Microdialysis vs. FSCV for Adenosine Measurement: A Comprehensive Technical Guide for Neuroscientists and Drug Developers
This article provides a comparative analysis of microdialysis and Fast-Scan Cyclic Voltammetry (FSCV) for measuring extracellular adenosine in the brain.
Microdialysis vs. FSCV for Adenosine Measurement: A Comprehensive Technical Guide for Neuroscientists and Drug Developers
Abstract
This article provides a comparative analysis of microdialysis and Fast-Scan Cyclic Voltammetry (FSCV) for measuring extracellular adenosine in the brain. Aimed at researchers and drug development professionals, it explores the foundational principles, methodological applications, and inherent limitations of each technique. The content details how methodological choices, from probe size to analytical parameters, critically impact data on adenosine's rapid neuromodulatory and neuroprotective roles. It further covers troubleshooting for common technical challenges and outlines validation strategies to ensure measurement specificity. By synthesizing key performance criteria—temporal resolution, spatial resolution, and invasiveness—this guide supports informed technique selection for advancing research in neurology and therapeutic development.
Understanding Adenosine Dynamics and the Imperative for Precise Measurement
The Critical Role of Adenosine as a Neuromodulator and Neuroprotector
Adenosine is a purine ribonucleoside that functions as a powerful endogenous neuromodulator and neuroprotector in the central nervous system. It operates through a complex receptor system, primarily including inhibitory A1 receptors (A1Rs) and facilitatory A2A receptors (A2ARs), which are differentially distributed throughout the brain [1]. Through these receptors, adenosine fine-tunes neuronal excitability, modulates neurotransmitter release, and integrates excitatory and inhibitory neurotransmission, positioning itself as a master regulator of brain homeostasis [1] [2]. Under pathological conditions, adenosine is released as part of the brain's endogenous protective mechanisms, with rapid increases observed during events such as seizures, ischemia, and traumatic brain injury [3] [4]. Understanding the precise dynamics of adenosine flux is therefore crucial for unraveling its neuroprotective functions and developing therapeutic strategies for neurological disorders.
The study of adenosine signaling presents significant methodological challenges due to its short half-life (8-15 seconds) and rapidly fluctuating extracellular concentrations [3]. This article provides a comparative analysis of the two primary techniques used to measure adenosine in the brain—microdialysis and fast-scan cyclic voltammetry (FSCV)—to equip researchers with the knowledge to select appropriate methodologies for investigating adenosine's critical roles in brain function and pathology.
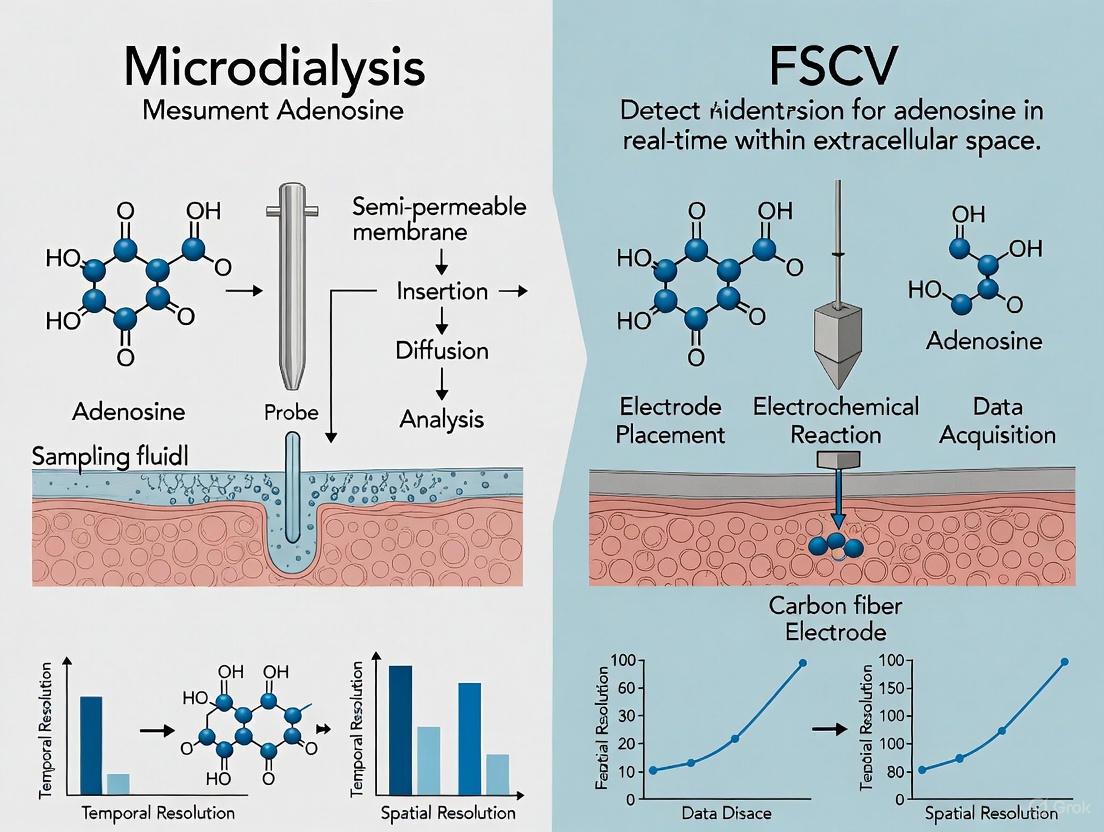
Methodological Comparison: Microdialysis vs. FSCV for Adenosine Measurement
Technical Foundations and Performance Characteristics
Table 1: Direct Comparison of Microdialysis and FSCV for Adenosine Measurement
| Feature | Microdialysis | Fast-Scan Cyclic Voltammetry (FSCV) |
|---|---|---|
| Temporal Resolution | Minutes (typically 2-10 min collection periods) [5] | Sub-second (10 measurements per second) [3] [5] |
| Spatial Resolution | Low (probes ~300 μm diameter, perfuse large areas) [6] | High (carbon fiber microelectrodes ~7 μm diameter) [6] [7] |
| Limit of Detection | ~5 nM [5] | Not explicitly quantified in results, suitable for micromolar fluctuations [6] |
| Key Advantage | Broad scope; multiplexed detection of many analytes simultaneously [6] | Real-time measurement of rapid adenosine dynamics during neural activity [3] |
| Primary Limitation | Cannot resolve rapid neurotransmitter release [3] | Limited to electroactive analytes; requires distinct electrochemical signature [7] |
| Tissue Impact | Significant tissue damage and foreign body response [6] | Minimal tissue damage; track often invisible via light microscopy [6] |
| Measurement Type | Indirect sampling of extracellular fluid [6] | Direct, real-time detection in extracellular space [5] |
| Ideal Application | Monitoring slow changes in basal adenosine levels (e.g., ischemia) [5] | Capturing rapid, transient adenosine release (e.g., seizure termination) [3] |
Practical Applications and Experimental Findings
The functional consequences of these methodological differences become evident when examining specific experimental applications. Microdialysis studies, for instance, have successfully measured peri-ictal adenosine increases of 6-31 fold three minutes after seizures in humans, revealing adenosine's role as an endogenous anticonvulsant [3]. However, this technique's temporal limitations obscure the precise relationship between adenosine dynamics and seizure events [3].
In contrast, FSCV provides a much finer-grained temporal view. In a swine model of epilepsy, simultaneous electrocorticography and FSCV demonstrated that adenosine rises by 260% compared to baseline approximately 2.6 seconds prior to electrographic seizure termination [3]. This precise timing suggests adenosine itself may be directly responsible for seizure cessation, a finding that was corroborated in human patients undergoing epilepsy surgery [3]. FSCV has also revealed a novel mode of rapid adenosine signaling that occurs over mere seconds, indicating that adenosine can operate on a time scale similar to classical neurotransmitters, not just as a slow neuromodulator [8] [5].
Experimental Protocols for Adenosine Measurement
FSCV Protocol for Rapid Adenosine Detection
Working Electrorode Preparation: Carbon fiber microelectrodes (CFMEs) are fabricated by aspirating a ~7μm carbon fiber into a 1.2 mm glass capillary tube, which is then pulled to a tapered point and sealed with epoxy resin [7]. The electrode is electrochemically pretreated to increase surface area and functional groups [7].
Waveform Application: For adenosine-specific detection, a triangular waveform is applied, scanning from a holding potential of -0.4 V to a switching potential of 1.45-1.5 V and back at a scan rate of 400 V/s, repeated at 10 Hz [5]. This waveform is optimized to detect adenosine's oxidation peaks while distinguishing it from interferents like ATP [5].
Signal Processing: The large background charging current is stable over time and is subtracted to reveal the faradaic current of adenosine oxidation [5]. Adenosine produces a characteristic cyclic voltammogram with two oxidation peaks—the primary at approximately 1.4 V and a secondary at 1.0 V—which serves as its electrochemical "fingerprint" [5].
In Vivo Measurement: The CFME is implanted into the target brain region with a Ag/AgCl reference electrode. The Wireless Instantaneous Neurochemical Sensor (WINCS) system can be used for simultaneous electrocorticography and electrochemical recording in large animal models or human patients [3].
Microdialysis Protocol for Adenosine Sampling
Probe Implantation: A microdialysis probe with a semi-permeable membrane (typically ~300 μm in diameter) is implanted into the target brain region [6]. Following implantation, a consistent wait time (often several hours) is observed before experimentation to allow stabilization of the system, as the initial tissue damage and foreign body response can affect analyte measurements [6].
Perfusion and Sample Collection: Artificial cerebrospinal fluid is perfused through the probe at a constant flow rate (typically 0.5-2 μL/min). Dialysate samples are collected from the outlet tubing over set intervals (usually 5-20 minutes), with the collection time determining the temporal resolution [6] [5].
Analytical Separation and Detection: Collected samples are typically analyzed using high-performance liquid chromatography (HPLC) to separate adenosine from other purines such as inosine, hypoxanthine, and uric acid [5]. Detection limits for adenosine can reach as low as 5 nM with appropriate analytical methods [5].
Adenosine Signaling Pathways and Measurement Workflows
Adenosine Neuroprotection Signaling Pathway
The following diagram illustrates the primary receptor-mediated mechanisms through which adenosine exerts its neuroprotective effects, highlighting the opposing roles of A1 and A2A receptors.
Experimental Workflow: FSCV vs. Microdialysis
This workflow compares the fundamental procedural differences between FSCV and microdialysis approaches for adenosine measurement, from initial setup to data output.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Research Reagent Solutions for Adenosine Research
| Item | Function & Application | Key Characteristics |
|---|---|---|
| Carbon Fiber Microelectrodes | Working electrode for FSCV measurements; detects adenosine oxidation current [3] [7] | ~7 μm diameter; minimal tissue damage; requires specific triangular waveform for adenosine [7] [5] |
| Adenosine Biosensors | Amperometric detection of adenosine via enzyme-coupled hydrogen peroxide production [3] [5] | Contain adenosine deaminase, purine nucleoside phosphorylase, and xanthine oxidase; not approved for human use [3] |
| Microdialysis Probes | Semi-permeable probes for sampling extracellular fluid from brain tissue [6] | ~300 μm diameter; causes significant tissue damage; enables broad analyte screening [6] |
| A1 Receptor Agonists (e.g., CPA) | Pharmacological activation of A1 receptors to study inhibitory neuromodulation [4] | Demonstrates anticonvulsant and neuroprotective effects in preclinical studies [1] [4] |
| A2A Receptor Antagonists (e.g., SCH 58261, KW-6002) | Pharmacological blockade of A2A receptors to study neuroprotection [1] [4] | Confers robust neuroprotection in adult animals; in clinical trials for Parkinson's disease [1] [4] |
| Adenosine Kinase Inhibitors | Increase extracellular adenosine by blocking major metabolic pathway [1] | Augments adenosine-mediated neuroprotection; potential therapeutic strategy [1] |
The comparative analysis of microdialysis and FSCV reveals that these techniques offer complementary, rather than competing capabilities for adenosine research. FSCV provides unprecedented temporal and spatial resolution for capturing rapid adenosine signaling dynamics, making it ideal for investigating moment-to-moment neuromodulatory functions, such as activity-dependent release and seizure termination [3] [5]. Conversely, microdialysis offers a broader analytical scope for monitoring basal adenosine levels and slower homeostatic changes, particularly in response to sustained challenges like ischemia [5]. The significant tissue damage associated with microdialysis implantation must be considered when interpreting results, whereas FSCV causes minimal disruption to the local cellular environment [6].
The choice between these methodologies should be guided by the specific research question. To fully elucidate adenosine's critical role as a neuromodulator and neuroprotector, researchers may increasingly employ these techniques in concert—using FSCV to capture rapid release and clearance kinetics while employing microdialysis to contextualize these events within slower metabolic and homeostatic processes. This integrated approach will advance our understanding of adenosine signaling across multiple temporal domains and facilitate the development of novel adenosine-based therapies for neurological disorders.
Key Technical Challenges in Measuring Extracellular Adenosine
The accurate measurement of extracellular adenosine is pivotal for advancing our understanding of its diverse roles in neuromodulation, neuroprotection, and immunoregulation [5] [9] [10]. The technical landscape is dominated by two primary methodologies: microdialysis and fast-scan cyclic voltammetry (FSCV). Each technique presents a unique profile of advantages and limitations, making the choice between them a fundamental consideration for researchers, scientists, and drug development professionals. This guide provides a comparative analysis of these core techniques, framing them within the context of a broader thesis on analytical approaches for adenosine measurement. By objectively comparing their performance against key technical challenges and providing supporting experimental data, this review aims to equip researchers with the information necessary to select the most appropriate method for their specific investigative goals.
Technical Comparison of Microdialysis and FSCV
The selection of an analytical technique dictates the temporal resolution, scope of analytes, and potential impact on the biological system under study. The following table provides a structured, quantitative comparison of microdialysis and FSCV.
Table 1: Technical Comparison of Microdialysis and Fast-Scan Cyclic Voltammetry (FSCV)
| Technical Parameter | Microdialysis | Fast-Scan Cyclic Voltammetry (FSCV) |
|---|---|---|
| Temporal Resolution | Minutes (typically 5-20 min sample collection) [5] | Sub-second (100 ms sampling rate) [5] [9] |
| Spatial Resolution | Low (probe diameter ~300 μm) [6] | High (carbon-fiber electrode diameter ~7 μm) [6] [5] |
| Limit of Detection | ~5 nM for adenosine [5] | Sub-micromolar for transient changes [5] |
| Analytical Scope | Broad (neurotransmitters, amino acids, peptides) [6] | Narrow (electroactive molecules only, e.g., DA, adenosine) [6] [5] |
| Tissue Damage | Significant (induces penetration injury/FBR) [6] | Minimal (track often invisible via light microscopy) [6] |
| Key Advantage | Ability to measure a wide range of non-electroactive molecules | Real-time tracking of rapid adenosine signaling events |
| Primary Limitation | Poor temporal resolution; significant tissue disruption | Limited chemical scope; signal confounded by interferents |
Experimental Protocols for Adenosine Measurement
Fast-Scan Cyclic Voltammetry (FSCV) for Adenosine
Objective: To detect rapid, transient changes in extracellular adenosine concentration in vivo with sub-second temporal resolution [9].
Workflow:
- Electrode Preparation: Carbon-fiber microelectrodes are fabricated by aspirating a single carbon fiber (diameter ~7 μm) into a glass capillary, which is then pulled to a fine tip and cut to an exposed length of 100-125 μm [9].
- Waveform Application: A triangular potential waveform is applied between the working carbon-fiber microelectrode and a Ag/AgCl reference electrode. For adenosine detection, the standard waveform scans from -0.4 V to 1.45 V or 1.50 V and back, at a rate of 400 V/s, repeated every 100 ms [5].
- Background Subtraction: The application of the waveform generates a large, stable background current. This current is digitally subtracted to reveal the Faradaic current from the oxidation of analytes, a process critical for identifying adenosine's electrochemical signature [5].
- Signal Identification: Adenosine is identified by its characteristic cyclic voltammogram (CV), which features two primary oxidation peaks. The largest peak appears near the switching potential at approximately 1.4 V, with a secondary oxidation peak at about 1.0 V [5].
- Data Visualization & Calibration: Data are often visualized in false color plots, where current is plotted against the applied potential and time. The electrode is calibrated post-experiment in a standard solution of known adenosine concentration (e.g., 1.0 μM) to convert oxidation current to concentration [5] [9].
Diagram 1: FSCV experimental workflow for adenosine detection.
Microdialysis for Adenosine
Objective: To sample and measure the basal concentration of adenosine and other small molecules from the extracellular fluid of a specific brain region over extended periods [6] [5].
Workflow:
- Probe Implantation: A microdialysis probe with a semi-permeable membrane (typical diameter ~300 μm) is surgically implanted into the target brain region (e.g., striatum) [6].
- Perfusion: Artificial cerebrospinal fluid (aCSF) is continuously perfused through the probe at a low, constant flow rate (typically 0.5 - 2.0 μL/min) [6].
- Equilibration & Sampling: Following implantation, a waiting period (often 60-180 minutes) is observed to allow the tissue to stabilize from the acute penetration injury [6]. After equilibration, dialysate is collected from the outlet tube into vials over defined intervals (e.g., 5-20 minutes).
- Sample Analysis: The collected dialysate samples are analyzed offline, typically using high-performance liquid chromatography (HPLC) to separate and quantify adenosine, often in conjunction with other purines like hypoxanthine and uric acid [5].
- Quantification: Adenosine concentration in the dialysate is quantified by comparing the sample's chromatographic peak to those of external standards of known concentration.
Diagram 2: Microdialysis workflow for measuring basal adenosine.
The Scientist's Toolkit: Research Reagent Solutions
Successful measurement of extracellular adenosine requires specific materials and reagents tailored to the chosen technique.
Table 2: Essential Research Reagents and Materials for Adenosine Measurement
| Item | Function/Description | Key Consideration |
|---|---|---|
| Carbon-Fiber Microelectrode [5] [9] | Working electrode for FSCV; 7 μm diameter enables high spatial resolution and minimal tissue damage. | Surface properties crucial for sensitivity and selectivity. |
| Microdialysis Probe [6] | Semi-permeable probe for sampling extracellular fluid; ~300 μm diameter. | Larger size causes significant tissue penetration injury. |
| Ag/AgCl Reference Electrode [9] | Provides a stable reference potential for electrochemical measurements in FSCV. | Essential for maintaining a consistent applied waveform. |
| Artificial CSF (aCSF) [6] | Physiological buffer used as perfusate in microdialysis. | Ionic composition must mimic the brain's extracellular environment. |
| SCH442416 [9] | Selective A2A adenosine receptor antagonist. | Used for pharmacological validation of adenosine's identity and function in FSCV studies. |
| Adenosine Standards [5] [9] | Solutions of known adenosine concentration for post-experiment calibration. | Critical for converting electrochemical current or chromatographic peak area to concentration. |
| HPLC System with UV/ECD [5] | Analytical instrument for separating and quantifying adenosine in microdialysis samples. | Provides the high sensitivity required for low nM detection limits. |
The choice between microdialysis and FSCV for measuring extracellular adenosine is not a matter of identifying a superior technique, but of aligning methodological strengths with specific research questions. This comparative analysis underscores that FSCV is unparalleled for investigating rapid, phasic adenosine signaling with high temporal and spatial fidelity, albeit with a narrower chemical scope. In contrast, microdialysis provides a broader biochemical profile of the extracellular environment, including non-electroactive molecules, but is constrained by its poor temporal resolution and significant impact on tissue integrity. For researchers and drug developers, this dichotomy means that studies focused on real-time neuromodulation or the dynamics of ischemic events [9] will benefit from FSCV, whereas investigations into tonic, basal adenosine levels or metabolic pathways are better served by microdialysis. A comprehensive understanding of the technical challenges inherent to each method is fundamental to designing rigorous experiments and generating reliable data in the complex field of adenosine research.
In the field of neuroscience and pharmacology, accurately measuring chemical concentrations in living tissues is paramount for understanding biological processes and drug actions. Two prominent techniques for in vivo sampling are microdialysis and fast-scan cyclic voltammetry (FSCV), each with distinct principles, capabilities, and limitations. Microdialysis is a minimally-invasive sampling technique that enables the continuous measurement of free, unbound analyte concentrations in the extracellular fluid of virtually any tissue [11]. Originally developed for monitoring neurotransmitters in the brain, its applications have expanded to encompass various tissues and research areas, including pharmacokinetics and environmental science [12] [13]. FSCV, an electrochemical method, provides real-time detection of electroactive substances with sub-second temporal resolution [8] [9]. This guide provides a comparative analysis of these techniques, focusing on their application in adenosine research, to inform researchers and drug development professionals in selecting the appropriate methodology for their specific experimental needs.
Historical Development and Fundamental Principles
Microdialysis: From Dialytrode to Quantitative Sampling
The conceptual foundation of microdialysis was laid in the early 1960s with the use of push-pull cannulas and implanted dialysis sacs in animal tissues [11]. The technique evolved significantly with the invention of the "dialytrode" in 1972, a device for long-term intracerebral perfusion in awake monkeys [11] [12] [14]. A major advancement came in 1974 with the development of the hollow fiber membrane, leading to the modern needle probe design [11]. Ungerstedt and colleagues further refined the probe design, enlarging the membrane surface area to improve sampling efficiency, which significantly contributed to the worldwide adoption of microdialysis, particularly for quantifying monoamines in neural tissue [15] [14].
The fundamental principle of microdialysis involves implanting a small probe with a semipermeable membrane into the tissue of interest [11]. The probe is perfused with an aqueous solution (perfusate) that closely mimics the ionic composition of the surrounding tissue fluid at low flow rates (typically 0.1-5 μL/min) [11]. Solutes below the membrane's molecular weight cutoff diffuse across the membrane along their concentration gradients, and the solution leaving the probe (dialysate) is collected for analysis [11]. This process allows for continuous monitoring of extracellular concentrations over periods ranging from hours to weeks [11] [12].
Fast-Scan Cyclic Voltammetry: Capturing Rapid Chemical Events
FSCV has emerged as a powerful complementary technique for monitoring electroactive neurotransmitters and neuromodulators, such as dopamine and adenosine, with high temporal resolution [6] [8]. This electrochemical method utilizes carbon-fiber microelectrodes that are significantly smaller than microdialysis probes (typically 7μm diameter), causing minimal tissue displacement [6]. FSCV operates by applying a rapid triangular waveform (e.g., from -0.4 V to 1.45 V and back at 400 V/s) to the microelectrode every 100 milliseconds [9]. As electroactive molecules oxidize and reduce at the electrode surface, they generate characteristic current profiles that enable both identification and quantification of the analyte [8] [16]. The technique's sub-second temporal resolution makes it ideal for capturing rapid chemical fluctuations, such as transient adenosine release events that last only a few seconds [8] [9].
Technical Comparison and Methodological Considerations
Comparative Technical Specifications
Table 1: Direct comparison of key technical characteristics between microdialysis and FSCV.
| Characteristic | Microdialysis | Fast-Scan Cyclic Voltammetry (FSCV) |
|---|---|---|
| Temporal Resolution | 1-30 minutes [14] | Sub-second (<100 ms) [8] [9] |
| Spatial Resolution | ~300 μm probe diameter [6] | 7 μm electrode diameter [6] |
| Analytical Scope | Broad (neurotransmitters, peptides, hormones, drugs) [11] [15] | Limited to electroactive compounds [6] |
| Invasiveness | Moderate (induces tissue trauma, foreign body response) [6] | Minimal (tissue track often invisible) [6] |
| Primary Measurements | Extracellular concentration (free, unbound fraction) [11] [13] | Real-time fluctuation dynamics [8] [9] |
| Key Advantage | Versatility in analyte detection [6] [15] | Excellent temporal resolution for rapid events [6] [8] |
Critical Methodological Protocols
Microdialysis Calibration Methods
Due to incomplete equilibrium between perfusate and extracellular fluid, microdialysis requires calibration to determine the recovery factor—the ratio between dialysate concentration and actual extracellular concentration [11] [12]. Several established methods exist:
- No-Net-Flux Method: The probe is perfused with at least four different analyte concentrations (Cin), and steady-state outlet concentrations (Cout) are measured. Recovery is determined by plotting (Cout - Cin) against Cin and calculating the slope of the regression line [11].
- Retrodialysis: The probe is perfused with an analyte-containing solution, and disappearance from the probe (Cin - Cout) is monitored. Recovery is calculated as (Cin - Cout)/Cin. For exogenous compounds, retrodialysis by drug is commonly used in clinical settings [11].
- Low-Flow-Rate Method: The probe is perfused at different flow rates, and extraction ratios are plotted against flow rates with extrapolation to zero flow [11].
FSCV Experimental Setup
FSCV for adenosine detection involves specific protocols:
- Electrode Preparation: Carbon-fiber microelectrodes (7μm diameter) are fabricated by aspirating a single carbon fiber into a glass capillary, pulled to a fine tip, and cut to 100-125μm length [9].
- Waveform Application: A triangular waveform from -0.4V to 1.45V and back at 400V/s is applied every 100ms against an Ag/AgCl reference electrode [9].
- Signal Processing: The large background current is subtracted, and electrodes are post-calibrated in known adenosine solutions (e.g., 1.0 μM) to convert current measurements to concentration values [9].
Figure 1: Adenosine Signaling Pathways and Detection Capabilities. This diagram illustrates how different modes of adenosine signaling are captured by FSCV versus microdialysis, leading to distinct but complementary functional interpretations.
Experimental Evidence: Adenosine Measurement Case Studies
Quantitative Comparison of Adenosine Measurements
Table 2: Experimental data from adenosine studies using microdialysis and FSCV under various physiological conditions.
| Experimental Condition | Technique | Adenosine Change | Temporal Dynamics | Reference |
|---|---|---|---|---|
| Cerebral Ischemia & Reperfusion | FSCV | 52% increase in transient frequency; 53% increase in cumulative concentration | Changes detected within seconds | [9] |
| Seizure Termination | FSCV | 260% increase compared to baseline | Elevation 2.6-7.5 seconds prior to electrographic termination | [17] |
| Permanent Focal Cerebral Ischemia | Microdialysis | Significant increase in extracellular levels | Changes measured over minutes to hours | [9] |
| Mechanical Stimulation | FSCV | Rapid transient release | Events lasting ~3 seconds | [9] |
Tissue Response and Methodological Limitations
Microdialysis-Induced Tissue Trauma
Microdialysis probe implantation (typically ~300μm diameter) causes significant tissue disruption compared to the minimal damage from FSCV microelectrodes (7μm diameter) [6]. This trauma triggers a foreign body response that evolves over hours to weeks, potentially compromising measurement accuracy [6]. Studies demonstrate that dopamine activity is significantly disrupted near microdialysis probes, with up to 90% reduction in evoked response amplitude when measured 200μm from the probe [6]. Consequently, most microdialysis protocols employ acute experiments within 24 hours of probe implantation to mitigate these effects [6].
FSCV Technical Limitations
While FSCV offers superior temporal resolution, it faces challenges including background current drift that typically limits continuous measurements to <90 seconds, though recent pre-conditioning protocols have extended this to 3 minutes [16]. Electrode fouling from protein adsorption and electrochemical byproducts can reduce sensitivity over time [16]. Additionally, FSCV is restricted to electroactive compounds, limiting its analytical scope compared to microdialysis [6].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key research reagents and materials for microdialysis and FSCV experiments.
| Item | Function/Application | Technical Specifications |
|---|---|---|
| Microdialysis Probes | Tissue implantation for solute sampling | 0.2-0.6mm diameter; 1-10mm membrane length; MW cut-off: 6kDa-3MDa [13] |
| Microdialysis Membranes | Molecular separation by size exclusion | Materials: cuprophane, PAES, PES, polyurethane, cellulose [13] |
| Carbon-Fiber Microelectrodes | Electrochemical detection for FSCV | 7μm diameter; 100-400μm active length [6] [9] |
| Perfusion Pumps | Controlled fluid delivery for microdialysis | Flow rates: 0.1-5μL/min (typical) [11] [13] |
| SCH442416 | A2A receptor antagonist for adenosine studies | 3mg/kg, i.p. administration in cerebral ischemia models [9] |
| Potentiostat | FSCV waveform application and current measurement | Capable of high scan rates (400V/s) and rapid sampling [9] |
Figure 2: Experimental Workflow Decision Tree. This flowchart provides a systematic approach for researchers to select the most appropriate technique based on their specific experimental requirements and the nature of their research question.
Microdialysis and FSCV represent complementary rather than competing approaches for adenosine measurement in neuroscience research. The selection between these techniques should be guided by specific experimental requirements: FSCV is optimal for capturing rapid, transient adenosine signaling with sub-second temporal resolution, particularly in studies of seizure termination, cerebral ischemia, and rapid neuromodulation [17] [9]. Microdialysis provides superior analytical versatility for measuring a broad range of compounds including nonelectroactive substances, enabling comprehensive profiling of extracellular environment changes over longer durations [11] [15]. Understanding their respective strengths, limitations, and methodological considerations allows researchers to make informed decisions, potentially employing these techniques in tandem to obtain a more complete understanding of adenosine dynamics in both normal physiological and pathological conditions.
Fast-scan cyclic voltammetry (FSCV) is an electroanalytical technique renowned for its exceptional temporal resolution in detecting neurotransmitters and neuromodulators within biological systems. Unlike traditional cyclic voltammetry with scan rates around 100 mV/s, FSCV operates at dramatically higher scan rates—up to 1×10⁶ V·s⁻¹—enabling the acquisition of complete voltammograms within milliseconds [18]. This capability makes FSCV particularly valuable for neuroscience research, where it allows real-time monitoring of neurochemical dynamics during behavioral tasks, pharmacological interventions, and physiological processes [19]. The technique's development and refinement have fundamentally advanced our understanding of chemical signaling in the brain, providing insights that were previously inaccessible with slower analytical methods.
The core principle underlying FSCV involves applying a rapid triangular voltage waveform to a microelectrode implanted in living tissue, driving the oxidation and reduction of electroactive molecules in the immediate environment [18]. The resulting current responses provide both quantitative and qualitative information about chemical concentrations and identities. When specifically applied to adenosine research, FSCV has revealed previously uncharacterized rapid signaling dynamics of this purinergic neuromodulator, demonstrating its transient inhibitory effects on other neurotransmitters and its potential role in seizure termination [8] [17]. This article explores the principles, historical context, and methodological applications of FSCV, with particular emphasis on its comparative advantages for adenosine detection relative to established techniques like microdialysis.
Historical Development of FSCV
The origins of FSCV trace back to foundational work in electrochemistry during the 1970s, when Ralph Adams pioneered the application of voltammetric methods to study oxidizable neurotransmitters in neural systems [19] [20]. Adams utilized carbon-paste electrodes to make the first voltammetric recordings of catecholamines in the lateral ventricle of anesthetized rats, demonstrating that neurochemicals could be quantified in situ without physically removing tissue [19]. These pioneering studies established that neural circuitry could remain intact during electrochemical measurements, preserving physiological relevance while obtaining chemical information.
A significant technological advancement came with the introduction of carbon-fiber microelectrodes (CFMEs) by Jean-Francois Pujol, which replaced the larger carbon-paste electrodes and minimized tissue damage while allowing faster scan rates due to reduced time constants [19]. The concurrent development of computer-controlled instrumentation enabled precise command of scan rates and rapid potential switching, facilitating the transition from slow analog voltammetry to digital FSCV [19]. A critical methodological breakthrough occurred in 1985 when Julian Millar introduced background-subtracted FSCV, recognizing that the stable background charging current could be subtracted to reveal faradaic currents resulting from rapid neurochemical changes [19]. This innovation enabled the high temporal resolution and sensitivity that define modern FSCV.
Mark Wightman subsequently popularized FSCV, particularly through his collaborations developing the technique for monitoring subsecond dopamine release in vivo [18] [20]. The convergence of carbon-fiber microelectrode technology, digital potentiostats, and background subtraction algorithms established FSCV as a powerful tool for neuroscience, allowing researchers to connect neurochemical dynamics with discrete behavioral events and pharmacological manipulations [19].
Table: Historical Milestones in FSCV Development
| Year | Development | Key Researchers | Significance |
|---|---|---|---|
| 1970s | First voltammetric measurements in brain tissue | Ralph Adams | Demonstrated feasibility of in situ neurochemical measurements |
| 1985 | Background-subtracted FSCV | Julian Millar | Enabled higher scan rates by eliminating charging current interference |
| 1980s-1990s | Carbon-fiber microelectrodes | Jean-Francois Pujol, Mark Wightman | Reduced tissue damage; enabled faster scan rates |
| 2000s-Present | Waveform optimization & multiplexing | Various groups | Expanded detectable analytes; improved selectivity |
Technical Principles and Methodologies
Fundamental Operating Principles
FSCV functions by applying a repetitive triangular waveform to a carbon-fiber microelectrode typically held at a potential that prevents faradaic reactions (often -0.4 V for dopamine detection) [21]. The voltage is rapidly ramped to a switching potential (typically +1.3 V) and back to the holding potential at scan rates of 400 V/s or higher, completing each cycle in approximately 10 ms when using a 10 Hz repetition rate [18] [21]. When the applied potential reaches the oxidation threshold for an electroactive species present at the electrode surface, electron transfer occurs, generating a measurable current. During the reverse scan, reduction of the oxidized species may occur, providing characteristic redox peaks that aid in chemical identification [21].
A defining feature of FSCV is the background charging current that results from the rapid voltage scanning. This current, which can be 10-100 times larger than faradaic currents from analytes, arises from capacitive effects at the electrode-electrolyte interface [21]. Since this background remains relatively stable between consecutive scans, it is subtracted to reveal the faradaic current attributable to analyte oxidation and reduction [19]. This background subtraction approach makes FSCV inherently differential, ideal for detecting concentration changes but unable to measure absolute basal levels without methodological modifications [21].
The voltammogram (current versus applied potential plot) provides characteristic signatures for different neurotransmitters based on their redox potentials. For example, dopamine exhibits oxidation peaks at approximately +0.6 V to +0.7 V and reduction peaks at approximately -0.2 V relative to a Ag/AgCl reference electrode [21]. These "electrochemical fingerprints" enable discrimination between different neurochemicals, particularly when combined with data analysis techniques like principal component regression [21].
Diagram Title: FSCV Electrochemical Process
Experimental Protocols for Adenosine Measurement
The detection of adenosine using FSCV requires specific methodological considerations. Researchers typically employ a specialized triangular waveform designed to encompass adenosine's redox potential, often scanning from -0.4 V to +1.5 V and back at 400 V/s or higher [8]. This waveform optimizes the electron transfer kinetics for adenosine while minimizing interference from other electroactive species. The experimental setup involves implanting a carbon-fiber microelectrode (diameter ~7μm) into the brain region of interest, along with a Ag/AgCl reference electrode and a stimulating electrode for evoked release studies [20].
In typical adenosine measurement protocols, baseline recordings establish stable background currents before experimental manipulations. Adenosine release can be evoked through various methods, including electrical stimulation, mechanical stimulation, or pharmacological challenges [8]. The recorded signals undergo background subtraction, and the resulting voltammograms are analyzed for characteristic adenosine features—oxidation peaks around +1.3 V to +1.4 V and reduction peaks near +0.8 V to +1.0 V [8]. Verification often includes pharmacological validation using receptor antagonists or enzyme inhibitors to confirm the identity of the measured species.
For ex vivo brain slice preparations, tissue is typically maintained in oxygenated artificial cerebrospinal fluid (ACSF) at physiological temperature [20]. The carbon-fiber microelectrode is positioned in the region of interest under visual guidance, and stimulation electrodes are placed to activate afferent pathways. Measurements of electrically evoked adenosine release provide information about the dynamics of purinergic signaling under controlled conditions that allow precise pharmacological manipulations [20].
Comparative Analysis: FSCV vs. Microdialysis for Adenosine Research
Technical Comparison
When evaluating FSCV against microdialysis for adenosine measurement, each technique demonstrates distinct advantages and limitations based on their underlying principles and methodological approaches.
Table: Technical Comparison of FSCV and Microdialysis for Adenosine Detection
| Parameter | FSCV | Microdialysis |
|---|---|---|
| Temporal Resolution | Subsecond (10-100 ms) [22] | Minutes (10-20 min) [6] |
| Spatial Resolution | Micrometer scale (7 μm electrodes) [6] | Millimeter scale (~300 μm probes) [6] |
| Detection Limit | ~1 nM for dopamine [18]; similar for adenosine | Low nanomolar range [6] |
| Invasiveness | Minimal tissue damage [6] | Significant tissue damage [6] |
| Chemical Specificity | Excellent for electroactive analytes [18] | Broad, but requires separation [6] |
| Measurable Analytes | Electroactive species only [18] | Extensive range including non-electroactive molecules [6] |
| Basal Level Measurement | Not possible without modifications [21] | Excellent for steady-state concentrations [6] |
Practical Research Applications
The complementary strengths of FSCV and microdialysis make them suitable for different research questions regarding adenosine signaling. FSCV has been particularly valuable in characterizing rapid adenosine signaling dynamics, such as the transient adenosine release that occurs within seconds of mechanical or electrical stimulation [8]. This rapid mode of adenosine release, undetectable by microdialysis, appears to modulate other neurotransmitters including dopamine and glutamate via A1 receptor activation [22]. For example, multiplexed measurements combining FSCV with genetically encoded sensors have demonstrated that adenosine exerts localized inhibitory effects on both dopamine and glutamate release within a 250 μm radius in the caudate putamen [22].
In epilepsy research, FSCV has revealed that extracellular adenosine concentrations increase significantly just prior to seizure termination (2.6-7.5 seconds before electrographic cessation), suggesting an active role for adenosine in seizure control [17]. These rapid dynamics would be completely missed by microdialysis sampling. Additionally, FSCV has been instrumental in characterizing activity-dependent adenosine release and its modulatory effects on oxygen dynamics and evoked dopamine release [8].
Conversely, microdialysis remains the preferred method for establishing basal adenosine concentrations and monitoring slow changes over extended periods [6]. The technique's ability to simultaneously measure multiple classes of compounds—including non-electroactive species—makes it valuable for comprehensive profiling of neurochemical changes under pathological conditions or in response to pharmacological treatments [6].
Advanced Applications and Current Research Directions
Multiplexed Sensing Approaches
Recent methodological advances have focused on multiplexing FSCV with complementary techniques to overcome its inherent limitation of detecting only electroactive species. Innovative approaches combine FSCV with genetically encoded fluorescent sensors, enabling simultaneous monitoring of electroactive and non-electroactive neurotransmitters [22]. For example, researchers have concurrently measured adenosine (via FSCV), dopamine (via FSCV), and glutamate (via iGluSnFR3.v857 fluorescent sensor) in brain slice preparations [22]. This integrated approach revealed coordinated neurotransmitter interactions, demonstrating that adenosine exerts transient inhibition on both dopamine and glutamate release through A1 receptor activation within a spatially constrained region (~250 μm) [22].
Other multiplexing strategies involve FSCV arrays with multiple carbon-fiber microelectrodes positioned in different brain regions to map neurochemical dynamics across neural circuits [22]. Additionally, enzyme-modified carbon-fiber microelectrodes expand FSCV's capabilities to non-electroactive analytes like glutamate, though these biosensors typically sacrifice some temporal resolution due to slower diffusion through enzymatic layers [22].
Waveform Optimization and Expanded Analytics
A significant research direction involves customizing triangular waveforms to enhance selectivity for specific analytes. While the traditional "dopamine waveform" (-0.4 V to +1.3 V) effectively detects catecholamines, modified potential windows improve detection of other substances including serotonin, histamine, and adenosine [18] [21]. For instance, the "sawhorse waveform" enables detection of neurotransmitters containing tyrosine residues [18]. These waveform optimizations consider several factors:
- Holding potential limitations by oxygen reduction [21]
- Switching potential constraints from water oxidation [21]
- Scan rate boundaries determined by electrode stability [21]
- Repetition rate limits imposed by adsorption processes [21]
Recent work has also explored carbon nanomaterial electrodes that exploit nanoscale structures to trap dopamine molecules, enhancing sensitivity through pre-concentration effects [21]. Additionally, methodological innovations like fast-scan controlled adsorption voltammetry (FSCAV) address FSCV's inability to measure basal neurotransmitter levels by incorporating controlled adsorption periods followed by redox scanning [23].
Diagram Title: FSCV Experimental Workflow
Essential Research Reagents and Materials
Successful implementation of FSCV requires specific instrumentation, electrodes, and chemical solutions optimized for neurochemical detection.
Table: Essential Research Reagents and Materials for FSCV
| Category | Specific Items | Function & Specifications |
|---|---|---|
| Electrodes | Carbon-fiber microelectrodes (7 μm diameter) [6] | Primary sensing element; minimal tissue damage |
| Ag/AgCl reference electrode [20] | Stable reference potential | |
| Bipolar stimulating electrode [20] | Evoked neurotransmitter release | |
| Instrumentation | Potentiostat (e.g., WaveNeuro, UNC UEI) [23] | Apply waveforms and measure currents |
| Data acquisition software (e.g., Demon Voltammetry) [20] | Signal processing and analysis | |
| Chemical Solutions | Artificial cerebrospinal fluid (ACSF) [20] | Physiological maintenance of tissue |
| 10x Krebs stock solution [20] | Electrochemical measurements | |
| Dopamine hydrochloride stock solution [20] | Electrode calibration | |
| Perchloric acid [20] | Solution preparation | |
| Specialty Items | Borosilicate capillary glass [20] | Electrode fabrication |
| Loctite 404 instant adhesive [20] | Component assembly |
FSCV represents a powerful analytical technique that has fundamentally advanced our understanding of rapid neurochemical dynamics, particularly in the realm of purinergic signaling. Its exceptional temporal resolution and chemical specificity have revealed novel aspects of adenosine signaling that were previously obscured by the limitations of slower techniques like microdialysis. The historical evolution of FSCV—from early voltammetric approaches to modern background-subtracted implementations with carbon-fiber microelectrodes—demonstrates how methodological innovations can unlock new biological insights.
While microdialysis maintains advantages for measuring basal concentrations and profiling multiple compound classes, FSCV provides unparalleled capacity to resolve subsecond neurochemical events. The ongoing development of multiplexed approaches, customized waveforms, and advanced electrode materials continues to expand FSCV's capabilities, promising further insights into the complex interplay between adenosine and other neurotransmitters in both health and disease. For researchers investigating rapid neuromodulatory processes, FSCV offers a uniquely powerful toolset that complements rather than replaces established neurochemical methods.
This guide provides a direct comparison of two primary techniques used in adenosine measurement for neuroscience research: Microdialysis and Fast-Scan Cyclic Voltammetry (FSCV). The analysis focuses on their core operational characteristics, strengths, and weaknesses to inform researchers and drug development professionals selecting methodologies for specific experimental needs.
Table 1: Core Characteristics of Microdialysis and FSCV
| Feature | Microdialysis | Fast-Scan Cyclic Voltammetry (FSCV) |
|---|---|---|
| Temporal Resolution | Minutes to hours [24] | Sub-seconds (< 1 second) [22] [24] |
| Spatial Resolution | Millimeters (mm) [24] | Micrometers (µm); discrete functional circuits [24] |
| Primary Measurement | Basal concentration [25] | Transient, stimulus-evoked release [24] |
| Key Strength | Broad, untargeted metabolite screening | Real-time monitoring of rapid neurotransmitter dynamics |
| Key Weakness | Poor resolution for transient signaling events | Inability to quantify basal concentrations [24] |
| Selectivity Mechanism | Physical separation via LC-MS/MS [25] | Electrochemical signature (cyclic voltammogram) [26] |
| Typical Sensitivity (LOD) | Low nanomolar (e.g., 0.02 nM) [25] | ~1 µM for enzyme-linked MEAs [24] |
Technique-Specific Experimental Protocols
Microdialysis with LC-MS/MS Detection
This protocol is adapted from methods used to quantify basal adenosine levels in murine brain microdialysates [25].
- Sample Collection: A microdialysis probe with a semi-permeable membrane is implanted in the brain region of interest (e.g., striatum or cortex) of an anesthetized or freely moving animal. The probe is perfused with a physiological solution, and analytes diffuse from the extracellular fluid into the dialysate, which is collected at timed intervals.
- Sample Preparation: A 20 µL dialysate sample is mixed with 5 µL of an internal standard (e.g., 50 nM 2-chloroadenosine) to control for variability in sample processing and analysis [25].
- LC-MS/MS Analysis:
- Chromatography: The sample is injected into a liquid chromatography (LC) system. A column-switching method is often employed to concentrate the sample and achieve high sensitivity, effectively separating adenosine from other compounds in the matrix.
- Mass Spectrometry: The eluent from the LC is directed into a tandem mass spectrometer (MS/MS) with an electrospray ionization (ESI) source operating in positive ion mode.
- Quantification: Adenosine is identified and quantified by monitoring its specific precursor ion (m/z 268) and a characteristic product ion (m/z 136) [25]. The internal standard (m/z 302) corrects for instrument drift.
FSCV with Enzyme-Linked Microelectrode Arrays (MEAs)
This protocol details the use of enzyme-based MEAs for real-time adenosine monitoring, offering a significant improvement in spatiotemporal resolution over traditional microdialysis [24].
- Microelectrode Array Preparation:
- A microelectrode array (MEA) with four platinum recording sites is used.
- The two distal sites are coated with an enzyme solution containing Adenosine Deaminase (ADA), Nucleoside Phosphorylase (NP), and Xanthine Oxidase (XO). This creates the adenosine-sensitive channels.
- The two proximal sites are coated with the same solution but without ADA. These serve as "sentinel" sites, detecting interfering substances and enzymatic breakdown products, but not adenosine itself [24].
- A size-exclusion layer (e.g., 1,3-phenylenediamine) is electroplated onto the sites to block interferents like ascorbic acid.
- In Vivo Measurement:
- The MEA is implanted into the target brain region.
- A constant potential (+0.7 V vs. Ag/AgCl) is applied to the recording sites.
- In the adenosine-sensitive sites, the enzyme cascade converts adenosine to uric acid, generating H₂O₂ as a reporter molecule. The H₂O₂ is oxidized at the platinum surface, producing a measurable current.
- The current from the sentinel sites is subtracted from the adenosine-sensitive sites, providing a selective and real-time measurement of extracellular adenosine concentration [24].
Diagram 1: Comparative experimental workflows.
The Scientist's Toolkit: Key Reagent Solutions
Table 2: Essential Reagents and Materials
| Item | Function/Description | Technique |
|---|---|---|
| 2-Chloroadenosine | Used as an internal standard (IS) during sample preparation to correct for analytical variability. | Microdialysis [25] |
| Adenosine Deaminase (ADA) | A key enzyme that catalyzes the conversion of adenosine to inosine, enabling its selective detection. | FSCV (Enzyme-linked MEA) [24] |
| 1,3-Phenylenediamine (mPD) | A size-exclusion polymer electroplated onto electrodes to block anionic interferents (e.g., ascorbic acid). | FSCV [24] |
| Tris Buffer | A standard physiological buffer (pH 7.4) used for in vitro calibrations and perfusions. | FSCV, Microdialysis |
| Nucleoside Phosphorylase (NP) & Xanthine Oxidase (XO) | Enzymes used in a cascade with ADA to ultimately convert adenosine to uric acid, producing a detectable H₂O₂ signal. | FSCV (Enzyme-linked MEA) [24] |
Performance Data & Head-to-Head Comparison
The quantitative performance of each technique reveals a clear trade-off between sensitivity and resolution.
Table 3: Quantitative Performance Comparison
| Performance Metric | Microdialysis + LC-MS/MS | FSCV (Enzyme-linked MEA) |
|---|---|---|
| Limit of Detection (LOD) | 0.02 nM (0.006 ng/mL) in mouse brain dialysate [25] | 0.96 µM in vitro; 0.04 µM in vivo [24] |
| Measured Basal Level | Low nanomolar range [25] | ~4.3 µM in rat cortex [24] |
| Linear Range | Not explicitly stated, but suitable for basal levels. | 0–15 µM (r² = 0.98) [24] |
| Temporal Resolution (Data Point) | Minutes per sample [25] | 4 Hz (MEA) [24]; can be <1 Hz for other FSCV [24] |
| Probe/Sensor Size | Large (mm-scale), causing significant tissue damage [24] | Small (µm-scale), suitable for discrete brain circuits [24] |
Diagram 2: Core strengths and weaknesses relationship.
Methodological Deep Dive: From Theory to Practical Application
Adenosine is a ubiquitous purinergic signaling molecule in the brain that regulates numerous physiological processes, including neurotransmission, blood flow, and the sleep-wake cycle, primarily through A1 and A2A receptor activation [22] [5]. Investigating its dynamics is crucial for understanding brain function, injury responses, and therapeutic development. This guide provides a comparative analysis of microdialysis and fast-scan cyclic voltammetry (FSCV) for adenosine measurement, detailing protocols and performance characteristics to inform researcher selection based on experimental needs.
Technical Comparison: Microdialysis vs. FSCV for Adenosine
The choice between microdialysis and FSCV represents a trade-off between temporal resolution and analytical scope. The table below summarizes key performance differences.
Table 1: Performance Comparison of Microdialysis and FSCV for Adenosine Measurement
| Feature | Microdialysis | Fast-Scan Cyclic Voltammetry (FSCV) |
|---|---|---|
| Temporal Resolution | Minutes (e.g., 1-1.5 min samples) [27] | Sub-second (100 ms) [5] [8] |
| Spatial Resolution | Low (hundreds of μm tissue damage) [6] | High (7-30 μm electrode diameter) [28] [6] |
| Measured Adenosine | Slower, tonic changes in basal levels [5] | Rapid, transient release events (seconds) [5] [8] |
| Analytical Scope | Broad (simultaneous sampling of many small molecules) [6] | Narrow (limited to electroactive species) [22] [6] |
| Tissue Impact | Significant (probe ~300 μm causes clear tissue trauma) [6] | Minimal (electrode track often invisible via light microscopy) [6] |
| Primary Data | Dialysate concentration (requires recovery calculation) | Direct extracellular concentration |
Microdialysis Protocol for Adenosine
Probe Selection and Configuration
Probe design significantly impacts recovery efficiency and experimental outcomes.
- Membrane Material and Configuration: Commercially available probes often use polyethersulfone (PES) or cuprophan membranes with a molecular weight cutoff (MWCO) of ~6 kDa in a concentric configuration [29]. Lab-made side-by-side configurations using regenerated cellulose (e.g., 13 kDa MWCO) are also used [29].
- Membrane Length: Standard active membrane lengths range from 1 mm to 4 mm [29] [27]. Shorter membranes are suitable for smaller brain regions but yield lower absolute recovery.
Table 2: Impact of Flow Rate on Microdialysis Recovery
| Flow Rate (μL/min) | Relative Recovery (%) | Sample Collection Interval | Application Context |
|---|---|---|---|
| 1-2 μL/min | Higher Recovery | 10-30 minutes | Measuring stable baseline levels |
| 2-4 μL/min | Moderate Recovery (e.g., 4-9%) [27] | 1-1.5 minutes [27] | Optimal for tracking dynamics |
Perfusate and Surgical Implantation
- Perfusate Composition: Use artificial cerebrospinal fluid (aCSF) to mimic ionic brain fluid. A standard recipe includes: 149 mM NaCl, 2.8 mM KCl, 1.2 mM CaCl₂, 1.2 mM MgCl₂, and 5.4 mM glucose, adjusted to physiological pH [29].
- Implantation and Equilibrium: Insert the probe into the target brain region using stereotaxic surgery. A post-implantation equilibrium period (typically 60-90 minutes) is critical for tissue stabilization before sample collection [27] [6].
HPLC Analysis of Adenosine
Analyze collected dialysate using HPLC with ultraviolet (UV) or mass spectrometry (MS) detection.
- HPLC System: Utilize microbore columns for enhanced sensitivity with minimal dead volume [27].
- Detection: UV detection is common, but LC-MS/MS provides superior specificity and lower detection limits for adenosine and its metabolites [29].
- In Vitro Recovery Calibration: Determine each probe's recovery rate before and after in vivo experiments by measuring adenosine diffusion from a standard solution into the perfusate [27]. This calibration is essential for estimating true extracellular concentrations.
Complementary Technique: FSCV for Adenosine
FSCV uses carbon-fiber microelectrodes (CFMEs) to detect electroactive molecules like adenosine with high spatiotemporal resolution.
FSCV Methodology and Electrode Design
- Electrochemical Detection: A triangular waveform (e.g., -0.4 V to 1.45 V vs. Ag/AgCl, 400 V/s) is applied to the CFME. Adenosine oxidizes irreversibly, producing characteristic peaks at ~1.4 V and ~1.0 V on the cyclic voltammogram for identification [5].
- Electrode Advancement: Standard 7 μm diameter CFMEs cause minimal tissue damage [6]. Recent work shows 30 μm cone-shaped CFMEs offer improved mechanical durability, higher sensitivity, and reduced glial activation, which is beneficial for chronic experiments [28].
Experimental Workflow for FSCV
The typical workflow for detecting adenosine with FSCV involves several key stages, from electrode preparation to data analysis.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for Adenosine Measurement Studies
| Item | Function/Description | Example Use |
|---|---|---|
| Microdialysis Probes | Semi-permeable membrane tip for solute sampling; concentric or side-by-side design [29]. | In vivo sampling of adenosine and other neurochemicals. |
| Artificial CSF (aCSF) | Ionic solution mimicking brain extracellular fluid; used as perfusate [29]. | Microdialysis perfusion fluid and in vitro recovery tests. |
| Carbon Fiber Microelectrode (CFME) | Miniaturized working electrode (7-30 µm diameter) for electrochemical detection [5] [28]. | FSCV detection of rapid adenosine transients. |
| HPLC with Microbore Column | Analytical system for separating and quantifying analytes in small-volume samples [27]. | Analysis of adenosine in collected microdialysate. |
| GRABAdo1.0m Sensor | Genetically encoded fluorescent sensor based on engineered GPCR [30] [31]. | Real-time imaging of adenosine dynamics via fiber photometry. |
| A1 Receptor Antagonist (DPCPX) | Pharmacological blocker of adenosine A1 receptors [22]. | Verifying receptor-specific mechanisms of adenosine action. |
Microdialysis remains the preferred method for broad-spectrum neurochemical sampling, including adenosine, over longer time courses. In contrast, FSCV is unparalleled for investigating the sub-second dynamics of rapid, transient adenosine signaling. Emerging technologies, particularly genetically encoded sensors like GRABAdo, are opening new frontiers for real-time adenosine imaging with high spatial resolution [30] [31]. Future advancements will likely involve the strategic multiplexing of these techniques, such as combining FSCV and fluorescent sensors, to simultaneously capture interactions between adenosine, dopamine, and glutamate, providing a more integrated view of neuromodulation in health and disease [22].
The measurement of adenosine, a ubiquitous neuromodulator in the brain, presents significant challenges due to its rapid signaling and low extracellular concentrations. Fast-scan cyclic voltammetry (FSCV) has emerged as a powerful technique for monitoring adenosine dynamics with high temporal resolution, offering distinct advantages over traditional methods like microdialysis. This guide provides a comparative analysis of FSCV methodologies for adenosine detection, focusing on waveform design, electrode fabrication, and data interpretation, framed within the broader context of adenosine measurement techniques. As research explores adenosine's role in regulating dopamine and glutamate release via A1 receptors [22], the demand for precise measurement techniques has intensified. This article objectively compares the performance of various FSCV approaches, supported by experimental data, to guide researchers and drug development professionals in selecting appropriate methodologies for their specific applications.
Fundamental Principles of Adenosine Detection with FSCV
Electrochemical Foundations
Fast-scan cyclic voltammetry detects electroactive neurotransmitters by applying a rapid, cyclic potential waveform to a carbon fiber microelectrode (CFME) and measuring the resulting current. Adenosine, like other electroactive analytes, undergoes oxidation and reduction at characteristic potentials, producing a unique voltammetric signature that allows for its identification and quantification [22]. The technique relies on the principle that different neurotransmitters oxidize or reduce at specific voltages, though this becomes challenging with structurally similar monoamines that share similar electrochemical properties [32].
Adenosine functions as a crucial neuromodulator in the brain, regulating processes including blood flow and the sleep-wake cycle through G protein-coupled receptors, primarily A1 receptors [22]. It exerts inhibitory effects on neurotransmitters such as dopamine and glutamate via these receptors, which limit adenyl cyclase formation and hyperpolarize neurons [22]. Understanding these modulatory effects requires techniques capable of capturing rapid neurochemical changes, which has led to the adoption of FSCV for adenosine research.
Comparative Advantage Over Microdialysis
When positioned within the broader methodology landscape for adenosine measurement, FSCV offers distinct advantages and disadvantages compared to microdialysis:
Table: Comparison of FSCV and Microdialysis for Adenosine Measurement
| Parameter | FSCV | Microdialysis |
|---|---|---|
| Temporal Resolution | Subsecond (100 ms) [22] | Minutes to hours [22] |
| Spatial Resolution | Extracellular space detection [22] | Macroscopic region sampling |
| Tissue Damage | Minimal with proper electrode design [33] | Significant due to larger probe size [22] |
| Analytical Specificity | Voltammetric signature identification | Chromatographic separation required |
| Real-time Monitoring | Excellent | Limited by sample collection and analysis |
| Simultaneous Analyte Detection | Limited to electroactive species [22] | Broad, including non-electroactive molecules |
While microdialysis can monitor multiple neurotransmitters simultaneously, its poor temporal resolution makes it unsuitable for capturing rapid adenosine transients, which occur on a timescale of seconds [22]. FSCV addresses this limitation with subsecond temporal resolution, enabling researchers to capture the rapid neuromodulatory effects of adenosine [22].
Electrode Fabrication and Design Considerations
Carbon Fiber Microelectrode Types and Performance
Electrode design critically influences the sensitivity, longevity, and biocompatibility of FSCV measurements. Recent advancements have focused on optimizing carbon fiber microelectrodes (CFMEs) to improve their performance for neurotransmitter detection, including adenosine.
Table: Comparison of Carbon Fiber Microelectrode Designs for Neurotransmitter Detection
| Electrode Type | Sensitivity (pA/µm²) | In Vivo Performance | Lifespan | Tissue Damage | Key Applications |
|---|---|---|---|---|---|
| 7 µm CFME (Standard) | 12.2 ± 4.9 [33] | 24.6 ± 8.5 nA dopamine signal [33] | Baseline | Minimal [33] | Acute measurements, phasic release |
| 30 µm Bare CFME | 33.3 ± 5.9 (2.7-fold increase) [33] | 12.9 ± 8.1 nA dopamine signal [33] | Moderate improvement | Significant tissue damage [33] | In vitro studies requiring high sensitivity |
| 30 µm Cone-Shaped CFME | Similar to 30 µm bare [33] | 47.5 ± 19.8 nA (3.7-fold improvement) [33] | 4.7-fold increase vs. 7 µm [33] | Reduced glial activation [33] | Chronic monitoring, long-term implantation |
Advanced Fabrication Techniques
The development of cone-shaped 30 µm CFMEs represents a significant advancement in electrode design. These electrodes are fabricated using electrochemical etching, where a direct current voltage of 10 V is applied to a 1 mm segment of carbon fiber submerged in Tris buffer [33]. During the 20-second etching process, a linear actuator moves the electrode upward at a constant speed, gradually exposing it to air and forming the desired cone shape with a final height between 100-120 µm [33]. This innovative geometry mitigates insertion-induced tissue damage while maintaining the mechanical robustness and sensitivity advantages of larger-diameter fibers.
Surface modifications further enhance electrode performance. Coatings such as PEDOT:Nafion have been shown to minimize the effects of in vivo biofouling and increase sensitivity to electroactive monoamine neurotransmitters [32]. These composite coatings combine the conductivity of PEDOT with the charge-selective properties of Nafion, improving both signal strength and specificity.
Waveform Design and Experimental Protocols
FSCV Waveforms for Adenosine Detection
FSCV employs specific voltage waveforms tailored to the electrochemical properties of target analytes. While standard FSCV waveforms for dopamine detection typically use a -0.4 V to +1.3 V sweep at 10 Hz [33], adenosine detection may require waveform optimization to maximize sensitivity and selectivity. The scan repetition rate is critical, with tonic concentration measurements often using slower rates (0.1 Hz) to allow for stable measurements [32], while phasic release measurements employ faster rates (10 Hz) to capture rapid transients.
Multiple cyclic square-wave voltammetry (M-CSWV) has emerged as a promising approach for measuring tonic concentrations of neurotransmitters [32]. This waveform applies a series of square-wave cycles at different potential ranges, enabling more stable measurements of baseline neurotransmitter levels compared to traditional FSCV. The development of N-shaped multiple cyclic square-wave voltammetry (N-MCSWV) and fast scan controlled-adsorption cyclic voltammetry (FSCAV) further expands the toolkit for tonic-level measurements [32].
Experimental Workflow for Multiplexed Adenosine Detection
The integration of FSCV with complementary techniques enables comprehensive investigation of adenosine's neuromodulatory effects. A representative experimental workflow for multiplexed detection of adenosine, dopamine, and glutamate involves:
This workflow enables researchers to investigate the spatial and temporal dynamics of adenosine neuromodulation. The expression of genetically encoded sensors via Sindbis viral vector provides rapid sensor expression within 18-24 hours [22], while CFME implantation allows simultaneous electrochemical detection. Local adenosine application (typically 30 seconds) reveals transient inhibitory effects, and A1 receptor blockade with DPCPX confirms the specific mechanism of action [22].
Data Interpretation and Analytical Approaches
Signal Processing and Discrimination Challenges
Interpreting FSCV data for adenosine detection involves several processing steps to extract meaningful neurochemical information from raw current measurements. Data processing typically includes background subtraction to isolate Faradaic currents from charging currents, filtering to reduce noise, and identification of characteristic oxidation and reduction peaks [33] [32]. The resulting voltammograms provide chemical information that can be used to identify and quantify neurotransmitters.
A significant challenge in adenosine detection is discriminating its signal from other electroactive species with similar oxidation potentials. This is particularly problematic in brain regions containing mixtures of structurally similar monoamines [32]. Traditional approaches rely on identifying unique voltammetric signatures through principal component analysis or other statistical methods, but these have shown limited success in resolving complex mixtures in the in vivo environment.
Advanced Computational Approaches
Deep learning algorithms have recently emerged as powerful tools for resolving concentrations of similar neurotransmitters from complex voltammetric data. DiscrimNet, a convolutional autoencoder, has demonstrated the ability to accurately predict individual tonic concentrations of dopamine, norepinephrine, and serotonin from both in vitro mixtures and the in vivo environment [32]. This approach significantly outperforms traditional shallow learning algorithms such as support vector regression (SVR), principal components regression (PCR), and partial least squares linear regression (PLSR) [32].
The DiscrimNet architecture combines labeled in vitro training data with unlabeled in vivo data to learn salient features that generalize well to new electrodes and experimental conditions [32]. This eliminates the need to retrain the model for each new electrode, facilitating broader application across research laboratories. The model has successfully predicted expected changes in dopamine and serotonin after cocaine and oxycodone administration in anesthetized rats, validating its utility for pharmacological studies [32].
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of FSCV for adenosine detection requires specific materials and reagents optimized for electrochemical measurements in biological systems.
Table: Essential Research Reagents and Materials for FSCV Adenosine Studies
| Item | Specification/Function | Application Notes |
|---|---|---|
| Carbon Fiber | AS4 (7 µm) for standard CFMEs; 30 µm for enhanced durability [33] | PAN-based fibers offer faster electron transfer kinetics [34] |
| Insulation Material | Silica tubing (ID = 20 µm, OD = 90 µm) [32] | Polyimide coating provides electrical insulation and mechanical support |
| Reference Electrode | Ag/AgCl in saline [32] | Essential for maintaining stable potential during FSCV measurements |
| Electrochemical Buffer | Tris buffer (pH 7.4) [33] | Provides stable electrochemical environment for in vitro characterization |
| Surface Coatings | PEDOT:Nafion composite [32] | Reduces biofouling and improves selectivity for cationic neurotransmitters |
| Adenosine Receptor Antagonists | DPCPX (8-cyclopentyl-1,3-dipropylxanthine) [22] | Selective A1 receptor antagonist for mechanism confirmation studies |
| Genetically Encoded Sensors | iGluSnFR3.v857 for glutamate [22] | Enables multiplexed detection with non-electroactive neurotransmitters |
| Data Acquisition System | NI USB-6363 (16-bit) [33] [32] | High-resolution analog-to-digital conversion for sensitive current measurements |
Adenosine Signaling Pathways and Neuromodulatory Mechanisms
Understanding adenosine's neurochemical effects requires knowledge of its signaling pathways and interactions with other neurotransmitter systems. Adenosine exerts its effects primarily through G protein-coupled receptors, with A1 receptors being particularly important for its neuromodulatory functions.
Adenosine released through mechanical stimulation or basal processes activates presynaptic A1 receptors, which subsequently limit adenyl cyclase formation and hyperpolarize neurons [22]. This leads to inhibited release of both dopamine and glutamate, with studies showing that adenosine's inhibitory effect operates within a constrained spatial range of approximately 250 μm [22]. The application of DPCPX, a selective A1 receptor antagonist, blocks this inhibitory effect, confirming the specific receptor mechanism involved [22].
FSCV methodology for adenosine detection has evolved significantly, with advancements in electrode design, waveform optimization, and data interpretation enhancing our ability to study this important neuromodulator. The development of cone-shaped 30 μm CFMEs addresses critical challenges in chronic monitoring by improving mechanical durability while minimizing tissue damage [33]. Multiplexing approaches that combine FSCV with genetically encoded fluorescence sensors enable simultaneous monitoring of multiple neurotransmitters, providing more comprehensive understanding of adenosine's neuromodulatory effects [22].
Future developments in FSCV for adenosine detection will likely focus on several key areas. First, the integration of advanced machine learning approaches like DiscrimNet will improve our ability to resolve adenosine signals from complex neurochemical mixtures [32]. Second, continued innovation in electrode materials and coatings will enhance sensitivity and longevity for chronic implantation studies. Finally, the translation of FSCV methodologies to clinical applications, particularly in closed-loop deep brain stimulation systems, represents an exciting frontier where real-time adenosine monitoring could optimize therapeutic outcomes for neurological disorders [33]. As these methodologies continue to mature, FSCV will remain an indispensable tool for unraveling the complex dynamics of adenosine signaling in the brain.
Adenosine is a ubiquitous neuromodulator in the brain, regulating critical functions such as sleep, blood flow, and neurotransmission. [22] Its signaling operates on distinct time scales: tonic signaling maintains stable, homeostatic levels over minutes to hours, while phasic signaling involves rapid, transient release events lasting only seconds. [5] [35] Understanding both modalities is essential for unraveling adenosine's dual role as a long-term homeostatic regulator and a rapid, activity-dependent neuromodulator.
The choice of measurement technique fundamentally shapes our understanding of these signaling modes. Fast-scan cyclic voltammetry (FSCV) and microdialysis represent two complementary approaches with divergent temporal resolutions and applications. This guide provides a comparative analysis of these methodologies, empowering researchers to select the optimal approach for their specific adenosine signaling questions.
Technical Comparison: Microdialysis vs. FSCV
The following table summarizes the core technical capabilities of microdialysis and FSCV for adenosine detection, highlighting their respective advantages for capturing tonic and phasic signaling.
Table 1: Technical Comparison of Adenosine Measurement Techniques
| Feature | Microdialysis | Fast-Scan Cyclic Voltammetry (FSCV) |
|---|---|---|
| Temporal Resolution | Minutes (5-10 minute sampling) [5] | Sub-second (100 ms) [5] |
| Spatial Resolution | Low (probe diameter ~300 μm) [6] | High (electrode diameter 7-30 μm) [33] [6] |
| Primary Signaling Mode | Tonic / basal concentrations [5] | Phasic / transient release [5] [35] |
| Limit of Detection | ~5 nM [5] | Sub-micromolar (e.g., detects ~0.04 μM transients) [35] |
| Tissue Damage | Significant (triggers foreign body response) [6] | Minimal (track often invisible via light microscopy) [6] |
| Key Advantage | Broad molecular profiling (e.g., neurotransmitters, metabolites) [6] | Real-time monitoring of rapid release and clearance kinetics [5] |
| Major Limitation | Inability to capture rapid, transient signaling events [5] | Limited to electroactive analytes; similar molecules can interfere [22] [5] |
Experimental Protocols for Adenosine Measurement
FSCV for Phasic Adenosine Detection
FSCV enables direct detection of adenosine with sub-second temporal resolution, making it ideal for capturing phasic signals. [5]
- Electrode Fabrication: Cylindrical carbon-fiber microelectrodes (CFMs) are fabricated using a single carbon fiber (diameter 7 μm) aspirated into a glass capillary. [32] [35] The fiber is trimmed to an exposed length of 50-100 μm. [32] [33] Electrodes are often coated with Nafion or carbon nanotubes to improve sensitivity and selectivity to cations like adenosine. [32] [35]
- Waveform Application: For adenosine detection, a triangular waveform is applied, scanning from -0.4 V to 1.45 V and back versus a Ag/AgCl reference electrode at a rate of 400 V/s. This scan is repeated every 100 ms. [5] [35]
- Data Collection & Processing: The resulting current is measured. A stable background current is subtracted to reveal the Faradaic signal of adenosine oxidation. The characteristic cyclic voltammogram for adenosine shows a primary oxidation peak at ~1.4 V. [5] Data is often visualized in false color plots, which show concentration changes over time. [5]
Microdialysis for Tonic Adenosine Levels
Microdialysis is used to sample the extracellular fluid to measure stable, tonic adenosine concentrations. [5] [6]
- Probe Implantation: A semi-permeable microdialysis probe (typically ~300 μm in diameter) is implanted into the brain region of interest. [6] Artificial cerebrospinal fluid (aCSF) is perfused through the probe at a low, constant flow rate (e.g., 0.2-2 μL/min). [5]
- Sample Collection: Molecules from the extracellular space diffuse across the membrane and are collected in the dialysate. Samples are typically collected over 5-10 minutes to obtain sufficient analyte volume for analysis. [5]
- Post-Hoc Analysis: Dialysate samples are analyzed offline, most commonly with High-Performance Liquid Chromatography (HPLC), to separate and quantify adenosine concentrations. [5]
Adenosine Signaling Pathways and Detection Workflows
Adenosine Receptor Signaling Pathways
Adenosine exerts its effects by binding to four G protein-coupled receptor (GPCR) subtypes, each with unique functions and distributions. [36] The diagram below illustrates the primary signaling pathways and their key roles.
Experimental Workflow for Multiplexed Sensor Measurements
Emerging approaches combine FSCV with genetically encoded sensors for a more comprehensive view of neuromodulation, as demonstrated in a 2025 study. [22] The workflow below outlines this multiplexed method for simultaneous adenosine, dopamine, and glutamate monitoring.
Essential Research Reagents and Materials
Successful investigation of adenosine signaling requires specific tools and reagents. The following table details key components for designing experiments.
Table 2: Essential Research Reagent Solutions for Adenosine Signaling Studies
| Reagent / Material | Function / Application | Example Specifications & Notes |
|---|---|---|
| Carbon Fiber Microelectrode (CFME) | Working electrode for FSCV; detects electroactive neurotransmitters. [5] [33] | 7 μm diameter standard; 30 μm cone-shaped variants improve longevity and biocompatibility. [33] |
| Adenosine Receptor Agonists/Antagonists | Pharmacological probes for receptor mechanism studies. [35] | DPCPX: A1 receptor antagonist (e.g., 6 mg/kg i.p.). CPA: A1 receptor agonist (e.g., 1 mg/kg i.p.). [35] |
| Genetically Encoded Fluorescent Sensors | Enable detection of non-electroactive neurotransmitters (e.g., glutamate). [22] | iGluSnFR3.v857: High-sensitivity glutamate sensor with fast kinetics, expressed via viral vectors (e.g., Sindbis). [22] |
| Triangular Waveform (FSCV) | Applied potential to oxidize/reduce adenosine for identification and quantification. [5] | Typical parameters: -0.4 V to 1.45 V, 400 V/s scan rate, 10 Hz repetition rate. [32] [5] |
| Microdialysis Probe | Semi-permeable probe for sampling extracellular fluid to measure tonic levels. [6] | ~300 μm diameter; requires post-hoc analysis (e.g., HPLC). [6] |
The comparative analysis of microdialysis and FSCV reveals a clear paradigm: technique selection dictates insight, with each method illuminating distinct facets of adenosine signaling. FSCV is unparalleled for capturing rapid, phasic adenosine transients that mediate moment-to-moment neuromodulation, while microdialysis provides a stable measure of tonic concentrations crucial for understanding homeostatic balance. [5] [35]
Future directions point toward multiplexed approaches, as exemplified by the combination of FSCV with genetically encoded sensors. [22] This powerful strategy allows researchers to simultaneously monitor adenosine alongside other neurotransmitters (e.g., dopamine, glutamate), providing a more integrated view of complex neuromodulatory interactions in real-time. Furthermore, technological innovations like cone-shaped, larger-diameter carbon fibers are enhancing the longevity and biocompatibility of chronic FSCV measurements, opening new possibilities for long-term studies of adenosine dynamics. [33] By leveraging the complementary strengths of these tools and embracing emerging methodologies, the scientific community can continue to decode the complex temporal language of adenosine signaling across health and disease.
Adenosine is a ubiquitous purinergic neuromodulator that regulates critical brain functions, including sleep, blood flow, and metabolic support [5] [37]. Its signaling occurs across multiple spatial and temporal scales—from rapid, localized transient release modulating specific synapses to slower, diffuse tonic signaling affecting broader brain regions [5] [38]. This spatial diversity presents a fundamental challenge for researchers: no single technique can optimally capture both local and global adenosine dynamics. Fast-scan cyclic voltammetry (FSCV) and microdialysis have emerged as complementary approaches, each with distinct strengths and limitations for spatial resolution. This guide provides a comparative analysis of these techniques, empowering researchers to select the appropriate methodology based on their specific spatial measurement requirements.
Technical Comparison: Microdialysis vs. FSCV
The selection between microdialysis and FSCV involves significant trade-offs between spatial resolution, temporal resolution, and analytical capabilities. The table below summarizes the core technical differences:
Table 1: Technical Comparison of Microdialysis and FSCV for Adenosine Measurement
| Feature | Microdialysis | Fast-Scan Cyclic Voltammetry (FSCV) |
|---|---|---|
| Spatial Resolution | Low (millimeter scale) [5] | High (micrometer scale) [5] |
| Temporal Resolution | Low (minutes) [5] | High (sub-second; 100 ms) [5] [22] |
| Measurement Type | Global, pooled sampling [5] | Local, point-source detection [5] |
| Primary Data | Absolute concentration (with recovery calibration) [39] | Relative concentration changes (current in nA) [5] |
| Key Strength | Measures absolute basal levels; identifies multiple analytes from one sample [5] [39] | Tracks rapid, phasic adenosine transients [5] |
| Major Limitation | Slow sampling misses rapid signaling; large probe size causes tissue disruption [5] [22] | Does not measure basal levels; primarily detects rapid concentration changes [5] |
| Typical LOD | ~5 nM [5] | Sub-micromolar (concentration changes) [5] |
Experimental Protocols for Adenosine Measurement
Microdialysis Protocol for Absolute Concentration Measurement
Microdialysis aims to determine the absolute concentration of adenosine in the extracellular fluid of a specific brain region.
- Probe Implantation: A semi-permeable microdialysis probe (typically several millimeters in length) is surgically implanted into the target brain region [5].
- Perfusate Collection: Artificial cerebrospinal fluid (aCSF) is perfused through the probe. Molecules from the extracellular fluid, including adenosine, diffuse across the membrane and are collected in the dialysate [5] [39].
- Sample Analysis: Dialysate aliquots are typically analyzed using High-Performance Liquid Chromatography (HPLC) to separate and quantify adenosine, often with detection limits as low as 5 nM [5].
- Critical Calibration Step: A key challenge is that the probe does not recover 100% of the analyte. The relative recovery of adenosine across the semipermeable membrane must be determined in vitro or in vivo to calculate the true absolute interstitial concentration [39].
FSCV Protocol for Transient Adenosine Dynamics
FSCV is designed to detect rapid, stimulus-evoked or spontaneous fluctuations in adenosine with high spatial and temporal precision.
- Electrode Preparation: Carbon-fiber microelectrodes (CFMEs) with a tiny exposed fiber tip (∼7 µm diameter, ∼100 µm length) are fabricated and preconditioned [5] [28].
- Waveform Application: The electrode is implanted into the tissue. A triangular waveform (e.g., scanning from -0.4 V to 1.45 V and back vs. a Ag/AgCl reference electrode at 400 V/s) is applied repeatedly at a high frequency (e.g., 10 Hz) [5].
- Electrochemical Detection: Adenosine is electroactive and undergoes oxidation at the electrode surface around 1.4 V, generating a measurable current proportional to its local concentration at that moment [5].
- Signal Processing: The stable background charging current is subtracted, revealing the faradaic current from adenosine oxidation. The resulting cyclic voltammogram provides a unique electrochemical signature that helps identify the analyte [5].
- Data Output: The result is a real-time trace of current changes (nA) that can be converted to relative concentration changes (µM) based on a post-calibration factor [5].
Visualizing Adenosine Signaling and Measurement Pathways
The following diagram illustrates the cellular sources and signaling pathways of adenosine, as well as the spatial domains accessed by different measurement techniques.
Adenosine Signaling and Measurement Dynamics
This diagram illustrates the two primary pathways of extracellular adenosine. Global tonic adenosine originates from the extracellular breakdown of ATP released from both neurons and astrocytes. It spreads diffusely, activating inhibitory A1 receptors and astrocytic A2B receptors to modulate overall brain state and metabolism [37] [38]. This pool is best measured by microdialysis. In contrast, local transient adenosine acts rapidly within specific synapses, primarily facilitating plasticity via A2A receptors [38]. This spatially restricted signaling is uniquely accessible to FSCV.
The Scientist's Toolkit: Essential Reagents and Materials
Successful measurement of adenosine requires specific reagents, tools, and biological models. The following table details key components of the experimental toolkit.
Table 2: Research Reagent Solutions and Essential Materials
| Item | Function/Description | Experimental Role |
|---|---|---|
| Carbon-Fiber Microelectrode (CFME) | 7 µm diameter carbon fiber sealed in a glass capillary [5] [28]. | FSCV working electrode for localized, high-temporal resolution detection [5]. |
| Microdialysis Probe | Semi-permeable membrane cannula (mm scale) [5]. | Sampler for pooling analytes from a larger tissue volume to measure global concentrations [5] [39]. |
| A2B Receptor Agonist (BAY 60-6583) | Selective pharmacological activator of adenosine A2B receptors [37]. | Tool to probe the specific role of astrocytic A2B receptors in metabolic signaling [37]. |
| A1 Receptor Antagonist (DPCPX) | Selective blocker of adenosine A1 receptors [22]. | Used to validate A1 receptor-mediated inhibitory effects on neurotransmitters like dopamine and glutamate [22]. |
| Ecto-5'-nucleotidase Inhibitor (α,β-methylene-ADP) | Inhibits the enzyme that converts AMP to adenosine [37]. | Used to probe the contribution of ATP-derived adenosine to the overall adenosine pool [37] [38]. |
| Adenosine Deaminase Inhibitor | Blocks the degradation of adenosine to inosine [39]. | A key component of "stop solution" for microdialysis to prevent analyte loss post-sampling [39]. |
The choice between microdialysis and FSCV is not a question of which technique is superior, but which is appropriate for the specific biological question. Researchers must align their tool selection with the spatial scale of the adenosine signaling they wish to study.
- Studying Global, Tonic Adenosine: For investigations of basal adenosine levels, their slow changes under pathological conditions like ischemia, or the simultaneous monitoring of multiple metabolites, microdialysis is the established and necessary method [5] [39]. Its ability to provide absolute concentration data is invaluable for pharmacokinetic and metabolic studies.
- Studying Local, Phasic Adenosine: For research focused on rapid, activity-dependent adenosine release, its transient modulatory effects on other neurotransmitters (e.g., dopamine, glutamate), and its actions within discrete synaptic environments, FSCV is the only technique that provides the requisite temporal and spatial resolution [5] [22].
The future of adenosine research lies in technological integration and innovation. Combining techniques, such as multiplexing FSCV with genetically encoded fluorescent sensors, allows for the simultaneous measurement of electroactive and non-electroactive signals, providing a more holistic view of neuromodulation [22]. Furthermore, advancements in electrode design, such as cone-shaped CFMEs, aim to improve biocompatibility and signal longevity for chronic studies [28]. By understanding the fundamental spatial considerations and the capabilities of each tool, researchers can effectively design experiments to unravel the complex roles of adenosine in health and disease.
The study of neurochemical signaling has been revolutionized by technologies that move beyond single-analyte measurement. Traditional methods like microdialysis provide a broad scope for detecting various substances but suffer from low temporal resolution and significant tissue disruption [6]. In contrast, fast-scan cyclic voltammetry (FSCV) offers excellent temporal resolution for electroactive analytes but remains limited in its ability to detect non-electroactive neurotransmitters simultaneously [22] [8]. The emerging paradigm of multiplexing FSCV with genetically encoded fluorescent sensors represents a transformative approach that overcomes these limitations, enabling researchers to capture complex, real-time interactions between multiple neurotransmitters and neuromodulators within intact neural circuits [22] [40]. This comparative analysis examines the technical capabilities, experimental applications, and performance characteristics of these integrated approaches within adenosine research, providing researchers with a framework for selecting appropriate methodologies based on specific experimental requirements.
Technical Comparison of Neurochemical Monitoring Approaches
Table 1: Comparison of Neurochemical Monitoring Techniques
| Technique | Temporal Resolution | Spatial Resolution | Key Detectable Analytes | Primary Limitations |
|---|---|---|---|---|
| Microdialysis | Minutes | Millimeters (probe diameter ~300 µm) | Broad range: DA, Glu, adenosine, peptides, amino acids [6] | Large probe size causes tissue damage; low temporal resolution [6] |
| FSCV | Sub-second (100 ms) | Micrometers (electrode diameter 7-30 µm) | Electroactive only: DA, adenosine [22] [8] | Limited to electroactive analytes; detects extrasynaptic spillover [22] |
| Fluorescent Sensors | Milliseconds | Synaptic level | Diverse: Glu, GABA, DA, adenosine, peptides [40] | No concentration profiles; photobleaching [22] |
| Multiplexed Approach | Sub-second | Synaptic to cellular | Combined: Simultaneous detection of electroactive and non-electroactive analytes [22] | Technical complexity; requires multiple instrumentation platforms [22] |
Table 2: Performance Characteristics in Adenosine Research
| Parameter | Microdialysis | FSCV | Multiplexed FSCV+Fluorescent |
|---|---|---|---|
| Adenosine Detection | Indirect via metabolites | Direct, rapid transients [8] | Direct with spatial context |
| Temporal Dynamics | Slow (minute scale) | Rapid (second scale) [8] | Simultaneous rapid and modulatory |
| Spatial Precision | Poor (tissue damage) [6] | Good (extracellular) | Excellent (synaptic to network) |
| Multiplexing Capacity | High (multiple analytes) [6] | Limited (electroactive only) | High (electroactive + non-electroactive) [22] |
Multiplexed Technical Approaches: Methodology and Workflow
Fast-Scan Cyclic Voltammetry (FSCV) for Electroactive Analytes
FSCV employs carbon fiber microelectrodes (CFMEs) typically ranging from 7-30 µm in diameter that are implanted into brain tissue to detect electroactive neurotransmitters such as dopamine and adenosine with sub-second temporal resolution [28] [8]. The technique applies rapid cyclic voltage sweeps (-0.4 to 1.3 V at 400 V/s) to the carbon fiber surface, causing oxidation and reduction of electroactive species adsorbed to the electrode [28]. The resulting currents are proportional to extracellular neurotransmitter concentrations, enabling precise quantification of transient release events [22]. Recent advancements in CFME design include cone-shaped 30 µm electrodes created through electrochemical etching, which demonstrate improved mechanical durability, 3.7-fold higher in vivo dopamine signals, and significantly reduced glial activation compared to conventional 7 µm CFMEs [28]. Standard FSCV experimental protocols involve preconditioning electrodes with a 1.5 V FSCV sweep, implementing a standard waveform (-0.4 to 1.3 V at 10 Hz), and processing data through background subtraction and principal component analysis to resolve individual neurochemical signals [28].
Genetically Encoded Fluorescent Sensors
Genetically encoded fluorescent sensors are engineered proteins that change their fluorescent properties upon binding specific neurochemicals, enabling real-time monitoring of neurotransmitter dynamics with high molecular specificity [40] [41]. These sensors typically utilize one of two primary design scaffolds: bacterial periplasmic binding proteins (PBPs) or G protein-coupled receptors (GPCRs) tagged with a fluorophore that activates after neuromodulator binding [22]. The readout mechanisms include (1) changes in fluorescence intensity, where sensors like iGluSnFR3.v857 and GCaMP utilize circularly permuted fluorescent proteins (cpFPs) that alter brightness upon conformational changes induced by ligand binding; (2) changes in FRET efficiency between two FPs; (3) translocation between cellular compartments; or (4) alterations in fluorescence spectral profiles [40] [41]. For adenosine research specifically, sensors have been developed based on modified GPCRs that emit fluorescence upon adenosine binding, enabling visualization of adenosine dynamics with high spatial resolution at the synaptic level [40].
Integrated Multiplexing Methodology
The multiplexed approach combines FSCV with fluorescent sensors in a complementary methodology that leverages the respective strengths of each technique [22]. A typical experimental workflow involves:
- Sensor Expression: Genetically encoded sensors (e.g., iGluSnFR3.v857 for glutamate) are expressed in target brain regions (e.g., caudate-putamen) using viral vectors such as Sindbis, which enables rapid sensor expression within 18-24 hours [22].
- Electrode Implantation: A CFME is implanted in the sensor-expressing region to monitor electrically stimulated dopamine and adenosine release [22].
- Stimulated Release: Electrical stimulation evokes neurotransmitter release, with FSCV detecting electroactive analytes (dopamine, adenosine) while fluorescence microscopy captures glutamate dynamics via the expressed sensors [22].
- Pharmacological Manipulation: Receptor-specific antagonists (e.g., DPCPX for A1 receptors) are applied to investigate modulation mechanisms [22].
- Spatial Mapping: Multiple measurements at varying distances from stimulation sites enable characterization of the spatial extent of neuromodulatory effects [22].
This integrated approach enables simultaneous monitoring of adenosine, dopamine, and glutamate, revealing complex interactions such as the inverse correlation between glutamate and dopamine release and the transient inhibitory effect of adenosine on both systems via A1 receptor activation within a constrained 250 µm range [22].
Experimental Protocols and Validation
Key Experimental Findings in Adenosine Modulation
Multiplexed approaches have yielded critical insights into adenosine neuromodulation, particularly regarding its spatial and temporal characteristics. Studies combining FSCV and iGluSnFR3 have demonstrated that local adenosine application produces transient inhibition of both electrically stimulated dopamine and glutamate release, with effects lasting approximately 30 seconds and complete recovery occurring within 10 minutes after adenosine administration [22]. This inhibition exhibits strict spatial confinement, observed only within a 250 µm radius from the application site, highlighting the regional nature of adenosine's modulatory effects [22]. Pharmacological validation using the A1 receptor antagonist DPCPX confirmed that adenosine's inhibition of both glutamate and dopamine release is mediated specifically through A1 receptor activation, indicating a coordinated modulatory mechanism rather than independent actions on each system [22]. Furthermore, these integrated measurements revealed an inverse spatial relationship between glutamate and dopamine release, with areas exhibiting high stimulated glutamate release displaying low stimulated dopamine release and vice versa, suggesting complex interactions between these neurotransmitter systems that were previously obscured when measured separately [22].
Technical Validation and Controls
Robust experimental protocols for multiplexed measurements incorporate several validation steps to ensure data reliability:
- Sensor Specificity Verification: Control experiments with receptor antagonists (e.g., DPCPX) confirm that observed effects are receptor-mediated rather than artifacts [22].
- Temporal Alignment: Precise synchronization of FSCV and fluorescence data acquisition systems enables accurate correlation of neurochemical dynamics across techniques [22].
- Spatial Mapping: Systematic measurements at varying distances from stimulation or drug application sites characterize the spatial extent of effects and validate measurement consistency [22].
- Electrode Performance Monitoring: Regular testing of CFME sensitivity and functionality ensures consistent detection performance throughout experiments [28].
Adenosine Signaling Pathways and Experimental Workflow
The diagram below illustrates the integrated experimental workflow for multiplexing FSCV with fluorescent sensors and the adenosine signaling pathway revealed through this approach.
Research Reagent Solutions for Multiplexed Neurochemical Monitoring
Table 3: Essential Research Reagents and Materials
| Reagent/Material | Function/Application | Example Specifications |
|---|---|---|
| Carbon Fiber Microelectrodes | FSCV detection of electroactive neurotransmitters | 7-30 µm diameter; cone-shaped tip for reduced tissue damage [28] |
| Genetically Encoded Sensors | Fluorescence detection of specific neurotransmitters | iGluSnFR3.v857 (glutamate); GRABDA (dopamine); iSeroSnFR (serotonin) [22] [40] |
| Viral Expression Vectors | In vivo delivery of sensor genes | Sindbis virus (rapid 18-24h expression) [22]; AAV vectors (long-term expression) |
| A1 Receptor Antagonists | Pharmacological blockade of adenosine signaling | DPCPX (8-cyclopentyl-1,3-dipropylxanthine) [22] |
| Data Acquisition Systems | Synchronized FSCV and fluorescence recording | Commercial FSCV systems (NI USB-6363) with custom LabVIEW software; fluorescence microscopes with appropriate filter sets [28] |
The multiplexing of FSCV with genetically encoded fluorescent sensors represents a significant advancement in neurochemical monitoring, offering researchers unprecedented capability to decipher complex neurotransmitter interactions in real-time within intact neural circuits. This integrated approach reveals coordinated neuromodulatory actions that remain invisible to single-technique methodologies, such as adenosine's simultaneous inhibition of dopamine and glutamate release via A1 receptors within spatially constrained domains [22]. While microdialysis continues to provide valuable broad-spectrum neurochemical analysis and standard FSCV maintains utility for specific electroactive analytes, the multiplexed paradigm enables a more comprehensive understanding of neural signaling dynamics across multiple neurotransmitter systems with superior temporal and spatial resolution. As fluorescent sensor technology continues to expand its color palette and specificity range [40] [41], and CFME designs evolve toward reduced tissue impact and chronic stability [28], this integrated approach will increasingly empower researchers to address fundamental questions in neuroscience and drug development with enhanced precision and analytical depth.
Troubleshooting Measurement Artifacts and Optimizing Data Quality
Mitigating Tissue Damage and the Foreign Body Response in Microdialysis
In vivo neurochemical monitoring provides critical insights into brain function and the efficacy of central nervous system (CNS) drugs. However, the implantation of monitoring devices, such as microdialysis probes, triggers a cascade of tissue trauma and a foreign body response (FBR) that can compromise data integrity [42] [6]. This response begins with immediate penetration injury upon probe insertion, followed by a persistent FBR characterized by inflammation, activation of immune cells, and the eventual formation of a fibrous capsule around the implant [43] [44]. This pathological FBR is driven by tissue-scale mechanical forces and is mediated by specific immune-cell signaling pathways, particularly through the Rac2-activated myeloid cells [44]. For researchers measuring nuanced signaling molecules like adenosine, this tissue disruption presents a significant challenge, as the very process of measurement can alter the neurochemical environment [5]. This guide provides a comparative analysis of microdialysis and fast-scan cyclic voltammetry (FSCV), focusing on their inherent propensity to cause tissue damage and the experimental strategies developed to mitigate these effects for reliable adenosine measurement.
Technical Comparison: Microdialysis vs. FSCV
The choice between microdialysis and FSCV involves a fundamental trade-off between analytical scope and minimal invasiveness. The following table outlines their core characteristics, with a particular emphasis on factors related to tissue damage and the FBR.
Table 1: Core Characteristics of Microdialysis and Fast-Scan Cyclic Voltammetry (FSCV)
| Feature | Microdialysis | Fast-Scan Cyclic Voltammetry (FSCV) |
|---|---|---|
| Basic Principle | Diffusion of analytes across a semi-permeable membrane into a perfusate [42] | Electrochemical oxidation and reduction of electroactive analytes at a carbon-fiber microelectrode surface [5] |
| Analytical Scope | Broad; any molecule smaller than the membrane cutoff (e.g., neurotransmitters, peptides, metabolites, drugs) [42] [6] | Narrow; limited to electroactive molecules (e.g., dopamine, adenosine, serotonin) [5] [6] |
| Temporal Resolution | Slow (minutes) [42] [5] | Very fast (sub-second; 100 ms) [5] [22] |
| Spatial Resolution | Low (probe membrane is several mm long) [42] | High (measures at a single point; electrode tip is ~7 µm in diameter) [5] [6] |
| Key Advantage | Unparalleled versatility for monitoring a wide range of neurochemicals [42] | Excellent temporal and spatial resolution for studying rapid neurotransmitter dynamics [5] [22] |
| Primary Invasiveness Concern | Large probe size (200-500 µm diameter) causes significant penetration injury and tissue disruption, triggering a pronounced FBR [42] [6] | Minimal tissue damage; the track of the carbon fiber is often invisible to light microscopy, causing negligible disruption [6] |
| Evidence of Tissue Impact | Creates a gradient of disrupted dopamine activity; electrically evoked release is abolished near the probe [42] [6] | Evoked dopamine responses are largely unaffected when measured with FSCV alone, confirming minimal tissue impact [6] |
Quantitative Data: Impact and Mitigation of the Foreign Body Response
The biological response to an implanted probe has quantifiable effects on measurement outcomes. The data below summarize key findings on the extent of tissue damage and the efficacy of mitigation strategies.
Table 2: Quantitative Evidence of Tissue Damage and Mitigation Efficacy
| Experimental Finding | Quantitative Data | Technique & Context |
|---|---|---|
| Gradient of Dopamine Disruption | Evoked dopamine response amplitude decreased by ~90% at a 200 µm distance from a microdialysis probe and was completely abolished at 0 µm separation [6]. | FSCV measurement near an implanted microdialysis probe in the rat striatum [6]. |
| Duration of Mitigation Effect | Retrodialysis of dexamethasone (DEX) preserved evoked dopamine release for up to 24 hours [45]. | DEX-enhanced microdialysis combined with FSCV validation in the rat striatum [45]. |
| Spatial Range of Adenosine Modulation | Transient adenosine application inhibited electrically stimulated dopamine and glutamate release only within a 250 µm radius [22]. | Multiplexed FSCV and genetically encoded sensors (iGluSnFR3.v857) in mouse brain slices [22]. |
| Key Molecular Driver of Pathological FBR | RAC2, a haematopoietic-specific Rho-GTPase, was significantly upregulated in severe human FBR (Baker-IV capsules) [44]. | Transcriptomic analysis of human fibrotic capsules from breast implants [44]. |
Experimental Protocols for Mitigating Tissue Damage
Dexamethasone-Enhanced Microdialysis
This protocol uses retrodialysis to deliver an anti-inflammatory agent directly to the probe track, mitigating the acute penetration injury and FBR [42] [45].
Methodology:
- Probe Implantation: Stereotactically implant a microdialysis probe (e.g., 220 µm outer diameter, 4 mm membrane length) into the target brain region (e.g., rat striatum or cortex) [42].
- DEX Perfusate Preparation: Prepare an artificial cerebrospinal fluid (aCSF) perfusate containing dexamethasone (DEX). A concentration of 10 µM is commonly used, delivered at a low flow rate (e.g., 0.5 µL/min) [42] [45].
- Retrodialysis Delivery: Begin perfusing the DEX-containing solution immediately after probe implantation. Evidence suggests that continuous delivery may not be necessary; perfusion for the first few hours post-implantation can provide benefits that last at least 10-24 hours [42] [45].
- Validation of Efficacy: The success of mitigation can be validated using side-by-side FSCV measurements of electrically evoked dopamine release, which should be preserved in tissue surrounding the DEX-treated probe, unlike untreated controls [42] [6].
Multiplexed FSCV and Fluorescence Sensors for Minimally Invasive Monitoring
This advanced protocol combines the high spatiotemporal resolution of FSCV with the specificity of optical sensors to study multiple neurotransmitters with minimal tissue disruption [22].
Methodology:
- Sensor Expression: Inject a Sindbis viral vector encoding a genetically encoded sensor (e.g., iGluSnFR3.v857 for glutamate) into the target brain region (e.g., caudate-putamen) of an animal model. Allow 18-24 hours for expression [22].
- Brain Slice Preparation: Prepare acute brain slices (e.g., 300-400 µm thick) from the injected animal and maintain in oxygenated aCSF [22].
- Multiplexed Measurement Setup: Implant a carbon-fiber microelectrode (CFME, ~7 µm diameter) into the slice within the region of sensor expression. Position the slice under a fluorescence microscope to monitor the optical sensor [22].
- Stimulation and Recording: Use electrical stimulation (e.g., a biphasic pulse, 300 µA, 2 ms per phase) to evoke neurotransmitter release. Simultaneously:
- Apply the FSCV waveform (e.g., -0.4 V to 1.45 V vs. Ag/AgCl at 400 V/s, repeated at 10 Hz) to detect electroactive transmitters like dopamine and adenosine [5] [22].
- Excite the fluorescence sensor with the appropriate wavelength and record emission changes to detect non-electroactive transmitters like glutamate [22].
- Pharmacological Manipulation: To investigate receptor mechanisms, bath-apply or locally puff-apply agents like an adenosine A1 receptor antagonist (e.g., DPCPX, 100 nM) to block the effects of endogenous or applied adenosine [22].
Signaling Pathways and Experimental Workflows
Key Signaling Pathway in the Foreign Body Response
The following diagram illustrates the molecular mechanism identified as a key driver of the pathological foreign body response to implants, which is critical for understanding the tissue environment around a microdialysis probe.
Diagram 1: FBR Signaling Pathway
Workflow for DEX-Enhanced Microdialysis
This workflow outlines the key steps for implementing the dexamethasone retrodialysis protocol to mitigate tissue damage.
Diagram 2: DEX Microdialysis Workflow
Workflow for Multiplexed FSCV & Fluorescence Sensing
This workflow shows the process for setting up a multiplexed experiment that minimizes invasiveness while allowing simultaneous measurement of multiple neurochemicals.
Diagram 3: Multiplexed Sensing Workflow
The Scientist's Toolkit: Essential Research Reagents & Materials
Successful implementation of these techniques requires specific reagents and materials. The following table lists key items for the featured experiments.
Table 3: Essential Research Reagents and Materials
| Item | Function / Application | Key Consideration / Rationale |
|---|---|---|
| Dexamethasone (DEX) | Potent anti-inflammatory glucocorticoid delivered via retrodialysis to mitigate FBR [42] [45]. | Local delivery avoids systemic effects; evidence suggests benefits persist after DEX perfusion stops [42]. |
| Carbon-Fiber Microelectrode (CFME) | Ultra-small working electrode (~7 µm diameter) for FSCV; causes minimal tissue damage [5] [6]. | The small size is crucial for high spatial resolution and negligible penetration injury compared to microdialysis probes [6]. |
| Artificial Cerebrospinal Fluid (aCSF) | Physiological perfusion fluid for microdialysis and maintenance of brain slices [42] [22]. | Ionic composition must mimic brain extracellular fluid to maintain tissue health and normal neurochemical signaling [42]. |
| iGluSnFR3.v857 | Genetically encoded fluorescent sensor for glutamate with high sensitivity and fast kinetics [22]. | Enables optical monitoring of a non-electroactive neurotransmitter with high spatiotemporal resolution when multiplexed with FSCV [22]. |
| 8-Cyclopentyl-1,3-dipropylxanthine (DPCPX) | Selective adenosine A1 receptor antagonist [22]. | Used for pharmacological validation to confirm that adenosine's effects are mediated via A1 receptors [22]. |
| Bovine Serum Albumin (BSA) / Human Serum Albumin (HSA) | Perfusate additive to reduce non-specific binding of hydrophobic drugs to microdialysis system components [46] [47]. | Critical for accurate measurement of lipophilic compounds; improves recovery by maintaining a concentration gradient [46] [47]. |
Identifying and Managing Electrochemical Interferences in FSCV
Fast-scan cyclic voltammetry (FSCV) has emerged as a premier technique for real-time monitoring of neurotransmitter dynamics with sub-second temporal resolution, enabling unprecedented insights into rapid neurochemical signaling events in the brain. [33] [22] This electrochemical method employs carbon-based microelectrodes to detect electroactive molecules, making it particularly valuable for studying neurotransmitters like dopamine and adenosine. [22] [48] However, a significant challenge complicating FSCV measurements is the presence of electrochemical interferences—substances that produce similar oxidation and reduction profiles to the target analytes, potentially leading to misinterpretation of neurochemical data. [32]
The interference problem becomes particularly acute when measuring adenosine, an important neuromodulator that regulates numerous brain functions and has been implicated in neuroprotection, sleep-wake regulation, and response to brain injury. [48] [49] Adenosine measurement is complicated by its electrochemical similarity to other purines and monoamines, as well as the complex metabolic pathways through which it is produced and degraded in the extracellular space. [49] Additionally, the very process of electrode implantation can trigger mechanosensitive release of adenosine, further complicating the interpretation of in vivo measurements. [48] This comparison guide examines the primary sources of interference in FSCV measurements, with particular focus on adenosine detection, and systematically evaluates the strategies researchers have developed to manage these challenges.
Electrochemical Interferences from Similar Neurotransmitters
The fundamental principle of FSCV—identifying molecules by their specific redox potentials—becomes challenging when measuring structurally similar neurotransmitters that oxidize or reduce at nearly identical voltages. [32] This is especially problematic in the in vivo environment, where mixtures of similar-appearing neurochemicals are abundant. [32] Dopamine and norepinephrine, for instance, exhibit significant overlap in their voltammetric signatures, making discrimination difficult in brain regions where both neurotransmitters are present. [32] Similarly, adenosine detection can be complicated by the presence of its metabolic precursors ATP and ADP, as well as other electroactive compounds with similar redox characteristics. [49]
Beyond interference from similar neurotransmitters, the electrode itself can contribute to background currents that obscure detection of target analytes. The carbon-fiber microelectrodes typically used in FSCV exhibit non-Faradaic charging currents that form the background against which neurotransmitter signals must be distinguished. [32] While background subtraction techniques can mitigate this issue, variability in electrode properties and performance can introduce additional sources of error and interference in measurements. [33]
Physiological and Metabolic Interferences
The metabolic pathway of adenosine production and degradation represents another significant source of potential interference in FSCV measurements. [49] Extracellular adenosine originates from multiple sources, including direct neuronal release and the rapid degradation of ATP released by both neurons and glial cells. [49] This means that measurements of adenosine fluctuations may reflect not only direct adenosine signaling but also ATP release and metabolism, complicating the interpretation of adenosine transients.
Mechanical stimulation from electrode implantation itself triggers adenosine release, which represents a significant methodological interference. [48] Studies have shown that simply lowering a carbon-fiber microelectrode 50 μm into brain tissue evokes transient adenosine release (averaging 0.8 ± 0.1 μM in brain slices and 3.3 ± 0.6 μM in vivo) that lasts approximately 18 seconds. [48] This mechanosensitive release is primarily activity-dependent and partially results from downstream breakdown of ATP, representing a significant confounding factor that must be considered when interpreting adenosine measurements obtained via FSCV. [48]
Table 1: Primary Sources of Interference in FSCV Adenosine Measurements
| Interference Category | Specific Examples | Impact on FSCV Measurements |
|---|---|---|
| Electrochemical Similarities | Dopamine, Norepinephrine, Serotonin | Overlapping voltammetric signatures complicate identification and quantification |
| Metabolic Precursors/Products | ATP, ADP, AMP, Inosine | Shared purine structure creates similar redox characteristics |
| Background Currents | Electrode charging currents, pH changes | Obscure detection of target analytes, require background subtraction |
| Physiological Confounders | Mechanosensitive release, Glial activation | Tissue response to electrode implantation alters neurochemical environment |
Electrode Fouling and Performance Degradation
A significant methodological interference in FSCV measurements comes from electrode fouling and performance degradation over time. Biofouling—the accumulation of proteins and other biomolecules on electrode surfaces—compromises sensor function by diminishing conductivity and blocking adsorption sites on the microelectrode surface. [34] This phenomenon is particularly problematic in chronic implantation scenarios where long-term measurements are desired. [33] The over-oxidation of carbon fibers during FSCV measurements further contributes to mechanical degradation and diminished electrode performance, creating an additional interference that must be managed for reliable measurements. [33]
The material properties of electrodes significantly influence their susceptibility to fouling. Traditional carbon-fiber microelectrodes often suffer from limited mechanical durability and reduced lifespan, hindering their use in chronic monitoring. [33] Even electrode geometry impacts tissue damage and subsequent glial activation, which can indirectly interfere with measurements by altering the local neurochemical environment. [33]
Comparative Analysis of Interference Management Strategies
Electrode Design and Material Innovations
Recent advances in electrode design and materials have yielded significant improvements in managing FSCV interferences. Conventional carbon-fiber microelectrodes (typically 7-10 μm in diameter) are increasingly being modified or replaced with novel designs that offer enhanced selectivity and reduced fouling. [33] [34]
Geometric modifications have proven effective in mitigating interference from tissue damage. Researchers have developed 30 μm diameter CFMEs with cone-shaped tips created via electrochemical etching, which demonstrated a 3.7-fold improvement in in vivo dopamine signals and significantly lower glial activation based on Iba1 and GFAP markers compared to unmodified 30 μm electrodes. [33] This geometric optimization reduced insertion-induced tissue damage, a common source of interference in in vivo measurements. [33]
Advanced carbon materials represent another promising approach. "All"-glassy carbon (GC) microelectrode arrays, in which both electrodes and interconnects are formed from a homogeneous GC layer, offer integrated chemical sensing while enhancing electrochemical durability by eliminating metal components. [50] These devices demonstrate exceptional electrochemical durability, enduring over 3.5 billion charge-balanced current pulses without failure. [50] Similarly, carbon-coated microelectrodes (CCMs) created through a unique low-temperature annealing process achieve high-performance voltammetry with extraordinary scalability and interoperability. [51] The mild annealing process (250°C for 1 hour) dramatically enhances electrochemical stability by reducing interlayer spacing from 4.0 to 3.7 Å and oxygen content from 15.9% to 8.7%, resulting in a coating highly resistant to water/ion infiltration that maintains stable performance. [51]
Surface coatings provide another strategy for interference management. Polymer coatings such as PEDOT:Nafion have been shown to minimize the effects of in vivo biofouling and increase sensitivity to electroactive monoamine neurotransmitters while excluding interfering substances. [32] These modified surfaces can enhance selectivity for target analytes while reducing interference from similarly electroactive compounds.
Table 2: Electrode-Based Interference Management Strategies
| Strategy | Mechanism of Action | Performance Advantages | Limitations |
|---|---|---|---|
| Cone-Shaped Geometry [33] | Reduces tissue damage during insertion | 3.7-fold improvement in in vivo signals; reduced glial activation | Specialized fabrication required |
| Carbon Coating with Annealing [51] | Stabilizes electrochemical interface | Low LOD (5 nM DA); high sensitivity (125.5 nA/μM); 100-channel scalability | Requires optimization of annealing parameters |
| PEDOT:Nafion Coatings [32] | Excludes interfering substances; reduces biofouling | Enhanced selectivity for monoamines; improved chronic stability | Potential compromise of temporal resolution |
| Glassy Carbon MEAs [50] | Eliminates metal components; homogeneous structure | Exceptional durability (>3.5 billion pulses); stable baseline | Complex fabrication process |
Waveform Optimization and Computational Approaches
Beyond physical electrode modifications, researchers have developed sophisticated waveform designs and computational approaches to discriminate between neurotransmitters and manage interferences.
Multiple Cyclic Square Wave Voltammetry (M-CSWV) has emerged as a valuable alternative to traditional FSCV for resolving tonic concentrations of neurotransmitters. [32] This approach enables measurement of tonic-level release of neurotransmitters, which involves pacemaker-like spontaneous firing of neurons that periodically release neurotransmitters into the extrasynaptic space. [32] Unlike phasic measurements that capture burst-firing events, M-CSWV targets the homeostatic imbalances that are increasingly recognized as important factors in neuropsychiatric disorders. [32]
Deep learning algorithms represent a cutting-edge approach to interference management. DiscrimNet, a convolutional autoencoder, has demonstrated remarkable capability in accurately predicting individual tonic concentrations of dopamine, norepinephrine, and serotonin from both in vitro mixtures and the in vivo environment in anesthetized rats. [32] This computational approach vastly outperforms traditional shallow learning algorithms (such as principal components regression, partial least squares linear regression, and support vector regression) that have previously been used for neurotransmitter discrimination. [32] The model generalizes well to data captured from electrodes unseen during model training, eliminating the need to retrain the model for each new electrode—a significant advantage for practical applications. [32]
Multiplexed sensing approaches that combine FSCV with other measurement techniques provide additional dimensionality to help resolve interferences. For example, researchers have successfully multiplexed FSCV with genetically encoded fluorescent sensors (iGluSnFR3.v857) to simultaneously monitor adenosine, dopamine, and glutamate. [22] This approach revealed that transient adenosine regulates dopamine and glutamate release via A1 receptors, exerting a regional inhibitory effect within a 250 μm radius in the caudate putamen. [22] By providing simultaneous measurement of multiple neurotransmitters, this multiplexing approach helps contextualize adenosine measurements and distinguish true adenosine signaling from interference.
Table 3: Performance Comparison of Discrimination Algorithms for FSCV
| Algorithm | Discrimination Principle | Applications | Advantages | Limitations |
|---|---|---|---|---|
| DiscrimNet (Deep Learning) [32] | Convolutional autoencoder architecture | Tonic concentration resolution of DA, NE, 5-HT | High accuracy in predicting in vivo concentrations; generalizes to new electrodes | Requires substantial training data; complex implementation |
| Principal Components Regression (PCR) [32] | Dimensionality reduction + regression | Phasic neurotransmitter release | Simpler implementation; established methodology | Limited success with in vivo mixtures |
| Partial Least Squares Regression (PLSR) [32] | Latent variable modeling | Phasic neurotransmitter release | Handles correlated variables well | Limited extension to tonic concentrations |
| Support Vector Regression (SVR) [32] | Kernel-based nonlinear regression | Phasic neurotransmitter release | Effective for nonlinear relationships | Suboptimal for complex mixtures |
Experimental Protocols for Key Studies
Protocol for Assessing Mechanically Evoked Adenosine Release
The characterization of mechanically evoked adenosine release provides a valuable case study in identifying and controlling for methodological interferences in FSCV:
Electrode Fabrication: Carbon-fiber microelectrodes were fabricated from T-650 carbon fibers as previously described, creating cylinder electrodes 50 μm long and 7 μm in diameter for use with FSCV. [48]
Stimulation Paradigm: After equilibration, 60 seconds of baseline data was collected, and the brain slice was mechanically stimulated by lowering the electrode 50 μm with a micromanipulator. [48]
Pharmacological Validation: To characterize the mechanisms of release, experiments included:
- Calcium-free conditions (aCSF without CaCl2 + 1 mM EDTA)
- Sodium channel blockade (0.5 μM tetrodotoxin, TTX)
- AMPA receptor antagonism (10 μM CNQX)
- NTDPase inhibition (POM-1 to assess ATP metabolism contribution)
- ENT transporter inhibition (NBTI) [48]
Measurement Conditions: FSCV was performed using a triangular waveform scanning from -0.4 V to 1.5 V and back at 400 V/s against an Ag/AgCl reference electrode. [48]
This protocol revealed that mechanically evoked adenosine is primarily activity-dependent and partially results from downstream breakdown of ATP, highlighting important considerations for distinguishing true adenosine signaling from artifact in FSCV measurements. [48]
Protocol for Cone-Shaped Electrode Fabrication and Testing
The development of cone-shaped electrodes illustrates an effective approach to mitigating interference from tissue damage:
Electrode Fabrication: 30 μm diameter carbon fibers were electrochemically etched using a direct current voltage of 10 V applied to a 1 mm segment of carbon fiber submerged in Tris buffer. [33] The carbon fiber underwent electrolysis for 20 seconds, resulting in partial erosion and detachment. A linear actuator moved the electrode upward at a constant speed during etching, gradually exposing it to air and forming the desired cone shape with a final height controlled between 100-120 μm. [33]
Performance Validation:
- In vitro sensitivity measurements in dopamine solutions
- In vivo dopamine detection in animal models
- Longevity assessment via erosion tests
- Biocompatibility evaluation through immunofluorescence analysis of brain tissue for Iba1 and GFAP markers [33]
Results: This geometric modification resulted in a 3.7-fold improvement in in vivo dopamine signals and significantly lower glial activation, with erosion tests revealing a 4.7-fold increase in lifespan compared to conventional 7 μm CFMEs. [33]
Protocol for Deep Learning Discrimination of Neurotransmitters
The implementation of DiscrimNet provides a computational approach to resolving interferents:
Electrode Preparation: Carbon fiber microelectrodes were fabricated using a standardized design with a single carbon fiber (AS4, diameter = 7 μm) inserted into a silica tube. [32] Electrodes were coated with PEDOT:Nafion deposition solution to minimize biofouling effects and increase sensitivity. [32]
Data Collection:
- In vitro data: M-CSWV measurements in solutions of DA, NE, and 5-HT at various concentrations
- In vivo data: Collation from prior experiments in anesthetized rats (nucleus accumbens, dorsal hippocampus, medial prefrontal cortex) [32]
Model Training: DiscrimNet was trained using both labeled in vitro data and unlabeled in vivo data, employing a convolutional autoencoder architecture to learn salient features for discrimination. [32]
Validation: Model performance was assessed by predicting neurotransmitter concentrations after pharmacological interventions (cocaine and oxycodone administration) and comparing against shallow learning algorithms. [32]
Signaling Pathways and Experimental Workflows
The accurate interpretation of FSCV data requires understanding the complex signaling pathways that regulate extracellular adenosine dynamics. The following diagram illustrates key pathways involved in adenosine release and metabolism that can contribute to interference in measurements:
Adenosine Signaling Pathways in FSCV
The experimental workflow for multiplexed FSCV and fluorescence measurements reveals how integrating multiple techniques can help resolve interferents:
Multiplexed FSCV Workflow
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key Research Reagents and Materials for FSCV Interference Management
| Item | Function/Role | Example Applications | Performance Considerations |
|---|---|---|---|
| Carbon Fiber Microelectrodes [33] [34] | Electrochemical sensing of neurotransmitters | Detection of DA, adenosine, and other electroactive analytes | 7-10 μm standard diameter; 30 μm for enhanced durability |
| PEDOT:Nafion Coating [32] | Surface modification to reduce biofouling | Chronic implantation studies; enhanced selectivity | Minimizes interference from anionic species; improves stability |
| iGluSnFR3.v857 Fluorescent Sensor [22] | Genetically encoded glutamate sensor | Multiplexed measurements with FSCV | High specificity for synaptic glutamate; fast binding kinetics |
| POM-1 (NTDPase Inhibitor) [48] | Inhibits ectonucleotidases | Distinguishing ATP-derived vs. direct adenosine release | Reduces adenosine from ATP metabolism by ~50% |
| DPCPX (A1 Receptor Antagonist) [22] | Blocks adenosine A1 receptors | Validating adenosine-mediated effects | Confirms A1 receptor involvement in observed modulation |
| TTX (Tetrodotoxin) [48] | Sodium channel blocker | Assessing activity-dependent release | Differentiates exocytotic vs. non-exocytotic release mechanisms |
| Tris Buffer [33] | Electrochemical medium | In vitro calibrations and testing | Maintains electrochemical stability and signal consistency |
The systematic management of electrochemical interferences in FSCV requires a multifaceted approach that addresses both technical and biological sources of confounding signals. Electrode design innovations—including geometric optimization, advanced carbon materials, and selective coatings—provide powerful tools for reducing interference at the source. Simultaneously, computational approaches like deep learning enable unprecedented discrimination between neurotransmitters with similar electrochemical signatures, while multiplexed sensing strategies provide additional dimensions of data to contextualize measurements.
For adenosine measurement specifically, researchers must remain cognizant of the complex metabolic pathways and multiple release mechanisms that contribute to extracellular adenosine dynamics. The demonstrated phenomenon of mechanically evoked adenosine release highlights the importance of controlling for methodological artifacts, particularly in studies investigating physiological adenosine signaling. As FSCV continues to evolve toward higher-density arrays, chronic implantation, and eventual human translation, effective interference management will remain paramount for extracting meaningful neurochemical information from complex biological environments.
Future developments will likely focus on the integration of multiple interference management strategies—combining optimized electrode designs with advanced computational analytics and multiplexed sensing modalities. Such integrated approaches promise to further enhance the selectivity and reliability of FSCV measurements, unlocking new possibilities for understanding complex neurochemical processes in both health and disease.
The accurate measurement of dynamic neurochemical processes requires meticulous optimization of sampling techniques. For adenosine research, the choice between microdialysis and fast-scan cyclic voltammetry (FSCV) represents a critical methodological crossroads, with probe design and flow parameters fundamentally dictating the temporal and spatial resolution achievable. This guide provides a comparative analysis of these technologies, focusing specifically on how flow rate and membrane surface area affect analyte recovery, with implications for experimental design in neurotransmitter research and drug development.
Monitoring adenosine signaling presents distinct challenges due to its rapid, transient release patterns and complex modulatory roles in processes ranging from neurotransmission to neuroprotection [5]. Two primary techniques have emerged for in vivo adenosine measurement: microdialysis and fast-scan cyclic voltammetry (FSCV). These methods operate on fundamentally different principles:
- Microdialysis utilizes a semi-permeable membrane implanted in tissue, through which a perfusion fluid is circulated. Analytes diffuse across the membrane according to concentration gradients, with recovery inversely related to flow rate and directly related to membrane surface area [52].
- Fast-Scan Cyclic Voltammetry (FSCV) employs carbon-fiber microelectrodes (CFMEs) that directly detect electroactive species via rapid potential scans, offering subsecond temporal resolution but limited to electroactive analytes like adenosine [21] [5].
The optimization of recovery parameters—flow rate and membrane surface area—is paramount for data quality in microdialysis, while FSCV optimization focuses on electrode design and waveform parameters.
Theoretical Foundations: The Recovery Principles
The Mathematics of Microdialysis Recovery
In microdialysis, the extraction efficiency of an analyte is governed by its diffusion from the extracellular fluid into the perfusion stream. The relative recovery (RR)—the ratio of analyte concentration in the dialysate to its concentration in the external medium—is quantitatively influenced by both flow rate and membrane surface area.
Relative Recovery (%) = f(Flow Rate, Membrane Surface Area, Diffusion Coefficient)
The relationship is characterized by:
- Inverse correlation with flow rate: Lower flow rates permit greater diffusion time, thereby increasing relative recovery but at the cost of extended temporal resolution and reduced sample concentration.
- Direct correlation with membrane surface area: Larger surface areas provide greater area for analyte exchange, enhancing recovery but increasing tissue displacement and damage.
The Spatial Considerations of Probe Size
The foreign body response triggered by probe implantation creates a gradient of disrupted neurochemical activity around the probe track. Studies comparing FSCV measurements at varying distances from implanted microdialysis probes have demonstrated that evoked dopamine response amplitude decreases by approximately 90% at a 200 μm separation compared to 1 mm [6]. This indicates that larger probes not only cause more initial damage but also create a larger zone of compromised neurochemical activity, potentially skewing concentration measurements.
Figure 1: Impact Pathway of Probe Implantation. This diagram illustrates how probe insertion triggers a cascade of tissue responses that ultimately affect recovery measurements, with key influencing factors shown in green.
Experimental Data & Comparative Performance
Quantitative Comparison of Microdialysis Systems
Table 1: Performance characteristics of conventional versus microfabricated microdialysis probes
| Parameter | Conventional Probe | Microfabricated Probe | Impact on Recovery |
|---|---|---|---|
| Probe Diameter | ~220 μm [6] | 45 μm thick × 180 μm wide [52] | 79% reduction in cross-sectional area minimizes tissue damage [52] |
| Membrane Length | Typically 1-4 mm | 4 mm [52] | Maintains surface area for exchange despite smaller footprint |
| Typical Flow Rate | 1-2 μL/min | 100 nL/min [52] | Lower flow rate increases relative recovery |
| Relative Recovery | Varies by design | 2-7% at 100 nL/min [52] | Optimized for high efficiency at low flow rates |
| Tissue Damage | Significant, creates chemical gradients [6] | Minimal due to reduced size [52] | Less disruption of native neurochemical environment |
FSCV as a Complementary Technique
Table 2: Key differences between microdialysis and FSCV for adenosine detection
| Characteristic | Microdialysis | FSCV |
|---|---|---|
| Temporal Resolution | Minutes (5-20 min samples) [5] | Subsecond (100 ms sampling rate) [22] [5] |
| Spatial Resolution | Limited by probe size (~220 μm) [6] | High (7 μm electrode diameter) [21] [28] |
| Adenosine LOD | ~5 nM [5] | Varies with electrode design |
| Key Advantage | Broad analyte screening [6] | Real-time transient detection [53] |
| Primary Limitation | Slow temporal resolution, tissue damage [6] | Limited to electroactive analytes [22] |
Detailed Experimental Protocols
Microfabricated Microdialysis Probe Protocol
Objective: To assess the performance of microfabricated microdialysis probes with enhanced recovery characteristics at low flow rates.
Materials & Methods:
- Probes: Si-based microfabricated probes (45 μm thick × 180 μm wide) with 4 mm nanoporous membrane section [52]
- Perfusion Fluid: Artificial cerebrospinal fluid (aCSF: 145 mM NaCl, 2.68 mM KCl, 1.10 mM MgSO₄, 1.22 mM CaCl₂, 0.50 mM NaH₂PO₄, 1.55 mM Na₂HPO₄, pH 7.4) [52]
- Flow Rate: 100 nL/min using precision syringe pump
- Analytical Method: LC-MS for dialysate analysis
- Experimental Model: In vivo rat striatum
- Stimulus: Intraperitoneal amphetamine injection (5 mg/kg)
Validation: The methodology demonstrated reliable detection of 13 neurochemicals simultaneously, with amphetamine evoking a 43-fold dopamine increase, comparable to conventional probes but with significantly less tissue disruption [52].
FSCV Adenosine Detection Protocol
Objective: To directly detect rapid adenosine transients with subsecond temporal resolution.
Materials & Methods:
- Electrodes: Carbon-fiber microelectrodes (7 μm diameter, ~100 μm exposed length) [28] or cone-shaped 30 μm variants for enhanced longevity [28]
- Waveform: Triangular potential scan from -0.4 V to 1.45 V and back at 400 V/s versus Ag/AgCl reference electrode [5]
- Sampling Rate: 10 Hz (every 100 ms)
- Data Visualization: Background-subtracted cyclic voltammograms and false color plots
- Identification: Characteristic oxidation peaks at 1.4 V (primary) and 1.0 V (secondary) [5] [53]
Validation: Specificity confirmed through pharmacological manipulations and peak ratio analysis (secondary:primary peak current ratio 0.49-0.89) [53].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key reagents and materials for adenosine research methodologies
| Item | Function/Application | Example Usage |
|---|---|---|
| Carbon-Fiber Microelectrodes (CFMEs) | Working electrode for FSCV; detects electroactive analytes | 7 μm standard or 30 μm cone-shaped for improved longevity [28] |
| Artificial Cerebrospinal Fluid (aCSF) | Physiological perfusion medium for microdialysis | Mimics extracellular fluid composition [52] |
| DPCPX (8-cyclopentyl-1,3-dipropylxanthine) | Selective A1 receptor antagonist | Confirms adenosine receptor-mediated effects [22] |
| Nafion Coatings | Cation-exchange polymer for electrode modification | Enhances selectivity against anionic interferents [5] |
| Boron-Doped Diamond Electrodes | Alternative electrode material | Wide potential window, reduced fouling for adenosine detection [54] |
| Flow Injection Analysis (FIA) System | Electrode calibration and in vitro characterization | Validates electrode performance pre-implantation [55] |
Technological Integration & Future Directions
The historical division between microdialysis and FSCV is narrowing with emerging technologies that combine their advantages. Recent demonstrations of multiplexed FSCV and genetically encoded fluorescence sensors enable simultaneous monitoring of adenosine, dopamine, and glutamate, revealing complex neuromodulatory interactions with high spatiotemporal resolution [22]. This approach leverages the subsecond temporal resolution of FSCV for electroactive analytes while incorporating optical sensors for non-electroactive neurotransmitters.
Advanced electrode designs continue to push methodological boundaries. The development of 30 μm cone-shaped CFMEs demonstrates a 3.7-fold improvement in in vivo dopamine signals compared to conventional 7 μm CFMEs, with significantly reduced glial activation and enhanced longevity [28]. Similarly, boron-doped diamond microsensors offer exceptional electrochemical properties with reduced fouling for adenosine detection in complex biological environments [54].
Figure 2: Methodology Integration Pathway. This diagram illustrates how the complementary strengths of microdialysis and FSCV are converging in next-generation integrated approaches for neurochemical monitoring.
The optimization of recovery in neurochemical sampling requires careful consideration of the fundamental trade-offs between flow rate, membrane surface area, temporal resolution, and tissue integrity. Microdialysis offers unparalleled versatility for screening multiple analytes but is constrained by relatively poor temporal resolution and significant tissue disruption. Conversely, FSCV provides exquisite temporal resolution for monitoring rapid adenosine transients but is limited to electroactive species. The emerging trend toward miniaturized probes, advanced electrode materials, and multiplexed sensing platforms represents a promising direction for achieving comprehensive neurochemical monitoring with minimal perturbation to the native biological environment. Researchers must align their methodological choices with specific experimental questions, recognizing that optimal recovery encompasses not only quantitative extraction efficiency but also preservation of the physiological system under investigation.
Fast-scan cyclic voltammetry (FSCV) has emerged as a powerful technique for monitoring neurochemical dynamics in vivo, offering unparalleled temporal and spatial resolution for detecting electroactive neurotransmitters such as dopamine, serotonin, and adenosine [56]. Unlike microdialysis, which provides averaged measurements over minutes, FSCV captures sub-second neurotransmitter fluctuations, making it ideal for studying phasic signaling events [57]. However, this technical advantage comes with a significant analytical challenge: accurately distinguishing the target analyte from interferent molecules with similar electrochemical properties in the complex brain environment [58]. The validation of analyte identity therefore becomes paramount, particularly when comparing FSCV against the higher chemical selectivity of microdialysis for tonic concentration measurements [59].
This comparison guide examines the two primary strategies for addressing this challenge: pharmacological validation, which uses receptor pharmacology to confirm chemical identity, and waveform optimization, which enhances selectivity through electrochemical engineering. For researchers studying adenosine measurement, understanding the complementary strengths and limitations of FSCV versus microdialysis is essential for experimental design and data interpretation within the broader context of neurochemical research.
Core Principles: FSCV versus Microdialysis for Adenosine Measurement
Table 1: Fundamental Methodological Differences Between FSCV and Microdialysis
| Parameter | Fast-Scan Cyclic Voltammetry (FSCV) | Microdialysis |
|---|---|---|
| Temporal Resolution | Sub-second (100 ms) [56] | Minutes per sample [59] |
| Spatial Resolution | Micrometer scale (7-10 μm electrodes) [34] | Millimeter scale (200-300 μm probes) [59] |
| Measurement Type | Phasic, transient release events [57] | Tonic, basal concentrations [59] |
| Selectivity Mechanism | Electrochemical signature + waveform design [56] | Physical separation + HPLC analysis [59] |
| Tissue Damage | Minimal with CFMEs [33] | Significant tissue disruption [59] |
| Adenosine LOD | ~50 nM with optimized waveforms [56] | ~1-5 nM with HPLC detection [59] |
| In Vivo Compatibility | Acute and chronic recordings possible [33] | Limited to chronic implantation [59] |
The selection between FSCV and microdialysis for adenosine research involves fundamental trade-offs. FSCV excels at capturing the rapid dynamics of adenosine signaling, such as its transient release during seizure termination [58] or its modulatory effects on dopamine transmission [56]. This makes FSCV ideal for studying the moment-to-moment functional role of adenosine in neural circuits. Conversely, microdialysis provides precise quantification of steady-state adenosine concentrations but cannot resolve rapid physiological changes due to its poor temporal resolution [59]. Additionally, the large probe size of microdialysis systems causes significant tissue damage and disrupts the blood-brain barrier, potentially altering the very neurochemical environment being measured [59].
Pharmacological Validation Strategies
Pharmacological validation serves as a cornerstone methodology for confirming analyte identity in FSCV experiments, particularly when transitioning from animal models to human studies where complete validation is challenging [58]. This approach uses receptor-specific drugs to manipulate neurotransmitter systems in predictable ways, creating unique pharmacological fingerprints for target analytes.
Table 2: Pharmacological Validation Protocols for Neurotransmitter Identification
| Neurotransmitter | Receptor Target | Drug Class | Experimental Effect | Validation Strength |
|---|---|---|---|---|
| Adenosine | A1 receptors | Antagonist (e.g., DPCPX) | Blocks adenosine-mediated inhibition | Confirms functional role |
| Adenosine | A2A receptors | Agonist (e.g., CGS-21680) | Enhances adenosine signaling | Corroborates release dynamics |
| Dopamine | D2 autoreceptors | Antagonist (e.g., raclopride) | Increases dopamine release | Confirms autoreceptor regulation |
| Dopamine | DAT transporter | Inhibitor (e.g., nomifensine) | Prolongs dopamine half-life | Verifies uptake mechanisms |
| Serotonin | SERT transporter | Inhibitor (e.g., fluoxetine) | Enhances serotonin signals | Distinguishes from dopamine |
The "Five Golden Rules" framework establishes comprehensive standards for validating FSCV measurements, especially in preclinical models [58]. This multi-faceted approach requires: (1) identification of neurotransmitter-specific electrochemical signatures using standard solutions; (2) confirmation of chemical identity through complementary techniques like microdialysis at the FSCV recording site; (3) anatomical validation through electrode placement verification; (4) kinetic validation of spontaneous or evoked neurotransmitter changes; and (5) pharmacological validation using receptor-specific drugs [58].
In human studies, where pharmacological validation may be limited by safety considerations, researchers increasingly rely on waveform optimization and advanced data analysis to compensate for the inability to fully apply all validation criteria [58]. This limitation highlights the importance of thorough preclinical characterization before transitioning to clinical applications.
Figure 1: Pharmacological Validation Workflow - This diagram illustrates the logical framework for pharmacological validation of FSCV signals, showing how different drug classes modulate neurotransmitter systems to produce predictable responses that confirm analyte identity.
Waveform Optimization Strategies
Waveform optimization represents the electrochemical engineering approach to enhancing analyte selectivity in FSCV. By carefully designing the voltage waveform applied to the working electrode, researchers can manipulate electron transfer kinetics and adsorption properties to favor specific neurotransmitters.
Table 3: Waveform Parameters and Their Impact on Selectivity
| Waveform Parameter | Effect on Selectivity | Impact on Adenosine Detection | Optimal Range for Adenosine |
|---|---|---|---|
| Holding Potential | Controls analyte adsorption | Negative potentials enhance sensitivity | -0.4 V to -0.6 V [56] |
| Switching Potential | Determines oxidation extent | Higher potentials improve signal | +1.4 V to +1.5 V [56] |
| Scan Rate | Affects current magnitude | Moderate rates balance S/N | 300-400 V/s [56] |
| Scan Pattern | Shapes CV fingerprint | Triangular vs. rectangular | Application-specific |
| Frequency | Sets temporal resolution | Lower frequencies reduce fouling | 10 Hz [56] |
Recent advances in waveform design have specifically addressed the challenge of adenosine detection in the presence of interferents like hydrogen peroxide and histamine, which share similar oxidation potentials [56]. The extended waveform pattern, which uses a more negative holding potential (-0.6 V) and more positive switching potential (+1.4 V), has demonstrated 5-fold improvement in sensitivity for various neurotransmitters compared to traditional waveforms [56]. This enhancement occurs through two mechanisms: the more negative holding potential increases electrostatic adsorption of cationic molecules, while the more positive switching potential generates additional oxygen-containing functional groups on the carbon electrode surface, creating more adsorption sites [56].
For adenosine-specific detection, researchers have developed specialized waveforms that exploit its unique electrochemical properties, particularly its distinct adsorption characteristics and electron transfer kinetics compared to catecholamines [56]. These optimized waveforms shift the voltammetric peaks of interferents while maintaining adenosine's characteristic signature, facilitating clearer identification in complex biological environments.
Figure 2: Waveform Optimization Logic - This diagram illustrates the relationship between waveform parameters, their electrochemical effects, and the resulting analytical outcomes for neurotransmitter detection.
Experimental Protocols and Methodologies
Carbon-Fiber Microelectrode Fabrication
The foundation of reliable FSCV measurements begins with consistent electrode fabrication. The standard protocol involves aspirating a single carbon fiber (7-10 μm diameter) into a borosilicate glass capillary, which is then pulled using a vertical electrode puller to create a sealed glass sheath [57]. The carbon fiber is trimmed to a length of 50-100 μm using a surgical blade under microscopic visualization [57]. Electrodes are subsequently conditioned before experiments by applying a triangular waveform (-0.4 V to +1.3 V, 400 V/s) at 60 Hz for 30 minutes in PBS buffer, which stabilizes the electrochemical response by generating oxygen-containing functional groups on the carbon surface [56].
Recent advancements in electrode design have addressed chronic implantation challenges. Studies demonstrate that 30 μm diameter carbon-fiber microelectrodes with cone-shaped tips, created through electrochemical etching, show 3.7-fold improvement in in vivo dopamine signals and significantly reduced glial activation compared to traditional 7 μm electrodes [33]. This geometric modification enhances mechanical robustness while minimizing insertion-induced tissue damage, particularly important for long-term adenosine monitoring studies.
FSCV Data Acquisition Protocol
Standard FSCV measurements employ a triangular waveform scanning from -0.4 V to +1.3 V and back at 400 V/s, repeated at 10 Hz frequency [56]. The electrode is held at -0.4 V between scans to promote adsorption of cationic analytes. Current data is typically sampled at 100 kHz using a National Instruments PCI-6052E card or similar data acquisition system [57]. The critical step in FSCV data processing is background subtraction, where a baseline current collected immediately before a neurochemical event is subtracted from subsequent scans to reveal the Faradaic current of interest [56].
For adenosine-specific detection, researchers modify this standard protocol by implementing a "modified adenosine waveform" with extended potential windows (-0.6 V to +1.4 V) and optimized scan rates to maximize the separation between adenosine and interferent signals [56]. This specialized approach capitalizes on adenosine's distinct adsorption properties to enhance selectivity.
Calibration and Signal Validation
Rigorous calibration is essential for quantitative FSCV measurements. Electrodes are calibrated in vitro using flow injection systems that deliver known concentrations of adenosine in artificial cerebrospinal fluid [57]. The resulting current responses are used to create calibration curves, establishing the relationship between concentration and electrochemical current for each specific electrode [56].
A significant challenge in FSCV quantification is the difference in electrode sensitivity between in vitro calibration and in vivo measurements, which can vary due to factors like tissue encapsulation and biofouling [56]. Recent approaches address this through paired FSCV-EIS (electrochemical impedance spectroscopy) measurements, which track changes in electrode impedance, reactance, and capacitance during experiments, providing real-time information about electrode performance drift [60]. This integrated approach represents a significant advancement toward in situ calibration strategies.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key Research Reagent Solutions for FSCV Validation
| Material/Reagent | Function | Application Notes |
|---|---|---|
| Carbon Fiber (7-10 μm) | Working electrode material | PAN-based fibers preferred for dopamine; pitch-based for larger currents [34] |
| Borosilicate Glass Capillaries | Electrode insulation | Provides mechanical support and electrical insulation [57] |
| Tris Buffer (pH 7.4) | Electrochemical medium | Maintains physiological pH for in vitro calibration [33] |
| Artificial CSF | Biological mimic | Closer to in vivo conditions than Tris buffer [33] |
| Adenosine Standards | Quantitative calibration | Used for electrode calibration and waveform optimization [56] |
| Receptor Agonists/Antagonists | Pharmacological validation | Confirms analyte identity through receptor interactions [58] |
| Graphene Oxide Coating | Electrode modification | Enhances sensitivity and stability when annealed [51] |
| Nafion Coatings | Anti-fouling layer | Reduces interference from anionic molecules [56] |
The selection of appropriate carbon fiber material significantly impacts FSCV performance. Polyacrylonitrile (PAN)-based fibers exhibit faster electron transfer kinetics and lower background currents, making them ideal for dopamine detection, while pitch-based fibers handle larger currents better, advantageous for detecting metabolites like DOPAC and ascorbic acid [34]. Recent innovations in electrode materials include carbon-coated microelectrodes (CCMs) created through electroplating of graphene oxide followed by mild annealing at 250°C, which demonstrate exceptional sensitivity (125.5 nA/μM for dopamine) and low detection limits (5 nM) [51].
For adenosine studies specifically, the development of fouling-resistant electrode coatings is particularly valuable, as adenosine measurements often require extended recording sessions to capture transient physiological events. These material advances, combined with optimized pharmacological and waveform strategies, provide researchers with an increasingly sophisticated toolkit for validating analyte identity in FSCV experiments.
In the field of neuroscience and drug development, establishing reliable neurochemical baselines is a fundamental prerequisite for meaningful experimental outcomes. This challenge is particularly acute when studying neuromodulators like adenosine, which plays critical roles in sleep regulation, seizure termination, and neuroprotection [61] [48]. The very techniques used to measure these analytes—whether microdialysis or Fast Scan Cyclic Voltammetry (FSCV)—inevitably disrupt the delicate physiological environment they aim to measure. Anesthesia protocols and surgical recovery times introduce additional variables that can significantly alter baseline neurochemistry, potentially compromising data interpretation and therapeutic development. This guide provides a comparative analysis of microdialysis and FSCV for adenosine measurement, focusing specifically on their interactions with anesthesia and post-surgical recovery processes, to help researchers design more reliable experimental paradigms.
Technical Comparison: Microdialysis vs. FSCV for Adenosine Research
The choice between microdialysis and FSCV involves significant trade-offs that directly impact baseline stability and measurement reliability, particularly in the context of anesthesia and recovery.
Table 1: Fundamental Technical Characteristics of Microdialysis and FSCV
| Characteristic | Microdialysis | Fast Scan Cyclic Voltammetry (FSCV) |
|---|---|---|
| Temporal Resolution | Minutes (5-30 min) [6] [62] | Subseconds (100 ms) [22] [16] |
| Spatial Resolution | ~300 μm probe diameter [6] | 7-30 μm electrode diameter [33] [6] |
| Primary Analyte Scope | Broad (dozens of neurotransmitters & metabolites) [6] | Limited to electroactive molecules [6] [22] |
| Tissue Damage | Significant, visible tracks [6] | Minimal with standard CFMEs; track often invisible [6] |
| Key Advantage | Broad analyte coverage from single sample | Real-time monitoring of rapid neurochemical transients |
| Key Limitation | Low temporal resolution; large probe size | Limited to electroactive species; electrode fouling |
The Tissue Damage Paradox and Baseline Establishment
The physical intrusion of measurement probes inevitably causes tissue damage, triggering neurochemical cascades that directly impact baseline establishment. Microdialysis probes (approximately 300 μm diameter) create substantial tissue disruption visible via light microscopy, inducing a foreign body response and creating gradients of disrupted neurotransmitter activity [6]. Studies reveal that implanting a microdialysis probe creates a gradient of disrupted dopamine activity, with up to 90% loss in evoked amplitude when measured 200 μm from the probe [6].
Conversely, carbon fiber microelectrodes (CFMEs) used in FSCV (typically 7 μm diameter) cause minimal damage, with tracks often invisible at light microscopy level and only observable with difficulty using electron microscopy [6]. However, recent research indicates that increasing CFME diameter to 30 μm improves mechanical durability but significantly increases tissue damage and glial activation, which can be mitigated through cone-shaped geometric modifications [33] [28].
Mechanically Evoked Adenosine Release: A Shared Confounding Factor
Both techniques face the challenge of mechanically evoked adenosine release during implantation. Research has demonstrated that mechanical stimulation (e.g., lowering an electrode 50 μm) evokes rapid, transient increases in extracellular adenosine concentrations (~0.8 μM in slices, ~3.3 μM in vivo) lasting approximately 18 seconds [48]. This release is partially activity-dependent (reduced by calcium chelation and tetrodotoxin) and partially dependent on extracellular ATP metabolism (reduced by NTDPase inhibition) [48]. This mechanically evoked adenosine likely serves a neuroprotective function but represents a significant confounding factor for baseline establishment that must be accounted for in experimental design.
Anesthesia and Temporal Considerations in Baseline Establishment
The timing of measurements relative to probe implantation and the use of anesthesia significantly influence measured baselines through multiple pathways.
The Implantation Timeline and Foreign Body Response
Microdialysis measurements demonstrate significant instability in the hours following probe implantation, with dopamine concentrations and their sensitivity to tetrodotoxin varying substantially over the first 24 hours [6]. This has led to standardized wait times (typically 12-24 hours) before collecting experimental data in microdialysis studies [6]. The foreign body response evolves over hours, days, and weeks post-implantation, making the selection of measurement timing critical for interpretation [6].
Table 2: Establishing Measurement Baselines: Temporal Considerations and Technical Responses
| Factor | Impact on Baseline | Evidence | Methodological Response |
|---|---|---|---|
| Post-Implantation Time | Neurochemical instability for 24+ hours; evolving foreign body response [6] | Microdialysis shows varying DA sensitivity to TTx over 24h [6] | Standardized wait times (12-24h) before data collection [6] |
| Mechanical Stimulation | Rapid adenosine release (3.3 μM in vivo) lasting ~18s [48] | Mechanical perturbation evokes transient adenosine via activity-dependent and ATP metabolism pathways [48] | Allow stabilization period post-implantation; account for release in data interpretation |
| Anesthesia Effects | Alters neurovascular coupling, neural activity, and neurotransmitter dynamics [58] | Limited direct adenosine studies; known confound in FSCV measurements [58] | Use awake recordings where possible; consistent anesthesia protocols; consider alternatives like decerebration |
| Electrode Properties | Larger electrodes (30μm) cause more tissue damage but offer better sensitivity [33] | 30μm CFMEs show reduced in vivo performance due to tissue damage; cone-shape modification improves biocompatibility [33] [28] | Use smaller electrodes (7μm) when possible; consider cone-shaped geometries for chronic studies |
Anesthesia Considerations
Anesthesia represents a significant confounding variable in neurochemical measurement, potentially altering neurovascular coupling, neural activity, and neurotransmitter dynamics [58]. While direct studies of anesthesia effects on adenosine baselines are limited, the known effects on other neurotransmitters suggest careful consideration of anesthesia protocols is essential. The trend toward awake, behaving animal models in FSCV research reflects the recognition of this confound [33] [58]. When anesthesia is necessary, consistent protocols and appropriate stabilization periods are essential for reliable baseline establishment.
Experimental Approaches and Methodological Details
FSCV Methodology for Adenosine Detection
Standard FSCV protocols for adenosine detection utilize carbon-fiber microelectrodes (7-30 μm diameter) fabricated by sealing a single carbon fiber in borosilicate glass capillary with epoxy, then trimming to expose 50-400 μm of active length [33] [48]. The typical waveform parameters include a triangular scan from -0.4 V to 1.3 V and back at 400 V/s, applied at 10 Hz frequency [33] [48]. Electrodes are typically pre-conditioned before experiments using extended electrochemical protocols to stabilize background currents [16].
For mechanical stimulation experiments, electrodes are positioned in tissue then lowered 50 μm using a micromanipulator while continuously recording [48]. Adenosine identification employs its characteristic electrochemical signature (oxidation peak at ~1.5 V) and pharmacological validation using receptor antagonists and enzyme inhibitors [48].
Microdialysis Coupled with Electrophoresis for Metabolite Monitoring
Advanced microdialysis approaches coupled with microchip electrophoresis and electrochemical detection (MD-ME-EC) enable simultaneous monitoring of adenosine and its metabolites (inosine, hypoxanthine, guanosine) [62]. This system uses PDMS-based microchips with 5-cm separation channels and carbon fiber working electrodes [62]. The separation buffer typically contains 35 mM sodium borate (pH 10), 15% DMSO, and 2 mM SDS, achieving separation of four purines within 85 seconds with detection limits of 10-33 μM [62]. This approach provides broader metabolite coverage but with lower temporal resolution than FSCV.
Adenosine Signaling Pathways and Experimental Workflows
The diagram below illustrates the complex pathways of adenosine production, release, and measurement following mechanical stimulation and during physiological processes, highlighting points of technical intervention and potential confounds.
Adenosine Dynamics and Measurement Pathways. This diagram illustrates the complex pathways from mechanical stimulation to adenosine detection, highlighting key technical considerations including tissue response, production mechanisms, measurement approaches, and experimental modulation points that impact baseline establishment.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Research Reagent Solutions for Adenosine Measurement Studies
| Reagent/Material | Function/Application | Example Use |
|---|---|---|
| Carbon Fiber Microelectrodes (CFMEs) | Working electrode for FSCV; 7-30 μm diameter [33] [48] | Detection of electroactive neurotransmitters (dopamine, adenosine) via oxidation/reduction currents |
| Microdialysis Probes | Continuous sampling of extracellular fluid; ~300 μm diameter [6] | Broad-spectrum collection of neurotransmitters and metabolites from brain extracellular space |
| Tetrodotoxin (TTX) | Sodium channel blocker [48] | Differentiation between action potential-dependent and independent release mechanisms |
| POM-1 | Nucleoside triphosphate diphosphohydrolase (NTDPase) inhibitor [48] | Inhibition of extracellular ATP breakdown to adenosine; tests ATP dependence of adenosine signals |
| DPCPX (8-cyclopentyl-1,3-dipropylxanthine) | Selective A1 receptor antagonist [22] | Blocks adenosine actions at A1 receptors; validates receptor-mediated effects |
| NBTI (S-(4-Nitrobenzyl)-6-thioinosine) | Equilibrative nucleoside transporter (ENT) inhibitor [48] | Inhibition of adenosine transport via ENT1/2; tests transporter contribution to extracellular adenosine |
| 5-Iodotubercidin | Adenosine kinase inhibitor [61] | Increases intracellular adenosine by blocking phosphorylation; used with intracellular sensors |
| Adenosine Deaminase | Enzyme that converts adenosine to inosine [62] | Validation of adenosine identity through enzymatic elimination |
Establishing reliable neurochemical baselines requires careful consideration of the fundamental trade-offs between microdialysis and FSCV, particularly regarding their interaction with anesthesia and post-surgical recovery processes. Microdialysis offers broad analyte coverage but suffers from significant tissue damage and poor temporal resolution, requiring extended recovery periods (12-24 hours) before stable measurements can be obtained. FSCV provides exceptional temporal resolution and minimal tissue damage but is limited to electroactive species and susceptible to mechanically evoked adenosine release immediately following implantation.
For researchers studying adenosine dynamics, the emerging approach of multiplexing techniques (combining FSCV with genetically encoded sensors) shows promise for overcoming the limitations of individual methods [22]. Additionally, geometric modifications to electrodes (e.g., cone-shaped tips) can significantly improve biocompatibility and reduce tissue damage, thereby enhancing the reliability of baseline measurements [33] [28]. Regardless of the technique selected, researchers must explicitly account for the effects of anesthesia, standardized post-implantation recovery times, and validate neurochemical signals through multiple pharmacological approaches to ensure data integrity and reproducible findings in adenosine research.
Head-to-Head Validation and Data-Driven Technique Selection
In the field of neuroscience and pharmacology, accurately measuring the dynamics of signaling molecules like adenosine is crucial for understanding brain function and developing new therapies. The choice of analytical technique fundamentally shapes the temporal scale of observable biological phenomena. Microdialysis and Fast-Scan Cyclic Voltammetry (FSCV) represent two prominent yet fundamentally different approaches for monitoring neurochemicals in vivo. Microdialysis has traditionally provided a "big picture" view of extracellular concentrations over minutes to hours, while FSCV captures rapid, subsecond chemical signaling events previously beyond analytical reach. This guide provides a direct technical comparison of these methodologies, focusing on their application in adenosine measurement research, to inform appropriate technique selection based on experimental objectives.
Microdialysis and FSCV operate on different physical principles, leading to their distinct performance characteristics, particularly regarding temporal resolution.
Microdialysis involves implanting a semi-permeable membrane probe into brain tissue. A perfusate is pumped through the probe, allowing molecules from the extracellular space to diffuse into the dialysate for subsequent collection and analysis, typically via HPLC [5] [63]. Its temporal resolution is ultimately limited by the time required to collect a sufficient sample volume for analysis.
Fast-Scan Cyclic Voltammetry (FSCV) employs a carbon-fiber microelectrode implanted into the brain tissue. A triangular voltage waveform is rapidly applied, causing electroactive molecules like adenosine to be oxidized and reduced. The resulting currents are measured, producing a characteristic signal for the analyte [5] [21] [18]. The entire scan takes less than 10 ms, enabling a very high sampling rate.
Table 1: Direct Comparison of Key Performance Specifications for Adenosine Measurement
| Performance Characteristic | Microdialysis | Fast-Scan Cyclic Voltammetry (FSCV) |
|---|---|---|
| Temporal Resolution | Minutes (1–20 minutes) [5] [63] | Subsecond (100 ms) [5] [21] |
| Spatial Resolution | Low (probe diameter ~300 µm) [6] | High (electrode diameter ~7 µm) [5] [6] |
| Limit of Detection for Adenosine | ~5 nM [5] | Not explicitly stated, but sensitive to nM changes [18] |
| In Vivo Applicability | Yes (acute and chronic) | Yes (typically acute) |
| Measurement Type | Direct, absolute concentration (total pool) [5] | Differential, relative change in concentration [21] |
| Key Advantage | Broad chemical scope; measures multiple analytes simultaneously [5] [6] | Excellent temporal resolution for rapid chemical events [5] |
| Primary Limitation | Poor temporal resolution; tissue damage from probe implantation [63] [6] | Limited analyte scope (electroactive species only); cannot measure basal levels [21] |
Experimental Protocols for Adenosine Detection
The distinct operational principles of microdialysis and FSCV necessitate completely different experimental setups and procedures.
Microdialysis Protocol for Adenosine
Microdialysis protocols for adenosine focus on capturing stable, basal levels and slower changes in concentration.
- Probe Implantation: A concentric microdialysis probe is stereotactically implanted into the brain region of interest. Probes typically have a membrane diameter of approximately 300 µm and a length of several millimeters [64] [6].
- Perfusion and Sample Collection: Artificial Cerebrospinal Fluid is perfused through the probe at low flow rates. Following an implantation wait time to stabilize the system, dialysate is collected in vials over defined intervals, typically ranging from 1 to 20 minutes [64] [63].
- Sample Analysis: The collected dialysate is analyzed, often using High-Performance Liquid Chromatography, which separates adenosine from other compounds like hypoxanthine and uric acid for quantification [5].
- Key Experimental Considerations: A consistent wait time after probe implantation is critical, as the initial tissue trauma can cause unstable neurotransmitter measurements [6]. Furthermore, to achieve a 1-minute temporal resolution, specialized equipment is required, including capillary columns and optimized mobile phases to eliminate interferences [64].
FSCV Protocol for Adenosine
FSCV protocols are designed to capture transient, stimulus-evoked or spontaneous adenosine release on a timescale of seconds.
- Electrode Preparation and Implantation: A carbon-fiber microelectrode is prepared and implanted into the target brain region. These electrodes are significantly smaller than microdialysis probes, with a typical diameter of 7 µm [5] [6].
- Waveform Application and Data Acquisition: A specific triangular waveform is applied, scanning from -0.4 V to 1.45 V or 1.50 V and back versus a Ag/AgCl reference electrode at a rate of 400 V/s. This scan is repeated 10 times per second [5] [3].
- Background Subtraction and Signal Processing: The large, stable background charging current is subtracted from the total current. The remaining Faradaic current is used to generate a background-subtracted cyclic voltammogram, which serves as an electrochemical fingerprint for adenosine [5] [21].
- Key Experimental Considerations: Adenosine is identified by its characteristic oxidation peaks at ~1.4 V and ~1.0 V. To confirm the identity of the measured analyte, pharmacological manipulations are often used [5]. FSCV is a differential technique, making it excellent for measuring changes in concentration but unable to quantify absolute basal levels [21].
Figure 1: The typical FSCV experimental workflow for adenosine detection, highlighting the key steps from electrode preparation to data output.
Biological Applications & Data Comparison
The different temporal resolutions of microdialysis and FSCV make them suitable for investigating distinct types of biological questions related to adenosine signaling.
Capturing Slow, Tonic Adenosine Signaling with Microdialysis
Microdialysis is well-suited for studying long-term shifts in basal adenosine levels. For instance, it has been used to measure adenosine buildup over 15 minutes during ischemic events like stroke [5]. Furthermore, a human microdialysis study documented a 6 to 31-fold increase in adenosine concentrations, but this rise was detected in samples collected three minutes after the seizure had already terminated [3]. This demonstrates the technique's utility for confirming adenosine involvement in prolonged processes but also its limitation in resolving rapid, phasic events.
Revealing Rapid, Phasic Adenosine Signaling with FSCV
FSCV has been instrumental in discovering a novel, rapid mode of adenosine signaling that occurs over just a few seconds [5] [8]. In a large animal model, FSCV recorded a 260% increase in adenosine relative to baseline just seconds before electrographic seizure termination [3]. This precise temporal correlation, which is impossible to establish with microdialysis, strongly suggests that adenosine itself plays a direct role in stopping seizures. This rapid release can be evoked by various stimuli or occur spontaneously, indicating a form of activity-dependent neuromodulation that operates on a much faster timescale than previously thought [5].
Table 2: Comparison of Experimental Findings on Adenosine Release
| Experimental Context | Measurement Technique | Temporal Profile of Adenosine Change | Biological Insight |
|---|---|---|---|
| Ischemia/Stroke | Microdialysis [5] | Buildup over 15+ minutes | Indicates role in neuroprotection and energy metabolism on a slow time scale. |
| Human Seizure (Post-Termination) | Microdialysis [3] | Increase measured 3 minutes after seizure end | Confirms adenosine is involved in the post-ictal period but cannot define its role in termination. |
| Seizure Termination (Animal Model) | FSCV [3] | 260% increase 2–7 seconds before seizure end | Suggests a direct, rapid mechanism for adenosine in seizure termination. |
| Neuronal Stimulation | FSCV [5] | Transient release lasting a few seconds | Reveals a novel, rapid, activity-dependent mode of adenosine signaling. |
Figure 2: A timeline comparison of the adenosine dynamics captured by FSCV (green) versus microdialysis (blue). FSCV resolves rapid, phasic release correlated with the seizure event itself, while microdialysis captures a slower, tonic change that becomes apparent after the event.
The Scientist's Toolkit: Essential Research Reagents & Materials
Successful implementation of microdialysis or FSCV requires specific materials and reagents. The following table details key components for each technique.
Table 3: Essential Research Reagents and Materials
| Item Name | Function/Application | Technique |
|---|---|---|
| Carbon-Fiber Microelectrode (CFM) | The working electrode for applying voltage and detecting Faradaic currents from electroactive analytes like adenosine. Typically 7 µm in diameter [5] [6]. | FSCV |
| Triangular Waveform | The specific voltage pattern applied to the CFM to oxidize and reduce adenosine. A common parameters are a scan from -0.4 V to 1.5 V at 400 V/s [5] [3]. | FSCV |
| Ag/AgCl Reference Electrode | Provides a stable, known reference potential against which the voltage at the CFM is controlled in an electrochemical cell [5] [21]. | FSCV |
| Concentric Microdialysis Probe | The device implanted into tissue; its semi-permeable membrane allows diffusion of analytes from the extracellular space into the perfusate. A common membrane diameter is 300 µm [64] [6]. | Microdialysis |
| Artificial Cerebrospinal Fluid (aCSF) | The solution perfused through the microdialysis probe. It is a balanced salt solution that mimics the ionic composition of native CSF [64]. | Microdialysis |
| High-Performance Liquid Chromatography | An analytical method used to separate and quantify the various chemical constituents collected in the microdialysis dialysate [5] [64]. | Microdialysis |
The comparative analysis between microdialysis and FSCV reveals a clear trade-off: breadth of chemical information versus superior temporal resolution. Microdialysis offers a versatile platform for monitoring a wide range of molecules, including non-electroactive substances, providing valuable data on basal concentrations and long-term shifts. FSCV, in contrast, provides a unique window into the rapid, subsecond world of chemical neurotransmission, which is essential for understanding the fine-grained dynamics of neuromodulators like adenosine.
Technique selection is not a matter of which is superior, but of which is appropriate for the biological question at hand. For studies of metabolic tone, slow neuromodulation, or the simultaneous monitoring of a broad chemical panel, microdialysis remains the tool of choice. For investigations into the rapid, phasic release of electroactive neurotransmitters and its correlation with behavioral or electrical events on a subsecond timescale, FSCV is indispensable. Future research may increasingly leverage these techniques in a complementary fashion to build a more complete model of adenosine signaling across multiple temporal domains.
In neuroscience and pharmacology research, the choice of analytical technique profoundly influences the interpretation of neurochemical signaling. The spatial resolution of a method determines whether a measurement reflects a localized signaling event or an averaged concentration from a heterogeneous tissue region. This guide provides a direct comparison between two established approaches for studying neurotransmitters like adenosine: microdialysis, representing bulk tissue analysis, and fast-scan cyclic voltammetry (FSCV), which resolves discrete chemical hotspots. Understanding their inherent differences in spatial resolution, temporal capability, and analytical scope is essential for designing rigorous experiments and accurately interpreting data on neuromodulation, particularly for the study of rapid adenosine signaling.
Bulk Tissue Analysis via Microdialysis
Microdialysis is a widely used technique for sampling chemicals from the extracellular space of living tissue.
- Principle: A probe with a semi-permeable membrane is implanted into the brain. A perfusate is pumped through the probe, and molecules from the extracellular fluid diffuse across the membrane into the dialysate, which is collected for offline analysis.
- Spatial Resolution: The spatial resolution of microdialysis is intrinsically low. Probe diameters are typically 200-300 μm, and they sample from a much larger area of tissue surrounding the probe [6]. The technique measures an averaged concentration from this heterogeneous tissue volume, effectively blurring discrete neurotransmitter release events [6].
- Tissue Damage Considerations: The implantation of a microdialysis probe causes significant tissue disruption, creating a gradient of altered neurochemical activity. Research shows that electrically evoked dopamine release is significantly suppressed within a 200 μm radius of the probe track and is completely absent immediately adjacent to the probe [6]. This penetration injury and the ensuing foreign body response can compromise the physiological relevance of the measurements.
Discrete Hotspot Analysis via Fast-Scan Cyclic Voltammetry (FSCV)
FSCV is an electrochemical technique designed to detect rapid changes in the concentration of electroactive neurotransmitters.
- Principle: A carbon-fiber microelectrode (CFME), typically 7 μm in diameter, is implanted in the brain. A rapid, repeating triangular voltage waveform is applied to the electrode, oxidizing and reducing electroactive molecules like adenosine and dopamine at characteristic potentials. The resulting current provides a "fingerprint" for the analyte and its concentration [65] [58].
- Spatial Resolution: FSCV offers high spatial resolution, capable of monitoring neurotransmitter dynamics at a single-carbon fiber level with a diameter of 7 μm [65] [28]. This allows it to detect neurotransmitter release from discrete hotspots. A 2025 study demonstrated that adenosine's inhibitory effect on dopamine and glutamate release was only observed within a 250 μm distance, showcasing FSCV's ability to resolve such regional effects [22].
- Tissue Damage Considerations: CFMEs cause minimal tissue damage; their tracks are often invisible under light microscopy [6]. To improve durability, 30 μm diameter CFMEs have been developed, but their larger size can cause more tissue damage and reduced signal in vivo. This can be mitigated by crafting the fiber into a cone-shaped tip, which reduces insertion trauma and glial activation, preserving the local microenvironment for more accurate measurement [28].
Table 1: Direct Comparison of Core Spatial and Technical Specifications
| Feature | Microdialysis (Bulk Analysis) | FSCV (Discrete Hotspots) |
|---|---|---|
| Spatial Resolution | Low (200-300 μm probe diameter, samples from larger tissue volume) [6] | High (7-30 μm electrode diameter, measures local release) [65] [28] |
| Temporal Resolution | Low (minutes to hours for sample collection) [6] | Very High (sub-second, typically 100 ms) [22] [65] |
| Measured Information | Average extracellular concentration from a bulk tissue region [6] | Real-time fluctuations at discrete release sites [22] |
| Primary Analytes | Broad (neurotransmitters, amino acids, neuropeptides, metabolites) [6] | Narrow (Electroactive molecules, e.g., adenosine, dopamine) [6] [8] |
| Tissue Impact | Significant penetration injury, foreign body response [6] | Minimal damage; tracks often invisible via light microscopy [6] |
| Key Spatial Finding | Creates a gradient of disrupted dopamine activity up to ~1 mm from probe [6] | Can resolve adenosine neuromodulation within a precise 250 μm radius [22] |
Experimental Protocols for Spatial Characterization
Protocol for Mapping Diffusion and Modulation with FSCV
The following methodology, adapted from a 2025 multiplexing study, outlines how FSCV is used to characterize the spatial extent of adenosine neuromodulation [22].
Sensor Expression & Electrode Implantation:
- A genetically encoded glutamate sensor (iGluSnFR3.v857) is expressed in the caudate-putamen region of a brain slice using a Sindbis viral vector for rapid expression.
- A carbon-fiber microelectrode (CFME) is implanted to monitor electrically stimulated dopamine and adenosine release.
Stimulation and Pharmacological Manipulation:
- Electrical stimulation is applied to evoke neurotransmitter release.
- Exogenous adenosine is applied locally via a micropipette for 30 seconds to simulate a transient neuromodulatory event.
- The A1 receptor antagonist DPCPX is perfused to block adenosine receptors and confirm the mechanism of action.
Multiplexed Data Acquisition:
- FSCV: The CFME records dopamine and adenosine dynamics with high temporal resolution using a triangular waveform (e.g., -0.4 V to +1.3 V and back).
- Fluorescence Imaging: Simultaneously, a microscope captures changes in glutamate fluorescence from cells expressing iGluSnFR3.
Spatial Analysis:
- Measurements are taken at varying distances from the site of adenosine application.
- The spatial range of adenosine's inhibitory effect is quantified by determining the distance at which the inhibition on evoked dopamine and glutamate release is no longer observed, which was found to be 250 μm [22].
Protocol for Assessing Tissue Damage and Signal Gradients
This protocol, derived from a review of microdialysis and FSCV effects, details how the spatial impact of probe implantation is quantified [6].
Dual-Probe Implantation:
- A microdialysis probe is implanted in the rat striatum.
- An FSCV carbon-fiber microelectrode is implanted at a series of defined separation distances from the microdialysis probe (e.g., 0 μm, 200 μm, 500 μm, 1000 μm).
Electrical Stimulation and Measurement:
- Dopamine release is evoked by electrical stimulation of dopaminergic pathways.
- The amplitude of the evoked dopamine signal is recorded by the FSCV electrode at each distance from the microdialysis probe.
Pharmacological Validation:
- The DA uptake inhibitor nomifensine is administered to determine if suppressed release near the probe can be pharmacologically reversed, indicating the presence of intact but dysfunctional terminals.
Data Analysis:
- A gradient of disrupted DA activity is mapped. One study found a ~90% loss in evoked DA response amplitude at a 200 μm separation, and a complete loss when the electrode was glued to the probe (0 μm) [6].
- Post-experiment immunohistochemistry is performed to correlate chemical measurements with histological evidence of tissue damage.
Diagram 1: Experimental workflow for mapping the spatial gradient of neurotransmitter disruption around a microdialysis probe.
The Scientist's Toolkit: Key Research Reagents and Materials
Table 2: Essential Materials for FSCV and Microdialysis Experiments
| Item | Function/Description | Example Use Case |
|---|---|---|
| Carbon-Fiber Microelectrode (CFME) | Working electrode for FSCV; typically 7 µm diameter for high spatial resolution [65]. | Detecting rapid, localized adenosine or dopamine transients [22] [8]. |
| Fused-Silica Chronic CFME | Miniaturized electrode sealed in fused-silica for long-term implantation with minimal tissue response [65]. | Longitudinal neurotransmitter measurements in behaving animals [65]. |
| iGluSnFR3.v857 | Genetically encoded fluorescence sensor for glutamate with high spatial resolution [22]. | Multiplexed detection of non-electroactive neurotransmitters alongside FSCV [22]. |
| DPCPX (8‐cyclopentyl‐1,3‐dipropylxanthine) | Selective A1 receptor antagonist [22]. | Pharmacological validation of adenosine's actions via A1 receptor blockade [22]. |
| Microdialysis Probe | Probe with semi-permeable membrane (200-300 µm) for sampling extracellular fluid [6]. | Bulk collection of a wide range of analytes from a tissue region [6]. |
| Nomifensine | Dopamine uptake inhibitor [6]. | Used to probe the integrity of dopamine terminals near an implanted probe [6]. |
Signaling Pathways and Spatial Logic of Neuromodulation
The spatial resolution of a measurement technique directly impacts the ability to resolve the operational logic of neuromodulatory pathways. The following diagram illustrates a specific adenosine-mediated pathway whose spatial characteristics were defined using high-resolution techniques.
Diagram 2: The spatial logic of rapid adenosine neuromodulation, as resolved by high-resolution techniques.
The comparative data reveals a fundamental trade-off. Microdialysis provides a comprehensive, broad-spectrum chemical profile but at the cost of spatial and temporal detail, and with significant tissue disruption that can alter the very system being measured [6]. Its low resolution makes it unsuitable for studying rapid, focal neuromodulation. In contrast, FSCV excels at capturing the fast, local dynamics of electroactive neurotransmitters like adenosine with minimal tissue impact, enabling the discovery of rapid signaling modes and precise mapping of neuromodulatory fields [22] [8].
The choice between these techniques is not a matter of which is superior, but which is appropriate for the specific research question. For defining the chemical milieu of a large brain region over extended periods, microdialysis remains valuable. However, for investigating the rapid, spatially precise interplay of neuromodulators like adenosine, dopamine, and glutamate at discrete hotspots, FSCV and its multiplexing with optical sensors are indispensable. The emerging trend of multiplexing FSCV with genetically encoded sensors represents the cutting edge, combining the high temporal resolution of electrochemistry with the molecular specificity and spatial resolution of fluorescence imaging to simultaneously monitor multiple neurotransmitters in real-time [22]. This integrated approach is paving the way for a more complete and nuanced understanding of brain chemistry.
The accurate measurement of specific analytes within complex biological systems represents a fundamental challenge in neuroscience and drug development. For neuromodulators like adenosine, which regulates critical processes from sleep to neuroprotection, the choice of detection methodology creates a significant trade-off between the ability to screen multiple analytes simultaneously and the capacity to monitor specific targets with high temporal and spatial precision [5]. This comparative analysis examines two principal approaches—microdialysis and fast-scan cyclic voltammetry (FSCV)—for adenosine measurement, focusing specifically on their analyte specificity, technical capabilities, and appropriate application contexts. While microdialysis offers a broad panel of detectable neurochemicals, FSCV provides direct, specific detection with superior temporal resolution, leaving researchers to navigate a landscape of complementary rather than competing technologies.
Technical Comparison: Microdialysis vs. FSCV for Adenosine Detection
The fundamental difference between these methodologies lies in their operational principles: microdialysis is a sampling technique that enables broad biochemical profiling, whereas FSCV is an electrochemical approach designed for direct, real-time measurement of specific electroactive molecules.
Table 1: Core Methodological Characteristics
| Feature | Microdialysis | Fast-Scan Cyclic Voltammetry (FSCV) |
|---|---|---|
| Basic Principle | Diffusion-based sampling across a semi-permeable membrane [6] | Direct electrochemical oxidation/reduction at an electrode surface [5] |
| Primary Advantage | Broad panel detection of numerous neurochemicals (electroactive and non-electroactive) [6] | High temporal and spatial resolution for specific electroactive analytes [5] |
| Adenosine Specificity | Requires separation via HPLC; potential interference from similar purines [5] | Inherent based on unique electrochemical "fingerprint" (voltammogram) [5] |
| Temporal Resolution | Minutes (5-10 minute typical collection intervals) [5] | Sub-second (100 ms sampling rate) [5] |
| Spatial Resolution | Low (probe diameter ~300 μm) [6] | High (carbon fiber electrode diameter 7-30 μm) [5] [33] |
| Tissue Impact | Significant; causes measurable tissue damage and disrupted neurotransmitter gradients [6] | Minimal; electrode tracks often invisible via light microscopy [6] |
Table 2: Performance Metrics for Adenosine Detection
| Performance Metric | Microdialysis | FSCV | Adenosine Biosensors |
|---|---|---|---|
| Limit of Detection | As low as 5 nM [5] | Demonstrated at sub-micromolar levels [5] | Not specified in results |
| Measurement Time Scale | Minutes to hours [5] | Seconds to sub-seconds [5] | ~2 seconds [5] |
| Key Interferents | ATP, AMP, other purines with similar retention times [5] | ATP (similar electroactive moiety), but distinguishable [5] | Dependent on enzyme specificity |
| In Vivo Application | Suitable for monitoring slow, basal concentration changes [5] | Ideal for detecting rapid, transient (seconds) adenosine signaling [5] | Used for monitoring within minutes during ischemic events [5] |
Experimental Protocols for Adenosine Detection
Microdialysis Protocol for Adenosine Measurement
Microdialysis enables the sampling of various neurochemicals from the extracellular space. The following protocol is adapted from established practices in the field [6]:
- Probe Implantation: A microdialysis probe (typically ~300 μm in diameter) is surgically implanted into the brain region of interest (e.g., striatum). The probe incorporates a semi-permeable membrane.
- Perfusion: Artificial cerebrospinal fluid (aCSF) is continuously perfused through the probe at a low, constant flow rate (e.g., 0.5-2 μL/min).
- Equilibration: Following implantation, a waiting period (often 1-24 hours) is observed to allow the tissue to stabilize, mitigating the acute effects of penetration injury on analyte levels [6].
- Dialysate Collection: The perfusate (dialysate) is collected from the outlet tube into vials over defined intervals (typically 5-20 minutes).
- Analyte Separation and Quantification: The dialysate sample is injected into a High-Performance Liquid Chromatography (HPLC) system. Adenosine is separated from other purines (e.g., ATP, ADP, AMP, inosine, hypoxanthine) and interfering compounds based on their interaction with the chromatographic column.
- Detection: The separated adenosine is typically quantified using an ultraviolet (UV) or mass spectrometry (MS) detector. The concentration is calculated by comparing the peak area/height to a standard curve.
Key Consideration: The large probe size causes significant tissue damage, disrupting local neurochemical gradients. Studies show that dopamine activity is profoundly suppressed within a 200 μm radius of the probe track, indicating that the measured adenosine levels may not reflect true physiological concentrations [6].
FSCV Protocol for Direct Adenosine Detection
FSCV directly detects electroactive molecules in real-time. The protocol for adenosine is as follows [5]:
- Electrode Preparation: A carbon-fiber microelectrode (CFME, typically 7 μm diameter) is fabricated and preconditioned. Some studies use modified electrodes (e.g., coated with Nafion and carbon nanotubes) to enhance sensitivity and selectivity over ATP [5].
- Waveform Application: The electrode is held at a negative holding potential (-0.4 V vs. Ag/AgCl). A triangular waveform is then applied, ramping to a positive switching potential (1.45-1.50 V) and back, at a high scan rate (400 V/s). This scan is repeated every 100 ms (10 Hz) [5].
- Background Subtraction: The applied waveform generates a large, stable background charging current. This background is digitally subtracted, revealing the Faradaic current from analytes undergoing oxidation/reduction.
- Signal Identification and Quantification: Adenosine undergoes a series of oxidations, producing a characteristic cyclic voltammogram (CV) with a primary oxidation peak at ~1.4 V and a secondary peak at ~1.0 V [5]. This "electrochemical fingerprint" allows it to be distinguished from other electroactive species like dopamine (oxidation ~0.6 V) or hydrogen peroxide (oxidation ~1.2 V, no secondary peak). Concentration is determined by correlating the current at the characteristic potential to a calibration curve.
Key Advantage: FSCV's sub-second temporal resolution has revealed a previously unappreciated mode of rapid adenosine signaling that lasts only a few seconds, underscoring its unique utility for studying fast physiological processes [5].
Diagram 1: FSCV detection workflow for adenosine.
Signaling Pathways and Physiological Context
Adenosine is a ubiquitous neuromodulator and a downstream product of ATP metabolism. It regulates physiological processes such as sleep, breathing, blood flow, and neurotransmission on a time scale of minutes to hours [5]. Its effects are primarily mediated through G-protein coupled receptors, with A1 receptors largely responsible for inhibitory effects and A2A receptors facilitating excitatory effects [5].
The discovery of rapid, transient adenosine release (on the scale of seconds) via FSCV has expanded the understanding of its signaling modalities [5]. This rapid release can be evoked electrically or mechanically, or occur spontaneously, and is activity-dependent. Functionally, this rapid adenosine signaling has been shown to modulate oxygen levels and evoked dopamine release, suggesting a fast modulatory role that complements its slower, tonic actions [5].
Diagram 2: Adenosine signaling pathways and timescales.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful experimentation in this field relies on specialized materials and reagents. The following table details key components for implementing FSCV and microdialysis.
Table 3: Key Research Reagent Solutions
| Item | Function/Description | Key Consideration |
|---|---|---|
| Carbon-Fiber Microelectrode (CFME) [5] [33] | Working electrode for FSCV; typically 7 μm diameter, can be modified for durability (e.g., 30 μm cone-shaped). | Small size minimizes tissue damage. Surface chemistry crucial for sensitivity and selectivity. |
| Ag/AgCl Reference Electrode [5] | Provides a stable, known reference potential for electrochemical measurements in FSCV. | Essential for maintaining potential accuracy in a biological environment. |
| Triangular Waveform [5] | The applied voltage profile (-0.4 V to +1.45 V vs. Ag/AgCl) used to oxidize/reduce adenosine in FSCV. | Specific parameters (scan rate, holding potential) are optimized for adenosine detection. |
| Artificial Cerebrospinal Fluid (aCSF) [6] | Ionic solution mimicking the brain's extracellular fluid; used as perfusate in microdialysis and in slice preparations. | Buffered to physiological pH (7.4) to maintain tissue viability and normal function. |
| Microdialysis Probe [6] | Semi-permeable membrane device implanted to collect dialysate from the extracellular space. | Large size (~300 μm) causes tissue damage, affecting the local chemical environment. |
| HPLC System with UV/MS Detector [5] | Analytical instrument for separating and quantifying adenosine from other compounds in microdialysate. | Required for resolving adenosine from structurally similar purine metabolites (e.g., ATP, inosine). |
| Nafion & Carbon Nanotube Coatings [5] | Electrode modifications used to enhance FSCV sensitivity for adenosine and improve selectivity over ATP. | Creates a permselective layer that can reject interfering anions. |
The choice between microdialysis and FSCV for adenosine research is not a matter of selecting a superior technology, but rather the appropriate tool for the specific biological question. Microdialysis remains the gold standard for broad-spectrum biochemical profiling and monitoring slow, tonic changes in basal adenosine levels over minutes to hours. In contrast, FSCV is unparalleled for investigating the dynamics of rapid, phasic adenosine signaling on sub-second timescales with high spatial fidelity and minimal tissue disruption. The emerging evidence of rapid adenosine signaling, uncovered primarily through FSCV, highlights how methodological capabilities can directly shape physiological understanding. Future advancements may lie in the continued refinement of electrode materials, such as diamond-based sensors [54] or geometrically optimized carbon fibers [33], and in the strategic combination of these techniques to correlate rapid electrochemical events with broader neurochemical landscapes.
The accurate measurement of dynamic biological processes, particularly in neurochemical research such as adenosine signaling, relies heavily on the temporal and spatial resolution of the analytical techniques employed. Within this domain, enzyme-linked biosensors and genetically encoded fluorescent reporters represent two distinct technological approaches with complementary strengths and limitations. This guide provides a comparative analysis of these methodologies, contextualized within the established framework of microdialysis and fast-scan cyclic voltammetry (FSCV) for adenosine research. For researchers and drug development professionals, selecting the appropriate technique is paramount, as the choice directly influences the observable timescales, detectable analyte spectrum, and ultimately, the biological conclusions that can be drawn. Enzyme-based biosensors leverage the specificity of biological recognition coupled with a physicochemical transducer, enabling them to detect a wide range of analytes with high selectivity [66]. In parallel, fluorescent biosensors, particularly those that are genetically encoded, offer unparalleled spatiotemporal resolution for monitoring molecular events in live cells, tracking changes in localization, intensity, or spectral properties of fluorescence [67] [68]. Understanding the performance benchmarks of these emerging technologies against traditional mainstays like microdialysis and FSCV provides a critical foundation for experimental design in modern biosensing applications.
Technical Performance Benchmarking
The comparative performance of enzyme-linked biosensors, fluorescent reporters, microdialysis, and FSCV is defined by key operational parameters. The following tables summarize these critical metrics to facilitate a direct comparison.
Table 1: Overall Technique Comparison for Adenosine and General Molecule Detection
| Technique | Temporal Resolution | Spatial Resolution | Key Analytes | Primary Advantages | Primary Limitations |
|---|---|---|---|---|---|
| Enzyme-Linked Biosensors [5] [66] | ~2 seconds | Low to Moderate (tissue region) | Neurotransmitters, Metabolites, Pollutants | High specificity, cost-effective, portable | Enzyme instability, limited lifespan, interference in complex matrices |
| Fluorescent Reporters (Genetically Encoded) [69] [67] [70] | Seconds to Minutes (e.g., 4 min for RBF) [69] | High (subcellular) | Ions, Metabolites, Neurotransmitters, Signaling Molecules | High spatiotemporal resolution, subcellular targeting, multiplexing potential | Requires genetic engineering, photobleaching, lower time resolution than FSCV |
| Microdialysis [5] [6] | Minutes (>5-10 min sampling) | Low (tissue region) | Broad spectrum of small molecules | Broad analyte scope, gold standard for slow changes | Low temporal/spatial resolution, tissue damage, invasive |
| Fast-Scan Cyclic Voltammetry (FSCV) [5] [8] | Sub-second (100 ms) | High (discrete brain regions) | Electroactive molecules (e.g., Adenosine, Dopamine) | Excellent temporal resolution, direct detection | Limited to electroactive species, challenging analyte identification |
Table 2: Direct Comparison of Biosensor Technologies
| Parameter | Enzyme-Linked Biosensors | Fluorescent Reporters |
|---|---|---|
| Detection Mechanism | Catalytic reaction producing detectable product (e.g., H₂O₂) [66] | Conformational change altering fluorescence (intensity, FRET, lifetime) [67] [68] |
| Key Example | Amperometric adenosine biosensor (2 s response) [5] | R-eLACCO2.1 (extracellular L-lactate), Guanidine RBF biosensor (4 min response) [70] [69] |
| Sensitivity | nM to µM range [66] | nM to mM range (dependent on biosensor affinity) [70] [68] |
| Multiplexing Capacity | Low, due to limited spectral outputs | High, via spectral separation (e.g., R-eLACCO2.1 with GCaMP) [67] [70] |
| In Vivo Applicability | Good, but prone to biofouling and interference | Excellent, with genetic targeting to specific cells/compartments [68] |
Experimental Protocols and Methodologies
Protocol for Enzyme-Linked Adenosine Biosensor Measurement
Amperometric enzyme-based biosensors for adenosine detection offer a balance of specificity and temporal resolution, operating on a timescale of seconds [5].
- Biosensor Construction: A platinum electrode is coated with a multi-enzyme cascade including adenosine deaminase, purine nucleoside phosphorylase, and xanthine oxidase. This cascade metabolizes adenosine to ultimately produce hydrogen peroxide (H₂O₂) [5].
- Signal Transduction: H₂O₂ is oxidized at the electrode surface, typically held at +0.5 V (vs. Ag/AgCl), generating a measurable current that is proportional to the ambient adenosine concentration [5].
- In Vivo Implantation and Measurement: The biosensor is implanted into the target brain region. Artificial cerebrospinal fluid (aCSF) can be perfused locally. The oxidation current is continuously recorded, and changes are correlated with biological stimuli or pharmacological interventions [5].
- Data and Analysis: Data is presented as a current-time plot. The sensor requires calibration to convert current (nA) to adenosine concentration (µM). The ~2-second response time is suitable for tracking adenosine shifts on the minute-timescale, such as during ischemic events [5].
Protocol for Genetically Encoded Fluorescent Biosensor Imaging
This protocol uses the red fluorescent extracellular L-lactate biosensor R-eLACCO2.1 as a paradigm, demonstrating the workflow for high-resolution imaging of metabolite dynamics [70].
- Biosensor Delivery and Expression: The gene encoding R-eLACCO2.1 is delivered to the target cells or tissue (e.g., the mouse somatosensory cortex) using viral vectors or transgenesis. The biosensor is engineered with an N-terminal leader sequence (e.g., Igκ) and a C-terminal glycosylphosphatidylinositol (GPI) anchor to ensure robust expression on the extracellular face of the plasma membrane [70].
- In Vivo Imaging Setup: A cranial window is established in an anesthetized or awake, behaving mouse. The region of interest is imaged using a two-photon or confocal microscope equipped with lasers suitable for exciting the red fluorophore (cpmApple in R-eLACCO2.1) [70].
- Signal Acquisition and Stimulation: Fluorescence is recorded over time (e.g., at 1-10 frames per second). To monitor activity-dependent changes, a physiological stimulus (e.g., whisker deflection or initiation of locomotion) is applied during imaging [70].
- Data Processing and Analysis: Fluorescence intensity (F) is measured in the region of interest. The response is calculated as ΔF/F₀, where F₀ is the baseline fluorescence. For biosensors compatible with fluorescence lifetime imaging (FLIM), changes in the nanosecond-scale fluorescence lifetime provide a ratiometric and more quantitative readout [70] [68]. Multiplexing is achieved by co-expressing a green biosensor like GCaMP to simultaneously record calcium activity and lactate dynamics [70].
Visualization of Techniques and Workflows
Biosensor Technology Decision Workflow
The following diagram illustrates the logical decision process for selecting the appropriate biosensing technology based on key experimental requirements.
Fluorescent Biosensor Mechanism
This diagram depicts the general working mechanism of a single-fluorophore, intensity-based genetically encoded biosensor, such as R-eLACCO2.1 or the guanidine RBF biosensor [69] [70] [68].
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of biosensor technologies requires specific reagents and instrumentation. The following table details key solutions for researchers in this field.
Table 3: Key Research Reagent Solutions for Biosensor Applications
| Item Name | Function/Description | Example Application |
|---|---|---|
| Guanidine-I Riboswitch Aptamer [69] | Biological recognition element for guanidine, fused to fluorogenic aptamer (Spinach2) in RBF biosensors. | Sensing environmental guanidine pollutants; studying cellular guanidine transport. |
| R-eLACCO2.1 Plasmid/Virus [70] | Genetically encoded red fluorescent biosensor for extracellular L-lactate. | Investigating lactate dynamics in vivo (e.g., testing ANLS hypothesis); multiplexed imaging with GCaMP. |
| CD59 GPI Anchor & Igκ Leader [70] | Genetic tags for efficient targeting and anchoring of biosensors to the extracellular plasma membrane. | Ensuring correct surface localization of extracellular biosensors like R-eLACCO2.1 for accurate measurement. |
| DFHBI Dye [69] | Cell-permeable, pro-fluorescent dye that binds to and activates fluorogenic RNA aptamers (e.g., Spinach2). | Enabling fluorescence turn-on in RNA-based fluorescent (RBF) biosensors for live-cell imaging. |
| Adenosine Enzyme Cocktail [5] | Immobilized enzymes (adenosine deaminase, etc.) that create a detection cascade on an electrode surface. | Fabricating amperometric biosensors for selective in vivo adenosine detection. |
| Carbon-Fiber Microelectrode [5] [6] | Miniaturized working electrode (~7 µm diameter) for high-resolution electrochemical measurements. | In vivo detection of electroactive analytes via FSCV or with enzyme-based biosensor coatings. |
The benchmarking data clearly illustrates a paradigm of technological trade-offs. No single technique universally supersedes all others; rather, they provide a complementary toolkit. Enzyme-linked biosensors offer proven, cost-effective specificity for a range of analytes, while fluorescent reporters provide unparalleled resolution for visualizing dynamics in live systems. The choice hinges on the specific biological question, particularly the required temporal resolution and the need for spatial mapping.
Future developments are poised to further blur the lines between these technologies. The integration of nanozymes (synthetic nanomaterials with enzyme-like activity) can enhance the stability and performance of traditional enzyme biosensors [66]. Similarly, advances in chemigenetic biosensors, which combine self-labeling protein tags with synthetic fluorophores, offer improved photostability and access to far-red spectra, expanding multiplexing capabilities [67] [68]. Furthermore, the fusion of sensing modalities, such as the combination of electrochemical and optical readouts, presents a powerful frontier for cross-validating and enriching data collection [71]. For the researcher investigating adenosine signaling or similar dynamic processes, the future lies in a strategic, multi-faceted approach that leverages the unique strengths of each biosensing technology to build a comprehensive and accurate picture of complex biological phenomena.
The selection of an appropriate measurement technique is a fundamental determinant of success in neuroscience and drug development research. For investigators studying adenosine, a ubiquitous neuromodulator with critical roles in sleep-wake regulation, neuroprotection, and tremor control, the decision most often centers on two established methodologies: microdialysis and fast-scan cyclic voltammetry (FSCV) [8] [72]. These techniques operate on different principles, offer distinct advantages, and present unique limitations that directly influence their suitability for specific research questions. Microdialysis provides a broad-spectrum chemical analysis but with limited temporal resolution, while FSCV delivers subsecond temporal resolution for a narrower range of electroactive analytes [6] [22]. This guide provides an objective, data-driven comparison of these techniques within the specific context of adenosine research, enabling scientists to make informed decisions aligned with their experimental objectives. By understanding the performance characteristics, technical requirements, and practical constraints of each method, researchers can optimize their approach to elucidate the complex roles of adenosine in brain function and pathology.
Technical Principles and Performance Characteristics
Fundamental Operating Mechanisms
Microdialysis is a sampling technique that involves implanting a semi-permeable membrane probe into brain tissue and perfusing it with an isotonic solution. Molecules in the extracellular fluid, including adenosine, diffuse across the membrane into the perfusate, which is collected for offline analysis typically using high-performance liquid chromatography (HPLC) [6]. This technique leverages the diffusion of substances down their concentration gradients, allowing for the recovery of a wide array of neurochemicals simultaneously. The size of the probe membrane pores determines the molecular weight cutoff, enabling selective sampling of small molecules like adenosine.
In contrast, Fast-Scan Cyclic Voltammetry (FSCV) is an electrochemical detection method that employs carbon fiber microelectrodes (CFMs) implanted directly into brain tissue. For adenosine detection, a triangular waveform (typically scanning from -0.4 V to +1.5 V and back at 400 V/s) is applied to the electrode at a frequency of 10 Hz [8] [72]. When adenosine molecules contact the charged carbon fiber surface, they undergo oxidation, generating a measurable current. The resulting voltammogram provides both quantitative concentration information and a characteristic "electrochemical fingerprint" that enables discrimination between adenosine and other electroactive species like dopamine, whose oxidation peaks occur at different potentials (+1.5 V for adenosine versus +0.6 V for dopamine) [72].
Comparative Performance Specifications
The table below summarizes the key performance characteristics of each technique for adenosine measurement, based on experimental data from the literature:
Table 1: Direct Comparison of Microdialysis and FSCV for Adenosine Measurement
| Performance Characteristic | Microdialysis | Fast-Scan Cyclic Voltammetry (FSCV) |
|---|---|---|
| Temporal Resolution | Minutes to hours [22] | Subsecond (≤100 ms) [22] [72] |
| Spatial Resolution | Low (probe diameter ~300 μm) [6] | High (fiber diameter 7-30 μm, length 50-400 μm) [6] [28] |
| Detection Limit | Varies with analytical method | Demonstrated for similar molecules: 20.2 nM for melatonin [16] |
| In Vivo Applicability | Well-established for chronic measurements after stabilization period [6] | Excellent for acute measurements; chronic use limited by electrode fouling [28] [16] |
| Chemical Specificity | High (with HPLC separation) [6] | Moderate (based on oxidation potentials) [22] [72] |
| Multiplexing Capability | Broad (simultaneous detection of numerous neurochemicals) [6] | Limited to electroactive species (e.g., dopamine, adenosine, serotonin) [6] [22] |
| Tissue Damage | Significant (probe tracks visible via light microscopy) [6] | Minimal (tracks often invisible via light microscopy) [6] |
| Measurement Type | Indirect (sampling with offline analysis) | Direct, real-time detection [72] |
Quantitative Experimental Comparisons
Direct methodological comparisons reveal how technical differences translate to varied experimental outcomes:
Table 2: Experimental Data Comparison for Neurotransmitter Measurement
| Experimental Context | Microdialysis Findings | FSCV Findings |
|---|---|---|
| Dopamine Measurement Near Implant | DA concentration and TTx sensitivity varies over 24h post-implantation [6] | 90% loss in evoked DA response amplitude at 200μm from microdialysis probe [6] |
| Adenosine Detection | Requires enzymatic conversion on biosensor (slow response: ~2 seconds) [72] | Direct detection at CFM (subsecond resolution) [72] |
| Temporal Dynamics | Minute-to-minute changes in extracellular concentration [22] | Rapid, transient adenosine signaling (seconds) [8] |
| Spatial Resolution | Measures averaged concentration across probe membrane surface | Regional inhibition effects observed within 250μm radius [22] |
Experimental Design and Methodological Protocols
Adenosine Measurement Using FSCV
Protocol for FSCV-based Adenosine Detection in vivo [72]:
Electrode Fabrication: A single polyacrylonitrile-based carbon fiber (T650, 5-7μm diameter) is aspirated into a borosilicate glass capillary and pulled to a microscopic tip using a pipette puller. The exposed carbon fiber is trimmed to 50-100μm length under a dissecting microscope.
Electrochemical Pre-conditioning: Electrodes are preconditioned using FSCV sweeps (-0.4 V to +1.5 V at 400 V/s, 30 Hz) in pH 7.4 Tris buffer until stable background currents are achieved.
Waveform Application: Implement a triangle waveform scanned between -0.4 V and +1.5 V at a rate of 400 V/s, applied at 10 Hz. The CFM is held at a bias potential of -0.4 V between scans.
Stereotactic Implantation: In anesthetized rats, implant the CFM in the target region (e.g., caudate putamen: AP +1.2, ML +2.5, DV -4.5 mm relative to bregma). Place an Ag/AgCl reference electrode in superficial cortical tissue.
Stimulation and Recording: Apply electrical stimulation to adenosine-releasing pathways (e.g., high-frequency stimulation of ventral tegmental area/substantia nigra: AP -5.3, ML +1.2, DV -7.5 mm).
Data Acquisition and Analysis: Record oxidation currents at peak potentials (+1.5 V for adenosine). Identify adenosine via its characteristic voltammogram and distinguish from interferents using principal component analysis.
Adenosine Measurement Using Microdialysis
Protocol for Microdialysis-based Adenosine Measurement [6]:
Probe Selection and Preparation: Select a microdialysis probe with appropriate molecular weight cutoff (typically 20-30 kDa) and membrane length (1-4 mm). Rinse the probe with sterile artificial cerebrospinal fluid (aCSF) before implantation.
Surgical Implantation: Anesthetize the animal and secure in a stereotactic frame. Perform craniectomy and implant the microdialysis guide cannula above the target region. Allow at least 24-48 hours post-surgical recovery before experiments to stabilize tissue response to implantation injury.
Probe Insertion and Perfusion: On the experimental day, carefully insert the microdialysis probe through the guide cannula. Begin perfusing with aCSF at a low flow rate (0.5-2 μL/min) using a precision syringe pump.
Sample Collection: After an equilibration period (typically 60-90 minutes), collect dialysate samples at defined intervals (10-30 minutes) into vials containing preservative if necessary. Maintain sample collection on ice or at refrigerated temperatures.
Analytical Separation: Analyze samples using HPLC with UV or mass spectrometry detection. For adenosine, utilize a C18 reverse-phase column with an isocratic or gradient mobile phase (e.g., phosphate buffer-methanol).
Quantification: Quantify adenosine levels by comparing peak areas with external standards. Apply no-net-flux method for absolute concentration determination if required.
Advanced Applications and Integration Approaches
Multiplexed Neurochemical Monitoring
No single technique provides a complete picture of complex neurochemical interactions. Consequently, researchers are increasingly developing multiplexed approaches that combine the strengths of multiple methods:
FSCV with Genetically Encoded Sensors: A recent innovative approach multiplexed FSCV with the genetically encoded glutamate sensor iGluSnFR3.v857 to simultaneously monitor adenosine, dopamine, and glutamate in brain slices [22]. This integration revealed a transient inhibitory effect of adenosine on both dopamine and glutamate release within a specific spatial range (250μm), mediated by A1 receptors. The combination allowed investigators to correlate subsecond adenosine dynamics (via FSCV) with glutamate fluctuations (via fluorescence) that would be inaccessible to FSCV alone.
FSCV with Microdialysis Validation: In melatonin detection studies, researchers used microdialysis to validate FSCV measurements, confirming that concentrations measured by FSCV (2.3±0.4μM) aligned with those obtained via microdialysis sampling [16]. This hybrid approach leveraged the temporal resolution of FSCV with the analytical specificity of microdialysis, providing enhanced confidence in the experimental findings.
Technical Innovations Addressing Methodological Limitations
Both techniques continue to evolve through technical innovations that address their inherent limitations:
Advanced Electrode Designs for FSCV: Conventional 7μm carbon fiber microelectrodes face limitations in mechanical durability and chronic performance. Recent research demonstrates that 30μm cone-shaped CFMEs offer a 3.7-fold improvement in dopamine detection sensitivity and significantly reduced glial activation (based on Iba1 and GFAP markers) compared to standard designs [28]. This geometric modification mitigates insertion-induced tissue damage while improving mechanical robustness for long-term measurements.
Waveform Optimization and Background Stabilization: Electrode fouling and background drift have traditionally limited FSCV measurement durations to <90 seconds. Through extended electrochemical preconditioning protocols and optimized waveforms, researchers have achieved stable melatonin detection for up to 3 minutes in vivo [16]. Similar approaches can be adapted for adenosine measurements to extend monitoring periods.
Decision Framework and Research Reagent Solutions
Technique Selection Algorithm
The following structured decision framework supports researchers in selecting the appropriate methodology based on specific experimental requirements:
Define Temporal Requirements: If studying rapid adenosine signaling dynamics (subsecond to seconds), FSCV is the mandatory choice [8]. For minute-to-hour scale fluctuations or baseline measurements, either technique is suitable, with microdialysis offering better stability for extended recordings.
Identify Spatial Resolution Needs: For mapping discrete brain regions with precision or investigating microdomains of adenosine release, FSCV with carbon fiber microelectrodes provides superior spatial resolution [6] [22]. For regional measurements averaging larger tissue volumes, microdialysis is appropriate.
Determine Multiplexing Requirements: If studying adenosine in isolation or with other electroactive species (dopamine, serotonin), FSCV is sufficient. If simultaneous measurement of multiple non-electroactive analytes (glutamate, GABA, neuropeptides) is required, microdialysis with HPLC is indicated [6] [22].
Consider Tissue Impact: For acute experiments where minimal tissue disruption is critical, FSCV causes significantly less damage [6]. For chronic preparations, despite initial implantation injury, microdialysis probes are suitable after the appropriate stabilization period (typically 24 hours).
Evaluate Analytical Complexity: FSCV provides immediate results with real-time analysis capabilities, while microdialysis requires subsequent offline analysis with associated equipment and expertise.
Essential Research Reagent Solutions
Successful implementation of either technique requires specific reagents and instrumentation:
Table 3: Essential Research Reagents and Materials
| Item | Function/Purpose | Technical Specifications |
|---|---|---|
| Carbon Fiber (T-650) | FSCV working electrode material | Polyacrylonitrile-based, 5-30μm diameter [28] [72] |
| Borosilicate Glass Capillaries | Electrode insulation and structural support | 0.4mm ID, 0.6mm OD [16] |
| Ag/AgCl Reference Electrode | Stable reference potential for FSCV | Teflon-coated silver wire, chloridized [72] |
| Microdialysis Probes | Semi-permeable membrane for solute sampling | ~300μm diameter, various membrane lengths [6] |
| Artificial CSF | Physiological perfusion fluid for both techniques | Isotonic, pH 7.4, containing essential ions [16] |
| Adenosine Standard | Quantitative calibration | High-purity, HPLC-grade for standard curves |
| DPCPX Antagonist | A1 receptor blockade for mechanistic studies | Validates adenosine receptor specificity [22] |
| WINCS System | Wireless FSCV data acquisition | FDA-recognized medical device standards [72] |
Microdialysis and FSCV represent complementary rather than competing approaches in adenosine research. The decision framework presented here enables researchers to align methodological selection with specific experimental questions, recognizing that technical capabilities directly constrain scientific insight. Microdialysis provides unparalleled chemical breadth for mapping complex neurochemical networks, while FSCV delivers unmatched temporal and spatial resolution for decoding rapid adenosine signaling dynamics. Emerging hybrid approaches that combine these techniques with genetic sensors and advanced electrode designs represent the future of neurochemical monitoring, offering multidimensional measurement capabilities that progressively overcome the limitations of individual methods. Through strategic technique selection based on performance characteristics and experimental requirements, researchers can optimize their approach to unravel the complex functions of adenosine in brain physiology and pathology.
Conclusion
Microdialysis and FSCV are not competing but complementary techniques that illuminate different facets of adenosine signaling. Microdialysis excels at providing a broad neurochemical profile and measuring stable, tonic levels, while FSCV is unparalleled for capturing rapid, phasic adenosine transients on a sub-second timescale. The choice between them must be guided by the specific research question—whether it concerns slow, homeostatic processes or rapid, activity-dependent neuromodulation. Future directions will be shaped by the growing trend of multiplexing these techniques with genetically encoded sensors and other modalities, offering a more integrated and holistic view of adenosine's complex roles in health, disease, and as a target for next-generation neurotherapeutics.
References
- 1. Adenosine as a neuromodulator in neurological diseases [https://pmc.ncbi.nlm.nih.gov/articles/PMC2950121/]
- 2. Adenosine as a neuromodulator in neurological diseases [https://www.sciencedirect.com/science/article/abs/pii/S1471489207001580]
- 3. Increased Cortical Extracellular Adenosine Correlates with Seizure Termination - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4104491/]
- 4. Neuroprotection by adenosine in the brain: From A1 receptor ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2096528/]
- 5. Fast-scan Cyclic Voltammetry for the Characterization of Rapid Adenosine Release - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4720017/]
- 6. A review of the effects of FSCV and microdialysis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4437820/]
- 7. Review—Recent Advances in FSCV Detection of Neurochemicals via Waveform and Carbon Microelectrode Modification - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8186302/]
- 8. Fast-scan Cyclic Voltammetry for the Characterization of ... [https://www.sciencedirect.com/science/article/pii/S2001037014000567]
- 9. Early changes in transient adenosine during cerebral ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0196932]
- 10. The progress and prospects of targeting the adenosine ... [https://biomarkerres.biomedcentral.com/articles/10.1186/s40364-025-00784-0]
- 11. Microdialysis [https://en.wikipedia.org/wiki/Microdialysis]
- 12. Microdialysis—theoretical background and recent ... [https://www.sciencedirect.com/science/article/pii/S0928098705000564#!]
- 13. Microdialysis as an Important Technique in Systems ... [https://link.springer.com/article/10.1208/s12248-017-0108-2]
- 14. Overview of Microdialysis - PMC - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC2538639/]
- 15. Overview of Brain Microdialysis - PMC - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC2953244/]
- 16. Real-Time Fast Scan Cyclic Voltammetry Detection and ... [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2020.602216/full]
- 17. Increased cortical extracellular adenosine correlates with ... [https://pubmed.ncbi.nlm.nih.gov/24483230/]
- 18. Fast-scan cyclic voltammetry [https://en.wikipedia.org/wiki/Fast-scan_cyclic_voltammetry]
- 19. Fast Scan Cyclic Voltammetry: Chemical Sensing in the Brain ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5750125/]
- 20. Measurement of Dopamine Using Fast Scan Cyclic ... [https://en.bio-protocol.org/en/bpdetail?id=2473&type=0]
- 21. Fundamentals of Fast-Scan Cyclic Voltammetry for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7028514/]
- 22. Multiplexing FSCV and iGluSnFR3 Sensors Reveals ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12230777/]
- 23. Fast Scan Cyclic Voltammetry - an overview [https://www.sciencedirect.com/topics/chemistry/fast-scan-cyclic-voltammetry]
- 24. Real-time monitoring of extracellular adenosine using enzyme ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7032657/]
- 25. An improved LC–S/MS method for the quantitation of ... [https://www.sciencedirect.com/science/article/abs/pii/S0731708512002701]
- 26. Microfabricated FSCV-Compatible Microelectrode Array for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2975426/]
- 27. Microdialysis update: optimizing the advantages - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC2269072/]
- 28. Improved longevity and in vivo performance of ... [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2025.1579380/full]
- 29. Experimental and numerical investigation of microdialysis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11223630/]
- 30. Protocol to study adenosine release in the adult mouse ... [https://www.sciencedirect.com/science/article/pii/S2666166725004022]
- 31. Protocol to study adenosine release in the adult mouse ... [https://pubmed.ncbi.nlm.nih.gov/40742815/]
- 32. Resolution of tonic concentrations of highly similar ... [https://www.nature.com/articles/s41380-024-02537-1]
- 33. Improved longevity and in vivo performance of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12411426/]
- 34. Carbon microelectrodes for the measurement of ... [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2025.1569508/full]
- 35. Characterization of Spontaneous, Transient Adenosine ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0087165]
- 36. Molecular Simulations and Drug Discovery of Adenosine ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9000248/]
- 37. Adenosine signalling to astrocytes coordinates brain ... [https://www.nature.com/articles/s41586-024-07611-w]
- 38. Different cellular sources and different roles of adenosine [https://www.sciencedirect.com/science/article/abs/pii/S0197018607001696]
- 39. Measurement of the endogenous adenosine concentration ... [https://pubmed.ncbi.nlm.nih.gov/18855606/]
- 40. Fluorescence Imaging of Neural Activity, Neurochemical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9940643/]
- 41. Decoding Molecular Network Dynamics in Cells: Advances ... [https://www.mdpi.com/2079-6374/15/9/614]
- 42. Dexamethasone-Enhanced Microdialysis and Penetration ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7752925/]
- 43. The foreign body response to biomaterial implants is ... [https://pubmed.ncbi.nlm.nih.gov/40516838/]
- 44. Allometrically scaling tissue forces drive pathological ... [https://www.nature.com/articles/s41551-023-01091-5]
- 45. Retrodialysis of Pharmacological Agents Mitigates Tissue ... [http://d-scholarship.pitt.edu/24621/]
- 46. Experimental Insights and Recommendations for Successfully ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12033007/]
- 47. Impact of protein binding on drug sampling by ... [https://www.sciencedirect.com/science/article/abs/pii/S1773224725004411]
- 48. Mechanical stimulation evokes rapid increases in extracellular ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4065624/]
- 49. Extracellular ATP/adenosine dynamics in the brain and its ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2023.1343653/full]
- 50. Advancing glassy carbon microelectrode arrays for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12492495/]
- 51. Stabilized carbon coating on microelectrodes for scalable ... [https://www.nature.com/articles/s41467-025-58388-z]
- 52. Microfabrication and In Vivo Performance of a Microdialysis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5111822/]
- 53. Automated Algorithm for Detection of Transient Adenosine ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5312768/]
- 54. In vitro adenosine detection with a diamond-based sensor [https://www.sciencedirect.com/science/article/abs/pii/S0925963505003122]
- 55. CADs for a Troubleshoot-Free Flow Cell for FSCV ... [https://www.hashemilab.com/post/novel-experimental-and-analysis-strategies-for-fast-voltammetry-2-a-troubleshoot-free-flow-cell]
- 56. Recent Advances in Fast-Scan Cyclic Voltammetry - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC7028521/]
- 57. Sampling phasic dopamine signaling with fast-scan cyclic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4311885/]
- 58. Defining a Path Toward the Use of Fast-Scan Cyclic ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2021.728092/full]
- 59. Software for near-real-time voltammetric tracking of tonic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9537688/]
- 60. Tracking Carbon Microelectrode Impedance during Fast- ... [https://pubmed.ncbi.nlm.nih.gov/40345216/]
- 61. A high-performance fluorescent sensor spatiotemporally ... [https://www.nature.com/articles/s41467-025-59530-7]
- 62. Continuous monitoring of adenosine and its metabolites ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6774626/]
- 63. Chapter 3.3 Improvement of the temporal resolution ... [https://www.sciencedirect.com/science/article/pii/S1569733906160154]
- 64. In vivo Monitoring of Dopamine by Microdialysis with One ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4835028/]
- 65. Acute and Chronic Electrodes for in Vivo Fast-Scan Cyclic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5783156/]
- 66. Advances in Enzyme-Based Biosensors: Emerging Trends ... [https://www.mdpi.com/2673-4591/106/1/5]
- 67. Advances in Multiplexed Live Imaging of Fluorescent Biosensors [https://pmc.ncbi.nlm.nih.gov/articles/PMC12467888/]
- 68. A guide to genetically-encoded redox biosensors [https://www.sciencedirect.com/science/article/abs/pii/S0003986124001887]
- 69. Guanidine Biosensors Enable Comparison of Cellular Turn ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7985839/]
- 70. A red fluorescent genetically encoded biosensor for in vivo ... [https://www.nature.com/articles/s41467-025-64484-x]
- 71. Emergent Biosensing Technologies Based on ... [https://www.mdpi.com/1424-8220/21/3/906]
- 72. Comonitoring of adenosine and dopamine using the Wireless ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2852872/]