Illuminating Synaptic Dynamics: A Comprehensive Guide to FRET-Based Protein Sensors for Neurotransmitter Release
This article provides a detailed guide for researchers and drug development professionals on Förster Resonance Energy Transfer (FRET)-based protein sensors for monitoring neurotransmitter release.
Illuminating Synaptic Dynamics: A Comprehensive Guide to FRET-Based Protein Sensors for Neurotransmitter Release
Abstract
This article provides a detailed guide for researchers and drug development professionals on Förster Resonance Energy Transfer (FRET)-based protein sensors for monitoring neurotransmitter release. We cover the foundational principles of FRET biosensor design, including donor-acceptor fluorophore pairs and neurotransmitter-binding domains. The methodological section explores practical applications, from live-cell imaging to in vivo measurements. We address critical troubleshooting and optimization strategies for signal fidelity and specificity. Finally, we present a comparative analysis against alternative techniques (e.g., electrophysiology, electrochemical methods, iGluSnFR), evaluating validation protocols, spatiotemporal resolution, and limitations. This resource aims to equip scientists with the knowledge to implement and advance these powerful tools for studying synaptic communication and neurological disorders.
Understanding FRET Biosensors: Core Principles and Molecular Design for Neurotransmitter Detection
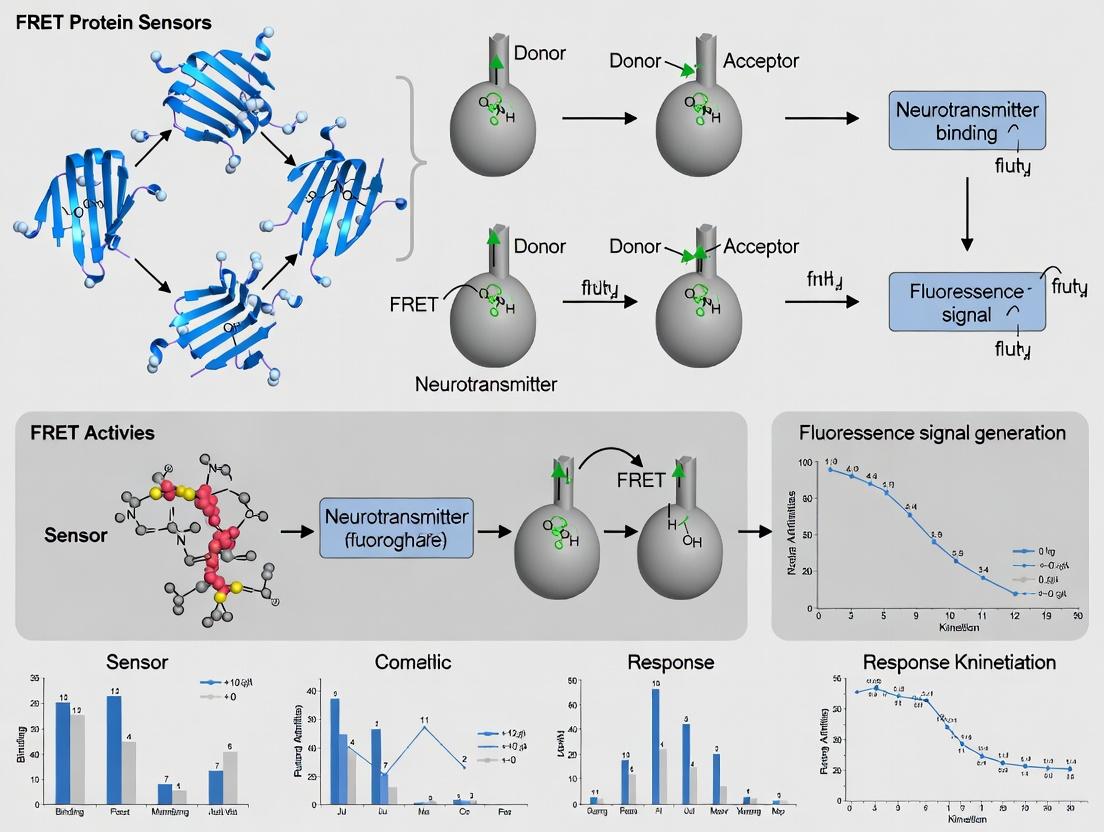
This Application Note details the principles and protocols of Förster Resonance Energy Transfer (FRET) within the broader thesis research focused on developing genetically encoded FRET-based protein sensors for monitoring real-time neurotransmitter release in synaptic clefts. The ability of FRET to act as a molecular ruler (1-10 nm) makes it indispensable for reporting dynamic protein-protein interactions and conformational changes in live neurons, a key requirement for studying the spatiotemporal dynamics of neurotransmission in health and disease.
FRET Mechanism & Quantitative Foundations
FRET is a non-radiative energy transfer from an excited donor fluorophore to a proximal acceptor fluorophore via dipole-dipole coupling. The efficiency of transfer (E) is exquisitely sensitive to the inverse sixth power of the distance (r) between the donor and acceptor, described by: E = 1 / [1 + (r/R₀)⁶] where R₀ is the Förster distance at which efficiency is 50%.
Table 1: Key FRET Quantitative Parameters for Common Pairs in Neurobiology
| Fluorophore Pair | Donor Ex (nm) | Acceptor Em (nm) | R₀ (nm) | Dynamic Range (ΔE) | Typical Use in Protein Sensors |
|---|---|---|---|---|---|
| CFP / YFP (e.g., Cerulean, Venus) | ~433 | ~528 | 4.9 - 5.2 | ~0.3 | Cameleon Ca²⁺ sensors, SNARE complex assembly |
| GFP / RFP (e.g., GFP, mCherry) | ~488 | ~610 | 5.1 - 5.5 | ~0.25 | General protein-protein interaction probes |
| Cy3 / Cy5 | ~550 | ~670 | 5.6 - 6.0 | ~0.35 | In vitro single-molecule studies of synaptic vesicles |
| T-Sapphire / dTomato | ~399 | ~581 | ~4.8 | ~0.28 | pH-sensitive synaptic vesicle release probes |
| Clover / mRuby2 | ~486 | ~605 | 5.8 - 6.2 | ~0.4 | High-signal variant for glutamate sensors (iGluSnFR) |
Application Notes: FRET-Based Neurotransmitter Release Sensors
The core thesis leverages FRET sensors designed as conformational switches. Neurotransmitter binding (e.g., glutamate, GABA) induces a structural change in a periplasmic binding protein (PBP) domain, altering the distance/orientation between fused donor and acceptor fluorescent proteins (FPs).
Key Design Considerations:
- Targeting: Sensors must be targeted to the extracellular membrane face (e.g., via GPI anchor, transmembrane domain) to report synaptic release.
- Kinetics: Sensor on/off rates must exceed the kinetics of neurotransmitter diffusion and reuptake.
- Affinity: Sensor Kd must match the expected millimolar range of synaptic cleft transmitter concentration.
- Photostability: Essential for prolonged imaging of neuronal activity.
Detailed Experimental Protocols
Protocol 4.1: Live-Cell FRET Imaging of Synaptic Glutamate Release
Objective: To measure action-potential-evoked glutamate release using a membrane-targeted iGluSnFR-3 variant in cultured hippocampal neurons.
Materials: See "The Scientist's Toolkit" (Section 6).
Methodology:
- Cell Preparation: Plate rat hippocampal neurons (DIV 0-1) on poly-D-lysine coated glass-bottom dishes. Transfect at DIV 7-10 with plasmid encoding the FRET-based glutamate sensor (e.g., super-ecliptic iGluSnFR-PM) using a calcium phosphate method or lipofection.
- Imaging Setup (Day 14-21): Use an epifluorescence or confocal microscope equipped with:
- 430/24 nm and 500/20 nm excitation filters for donor and acceptor direct excitation.
- A 455 nm dichroic mirror.
- Emission filters: 483/32 nm (donor, CFP) and 542/27 nm (acceptor, YFP).
- A fast perfusion system for buffer exchange.
- A field stimulation electrode for electrophysiological triggering.
- Acquisition:
- Maintain cells in HEPES-buffered saline (HBS) at 32°C.
- Acquire donor (IDD) and FRET (IDA) channel images simultaneously using a dual-view emission splitter or sequentially with <500 ms intervals.
- Acquire a reference acceptor (IAA) channel image via direct 500 nm excitation.
- Apply a 1 ms, 40 mA field stimulus at 10-20 Hz for 1-2 s to trigger action potentials.
- Record at 5-20 Hz frame rate.
- FRET Calculation & Analysis:
- Correct for background, bleed-through (crosstalk), and direct acceptor excitation.
- Calculate FRET ratio as R = IDA / IDD or normalized FRET efficiency (E) using acceptor sensitization methods.
- Plot ΔR/R₀ or ΔE over time to visualize glutamate transients.
Protocol 4.2: Acceptor Photobleaching FRET for Sensor Validation
Objective: To confirm FRET occurrence and quantify baseline efficiency in fixed or live cells expressing the sensor.
Methodology:
- Image a region of interest (ROI) containing the sensor-expressing membrane.
- Acquire pre-bleach donor (IDpre) and FRET channel images.
- Bleach the acceptor fluorophore in the ROI using high-intensity laser light at the acceptor's excitation peak (e.g., 514 nm for YFP) for 30-60 seconds.
- Acquire post-bleach donor (IDpost) image immediately.
- Calculate FRET Efficiency: E = 1 - (IDpre / IDpost). A significant increase in donor fluorescence after bleaching indicates positive FRET.
Visualizations
Title: The FRET Energy Transfer Process
Title: FRET Sensor Mechanism for Neurotransmitter Detection
Title: Live-Cell FRET Imaging Protocol Workflow
The Scientist's Toolkit: Key Reagent Solutions
| Item | Function & Role in FRET Neurotransmitter Research |
|---|---|
| Genetically Encoded FRET Sensors (e.g., iGluSnFR, GABA-SnFR, dLight) | Engineered fusion proteins containing a neurotransmitter-binding domain flanked by donor/acceptor FPs. The core reporting tool. |
| Poly-D-Lysine Coated Dishes | Provides a positively charged substrate for optimal adhesion and growth of primary hippocampal neurons. |
| Neurobasal/B-27 Culture Medium | Serum-free medium formulation optimized for long-term survival and health of primary neurons. |
| Calcium Phosphate Transfection Kit | Efficient method for plasmid DNA delivery into post-mitotic primary neurons, crucial for sensor expression. |
| HEPES-Buffered Saline (HBS) Imaging Solution | Maintains physiological pH (7.4) outside a CO₂ incubator during live-cell imaging. |
| Tetrodotoxin (TTX) | Sodium channel blocker. Negative control to confirm that detected signals are action-potential dependent. |
| CNQX/D-AP5 (or other receptor antagonists) | Blocks postsynaptic ionotropic glutamate receptors. Ensures sensor signal originates from released transmitter, not secondary network activity. |
| Sulforhodamine 101 (SR101) | Used in acute brain slices to selectively stain astrocytes, aiding in the identification of neuronal structures for ROI selection. |
| Alexa Fluor 594-conjugated α-bungarotoxin | Labels nicotinic acetylcholine receptors at neuromuscular junctions, useful for identifying specific synaptic regions in certain models. |
This Application Note details the design, optimization, and use of genetically encoded FRET-based sensors for monitoring real-time neurotransmitter dynamics in vitro and in vivo. These sensors are central to a thesis focused on elucidating the spatiotemporal precision of synaptic transmission and the effects of pharmacological agents. The core sensor architecture consists of a specific neurotransmitter-binding domain, a conformational-coupling linker, and a paired donor-acceptor fluorophore system.
Core Components: Function and Selection Criteria
Binding Domain
The binding domain confers specificity. It is typically derived from native neurotransmitter receptors or bacterial periplasmic binding proteins engineered for high affinity and selectivity.
Table 1: Common Binding Domains for Neurotransmitter FRET Sensors
| Neurotransmitter | Typical Binding Domain Source | Engineered Affinity (Kd, nM) | Key Selectivity Feature |
|---|---|---|---|
| Glutamate | Glutamate receptor ion channel (GluA2) | 100 - 10,000 | Distinguishes from Aspartate |
| GABA | GABAB receptor | 500 - 5,000 | Low affinity for glycine |
| Dopamine | Dopamine receptor D2, bacterial PBP | 50 - 5,000 | Varies by subtype (D1 vs D2) |
| Acetylcholine | Muscarinic (M3) receptor | 10 - 1,000 | Muscarinic vs. Nicotinic |
| Serotonin | Serotonin receptor (5-HT1A) | 200 - 2,000 | High selectivity over catecholamines |
Linker
The linker connects the binding domain to the fluorescent proteins (FPs) and transduces the binding-induced conformational change into a change in FRET efficiency. Optimal length and rigidity are empirically determined.
Table 2: Linker Design Parameters and Impact
| Parameter | Options | Impact on Sensor Performance |
|---|---|---|
| Length | 5-25 amino acids | Shorter linkers often yield larger ∆FRET |
| Composition | Flexible (GGGGS), Rigid (EAAAK) | Flexible: larger dynamic range; Rigid: faster kinetics |
| Cleavage Site | Protease-sensitive (e.g., TEV) | Allows for modular assembly and testing |
Fluorophore Pair
The donor and acceptor fluorescent proteins are chosen for spectral overlap (high Förster radius, R0), brightness, photostability, and minimal cross-talk.
Table 3: Common FRET Fluorophore Pairs for Neurotransmitter Sensors
| Donor | Acceptor | Förster Radius (R0, Å) | Typical ∆FRET (%) | Advantages |
|---|---|---|---|---|
| ECFP | Venus | ~49 | 10-25% | Classic pair, well-characterized |
| Cerulean | Citrine | ~52 | 15-30% | Improved brightness & photostability |
| mTurquoise2 | cpVenus | ~59 | 20-40% | High quantum yield, large dynamic range |
| EGFP | mRuby2 | ~54 | 15-35% | Reduced pH sensitivity, red-shifted emission |
Experimental Protocols
Protocol 1:In VitroCharacterization of Sensor Affinity and Dynamic Range
Purpose: To determine the dissociation constant (Kd) and maximum FRET response (∆FRETmax) of a purified sensor protein.
Materials:
- Purified sensor protein in HEPES-buffered saline (HBS: 20 mM HEPES, 150 mM NaCl, pH 7.4).
- Titration series of neurotransmitter stock solution (e.g., 0, 0.1, 0.5, 1, 5, 10, 50, 100, 500 µM glutamate in HBS).
- Plate reader or fluorometer capable of dual-emission readings (e.g., donor excitation at 433 nm, collect emission at 475 nm and 535 nm).
Procedure:
- Dilute purified sensor to a final concentration of 1 µM in HBS in a 96-well plate.
- For each well, add 1 µL of the appropriate neurotransmitter stock to 99 µL of sensor solution. Include a no-neurotransmitter control.
- Incubate for 5 minutes at room temperature to reach equilibrium.
- Excite the sample at 433 nm and record fluorescence intensity at 475 nm (F475, donor channel) and 535 nm (F535, acceptor channel).
- Calculate: FRET Ratio (R) = F535 / F475.
- Analyze: Plot R vs. neurotransmitter concentration [L]. Fit data to the Hill equation: R = Rmin + (Rmax - Rmin) / (1 + (Kd / [L])n), where Rmin/max are the minimum and maximum ratios, and n is the Hill coefficient.
- ∆FRET = ((Rmax - Rmin) / Rmin) * 100%.
Protocol 2: Live-Cell Imaging of Neurotransmitter Release in Cultured Neurons
Purpose: To monitor action-potential-evoked neurotransmitter release using a FRET sensor expressed in neurons or neighboring cells.
Materials:
- Primary neuronal culture (e.g., hippocampal neurons, DIV 14-21).
- Lentivirus or plasmid encoding the FRET sensor (e.g., iGluSnFR for glutamate).
- Imaging setup: Epifluorescence or confocal microscope with a dual-emission beam splitter (e.g., DV2, Photometrics), 445 nm laser or LED, 40x/1.3 NA oil objective.
- Perfusion system with modified ACSF (Artificial Cerebrospinal Fluid) and high-K+ (50 mM KCl) ACSF for stimulation.
- Field stimulation electrode (for electrically evoked release).
Procedure:
- Transduction: Transduce neurons with sensor virus at DIV 7-10. Allow 5-7 days for expression.
- Imaging Setup: Mount culture dish on the microscope stage. Continuously perfuse with normal ACSF (30°C, 5 mL/min). Focus on healthy, moderately expressing neurons.
- Acquisition: Use time-lapse imaging with 100-500 ms exposure. Acquire donor and acceptor channels simultaneously via the beam splitter.
- Stimulation: To evoke release, rapidly switch perfusion to high-K+ ACSF for 5-10 seconds, or apply a 1 ms, 20 V/cm field stimulus.
- Analysis: For each time point (t), calculate the FRET Ratio R(t) = IAcceptor(t) / IDonor(t).
- Normalize: Express data as ∆R/R0 = (R(t) - R0) / R0, where R0 is the baseline ratio before stimulation.
Visualizations
Diagram 1: Conformational change in a FRET-based neurotransmitter sensor.
Diagram 2: Workflow for live-cell imaging of neurotransmitter release.
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for FRET Sensor Development & Application
| Item / Reagent | Function / Purpose | Example Product / Note |
|---|---|---|
| Engineered PBPs or Receptor Domains | Provides the specific, high-affinity binding core for the target neurotransmitter. | GltI (for glutamate), OpuAC (for choline), Dopamine D2 receptor fragment. |
| Fluorescent Protein Variants | Donor/Acceptor pair with optimal spectral properties (high R0, brightness, photostability). | mTurquoise2 (donor), cpVenus (acceptor), mNeonGreen, mRuby3. |
| Flexible Cloning System (e.g., Gibson Assembly) | Enables rapid modular assembly of binding domain, linker, and FP gene fragments. | Commercial Gibson Assembly Master Mix. |
| HEK293T Cell Line | Standard mammalian cell line for initial sensor protein expression and in vitro characterization. | High transfection efficiency. |
| Neuronal Cell Culture Systems | Primary neurons or iPSc-derived neurons for physiological validation of sensor function. | Rat hippocampal neurons, human cortical neurons. |
| Fast Perfusion System | For rapid application and washout of neurotransmitters or drugs during imaging experiments. | Ala Scientific, Warner Instruments. |
| Dual-Emission Imaging System | Microscope setup capable of simultaneous donor/acceptor emission capture. | Photometrics DV2 beam splitter, Optosplit II. |
| Rationetric Analysis Software | Software to calculate FRET ratios (IA/ID) and ∆R/R0 from time-lapse images. | ImageJ/FIJI with Ratio Plus plugin, MetaMorph, custom Python/Matlab scripts. |
| Selective Pharmacological Agents | Agonists and antagonists to validate sensor specificity and probe endogenous receptors. | NBQX (AMPA receptor antagonist), Sulpiride (D2 antagonist). |
Application Notes
Förster Resonance Energy Transfer (FRET)-based biosensors are indispensable tools for monitoring real-time neurotransmitter release and intracellular signaling dynamics. The choice of donor-acceptor pair critically influences the sensor's performance, including its dynamic range, photostability, and compatibility with instrumentation. This article, framed within a thesis on developing FRET sensors for neurotransmitter release, details classic and modern fluorescent protein pairs.
Classic Pairs: CFP/YFP and GFP/RFP
The CFP (cyan) and YFP (yellow) pair, exemplified by the original cameleon calcium sensors, has been a cornerstone. Its spectral profile allows good separation of donor emission and acceptor excitation but suffers from pH sensitivity (YFP pKa ~6.9), vulnerability to photobleaching, and significant spectral bleed-through (SBT). The GFP/RFP pair offers a larger Stokes shift, reducing direct acceptor excitation, but early variants like GFP/DsRed had issues with acceptor oligomerization and slow maturation.
Modern Alternatives: Clover/mRuby2
Engineered for optimized FRET, Clover/mRuby2 is now a gold standard. Clover is a bright, monomeric, pH-stable GFP variant. mRuby2 is an exceptionally bright and photostable monomeric RFP. Together, they provide a high Förster radius (~5.3 nm), excellent photon output for improved signal-to-noise ratio, and reduced photobleaching, making them superior for long-term imaging of synaptic activity.
Table 1: Quantitative Comparison of Common FRET Pairs
| Pair (Donor/Acceptor) | Ex Max (nm) | Em Max (nm) | Förster Radius (R0, nm) | Brightness (Relative) | pKa (Acceptor) | Key Advantages | Key Limitations |
|---|---|---|---|---|---|---|---|
| CFP / YFP (e.g., Cerulean/Venus) | 433 / 515 | 475 / 528 | ~5.2 | Moderate | ~6.9 | Well-characterized, many existing biosensors | pH sensitive, photobleaching, high SBT |
| GFP / RFP (e.g., EGFP/mCherry) | 488 / 587 | 507 / 610 | ~5.1 | Moderate | ~4.5 | Large Stokes shift, reduced direct excitation | Lower R0, some acceptor oligomerization in early variants |
| Clover / mRuby2 | 505 / 559 | 515 / 600 | ~5.3 | High | ~5.0 | Very bright, photostable, high FRET efficiency, monomeric | Requires filters for green/red separation |
Experimental Protocols
Protocol 1: Calibration of FRET Sensor ResponseIn Vitro
This protocol outlines how to determine the maximum dynamic range of a purified FRET sensor protein.
- Protein Purification: Express the FRET sensor (e.g., a glutamate sensor with Clover/mRuby2) in E. coli and purify via His-tag affinity chromatography.
- Buffer Setup: Prepare a physiologically relevant buffer (e.g., 20 mM HEPES, 100 mM KCl, pH 7.4).
- Spectrofluorometry: Place purified protein in a cuvette. Set donor excitation (e.g., 473 nm for Clover). Record emission spectra from 500-650 nm.
- Baseline & Saturation: Record the baseline spectrum. Add saturating concentration of the analyte (e.g., 10 mM glutamate). Record the saturated spectrum.
- Calculation: Determine the emission ratio (Acceptor emission peak / Donor emission peak) for baseline (Rmin) and saturated (Rmax) states. Dynamic Range (DR) = Rmax / Rmin.
Protocol 2: Live-Cell Imaging of Neurotransmitter Release Using a FRET Sensor
This protocol describes imaging presynaptic neurotransmitter release in cultured neurons.
- Culture & Transfection: Culture hippocampal neurons from rat E18 embryos. Transfect at DIV 7-10 with the FRET sensor plasmid using calcium phosphate or lipofection.
- Imaging Setup (24-48h post-transfection): Use an epifluorescence or confocal microscope equipped with:
- A 440 nm laser (for CFP) or 473 nm laser (for Clover).
- Beam splitter and simultaneous dual-emission detection.
- Filter sets: Donor channel (e.g., 480/40 nm for CFP, 500/30 nm for Clover); Acceptor channel (e.g., 535/30 nm for YFP, 600/50 nm for mRuby2).
- Acquisition: Perfuse with aCSF at 37°C. Acquire images at 2-10 Hz. Establish a 30s baseline.
- Stimulation: Deliver field stimulation (e.g., 1 ms pulses at 20 Hz for 2s) to evoke action potentials and vesicle release.
- Analysis: For each time point t, calculate the FRET ratio R(t) = Intensity(Acceptor channel) / Intensity(Donor channel). Normalize as ΔR/R0 = [R(t) - R0] / R0, where R0 is the average baseline ratio. Plot ΔR/R0 over time.
The Scientist's Toolkit
Table 2: Essential Research Reagents & Materials
| Item | Function in FRET Sensor Research |
|---|---|
| FRET Sensor Plasmid (e.g., pCAG-Clover-mRuby2-iGluSnFR) | Encodes the genetically encoded FRET biosensor under a strong promoter for neuronal expression. |
| Neurobasal/B27 Culture Medium | Supports long-term survival and health of primary neuronal cultures. |
| Poly-D-lysine Coated Coverslips | Provides a charged substrate for neuron adhesion and growth. |
| Transfection Reagent (e.g., Lipofectamine 2000, Calcium Phosphate) | Facilitates delivery of plasmid DNA into hard-to-transfect primary neurons. |
| Tetrodotoxin (TTX) & 4-AP | Pharmacological tools to block voltage-gated Na+ channels (TTX) or K+ channels (4-AP to enhance release) for control experiments. |
| Recombinant Neurotransmitter (e.g., L-Glutamate) | Used for in vitro calibration and as a positive control for sensor application. |
| Mounting Medium with Nuclease | For immobilizing and sealing samples during imaging; nuclease prevents clogging of microfluidic perfusion systems. |
Visualizations
Diagram 1: FRET Sensor Conformational Change Mechanism
Diagram 2: Workflow for Validating a FRET Neurotransmitter Sensor
Diagram 3: Spectral Overlap Visualization of FRET Pairs
Within the broader thesis on developing FRET-based protein sensors to monitor real-time neurotransmitter release, a critical design choice concerns the sensing paradigm. This article compares two principal mechanisms: conformational change sensors and cleavage-based sensors, exemplified by the SNIFIT (Signal Amplification by Integrated-FLIT) technology. The selection of paradigm dictates sensitivity, kinetics, reversibility, and applicability in complex biological environments like synaptic clefts.
Core Principles & Comparative Analysis
Conformational Change Sensors
These are single-polypeptide sensors where ligand binding induces a conformational shift that alters the distance or orientation between a donor and acceptor fluorophore, modulating FRET efficiency. They are intrinsically reversible.
Cleavage-Based SNIFIT Sensors
SNIFITs are bipartite systems. A ligand-binding domain is anchored to the membrane, while a fluorophore-labeled “reporter” unit is recruited from the cytosol. Ligand binding creates a docking site for the reporter. A concomitant, protease cleavage event (e.g., by Tobacco Etch Virus protease, TEVp) liberates the reporter, leading to a permanent, amplified fluorescence change.
Quantitative Comparison Table
Table 1: Quantitative Comparison of Sensing Paradigms
| Parameter | Conformational Change FRET Sensors | Cleavage-Based SNIFIT Sensors |
|---|---|---|
| Reversibility | Fully reversible | Irreversible (single-use) |
| Kinetics (Response Time) | Fast (ms-s), limited by binding | Slower (min), limited by cleavage/recruitment |
| Signal-to-Noise Ratio (SNR) | Moderate (~10-50% ΔFRET) | High (Amplified, >1000% ΔFluorescence) |
| Cellular Context | Cytosol, membrane-tethered | Primarily cell surface |
| Primary Application | Real-time dynamics, kinetics | High-sensitivity endpoint detection, imaging |
| Common Neurotransmitters Detected | Glutamate (iGluSnFR), GABA, dopamine | Extracellular cAMP, IP₃ |
Table 2: Performance Metrics from Recent Studies (2023-2024)
| Sensor Type | Sensor Name | Ligand | ΔFRET/ΔF (%) | Kd / EC₅₀ | Reference |
|---|---|---|---|---|---|
| Conformational | iGluSnFR3 | Glutamate | ~400% ΔF | 4.2 µM | Marvin et al., 2023 |
| Conformational | GRABDA2h | Dopamine | ~90% ΔFRET | 130 nM | Sun et al., 2020/2022 |
| SNIFIT | SNIFIT IP₃R | IP₃ | >1000% ΔF (Cleavage) | ~10 nM | Aoki et al., 2023 |
| SNIFIT | cAMPSnif | cAMP | ~1500% ΔF (Cleavage) | 2.1 µM | Harvey et al., 2024 |
Experimental Protocols
Protocol: Validating a Conformational FRET SensorIn Vitro
Aim: To determine the ligand affinity (Kd) and dynamic range of a purified FRET-based neurotransmitter sensor.
Materials: See "The Scientist's Toolkit" (Section 6).
Procedure:
- Sensor Purification: Express the His-tagged sensor protein (e.g., glutamate sensor) in HEK293T cells or E. coli. Purify using Ni-NTA affinity chromatography. Dialyze into assay buffer (e.g., 20 mM HEPES, 150 mM NaCl, pH 7.4).
- Fluorometer Setup: Load purified sensor (100 nM final) into a quartz cuvette in a spectrofluorometer. Set thermostatic control to 37°C.
- Spectral Scan: Excite the donor (e.g., at 433 nm for CFP). Acquire emission spectra from 450-600 nm without ligand to establish baseline FRET.
- Titration: Add increasing concentrations of ligand (e.g., glutamate, from 1 nM to 10 mM) in a logarithmic series. After each addition, mix gently and record the emission spectra.
- Data Analysis: Calculate the donor/acceptor emission ratio (e.g., 475 nm/527 nm for CFP/YFP) for each spectrum. Plot the ratio against ligand concentration. Fit the data to a four-parameter logistic equation to determine EC₅₀ (~Kd) and maximum ΔRatio.
Protocol: Implementing a SNIFIT Sensor in Live Cells
Aim: To monitor cAMP production at the plasma membrane using the cAMPSnif system.
Procedure:
- Plasmid Transfection: Co-transfect HEK293 cells (or primary neurons) with three plasmids:
- Anchor: Plasmid encoding the extracellular cAMP-binding protein (e.g., EPAC1-cAMP binding domain) fused to a transmembrane domain and the TEVp recognition sequence.
- Reporter: Plasmid encoding GFP (or another fluorophore) fused to a nuclear export signal (NES) and a TEVp cleavage site.
- Protease: Plasmid encoding the constitutively active, membrane-tethered TEVp.
- Expression (24-48 hrs): Allow for protein expression and system assembly. The reporter is cytosolic but excluded from the nucleus by the NES.
- Pre-Stimulation Imaging: Using confocal or TIRF microscopy, acquire a baseline image of GFP fluorescence at the cell membrane.
- Stimulation & Cleavage: Stimulate cells with an agent that elevates extracellular cAMP (e.g., Forskolin 10 µM + IBMX 100 µM). cAMP binding to the anchor induces a conformational change that recruits the reporter into proximity with the tethered TEVp, leading to cleavage.
- Post-Stimulation Imaging: Monitor GFP fluorescence over time (5-60 min). Cleavage releases GFP from its membrane tether, causing a loss of membrane-localized fluorescence and a diffuse cytosolic signal. Quantify the decrease in membrane fluorescence intensity over time.
Signaling Pathway & Workflow Diagrams
Diagram 1: Conformational Change FRET Sensor Cycle
Diagram 2: SNIFIT Sensor Activation & Cleavage
Diagram 3: Sensor Paradigm Selection Logic
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions
| Item | Function/Description | Example Product/Catalog # |
|---|---|---|
| FRET Sensor Plasmids | Genetically encoded constructs for conformational sensors (e.g., CFP-YFP pairs). | Addgene: #41791 (iGluSnFR), #130992 (GRABDA) |
| SNIFIT Component Plasmids | Tripartite system plasmids (Anchor, Reporter, TEV Protease). | Available from leading authors (e.g., Aoki lab) or custom-built. |
| TEV Protease (Recombinant) | For in vitro validation of SNIFIT cleavage efficiency. | Thermo Fisher, AC751 |
| High-Affinity Ligand Agonists/Antagonists | For sensor calibration and control experiments. | Tocris Bioscience (e.g., Glutamate #0218, Forskolin #1099) |
| Ni-NTA Agarose | Purification of His-tagged sensor proteins for in vitro characterization. | Qiagen, #30210 |
| Fluorometer/Cuvettes | For precise in vitro spectral measurements and Kd determination. | Horiba PTI QuantaMaster, Starna Cells cuvettes |
| TIRF/Confocal Microscope | For live-cell imaging of membrane-localized sensor dynamics. | Nikon/Zeiss/Olympus systems with environmental control |
| Image Analysis Software | For quantifying FRET ratios or membrane fluorescence loss over time. | Fiji/ImageJ, MetaMorph, NIS-Elements |
This document provides application notes and protocols for FRET-based genetically encoded sensors used to monitor the dynamics of five critical neurotransmitters: Glutamate, GABA, Dopamine, Acetylcholine, and Norepinephrine. These tools are essential for a thesis focused on understanding the spatiotemporal precision of neurotransmitter release in both physiological and pathological contexts, with direct implications for neuroscience research and CNS drug development.
Table 1: Key FRET-Based Neurotransmitter Sensors
| Neurotransmitter | Sensor Name(s) | Key Domain Architecture (Ligand-Binding / FRET Pair) | Reported Affinity (Kd/EC50) | Dynamic Range (ΔR/R %) | Primary Reference (Year) |
|---|---|---|---|---|---|
| Glutamate | iGluSnFR variants (SF-iGluSnFR, iGluSnFR3) | GltI glutamate-binding protein / cpGFP | ~5 µM (SF-iGluSnFR) | ~400% (iGluSnFR3) | Marvin et al., 2018; Aggarwal et al., 2023 |
| GABA | iGABASnFR | GABA-binding protein (Atu2422) / cpGFP | ~10 µM | ~500% | Marvin et al., 2019 |
| Dopamine | dLight1, GRABDA | D2-like or D1-like dopamine receptor / cpGFP | 90 nM (dLight1.1) 130 nM (GRABDA1h) | ~340% (dLight1.3b) ~90% (GRABDA1h) | Patriarchi et al., 2018; Sun et al., 2020 |
| Acetylcholine | GACh, GRABACh | Muscarinic M3 receptor (M3R) / cpGFP | 2 µM (GACh2.0) 0.3 µM (GRABACh3.0) | ~70% (GACh2.0) ~130% (GRABACh3.0) | Jing et al., 2018; Wu et al., 2023 |
| Norepinephrine | GRABNE | α1A-adrenergic receptor / cpGFP | 80 nM (GRABNE1h) | ~90% (GRABNE1h) | Feng et al., 2019 |
Detailed Experimental Protocols
Protocol 3.1: In Vitro Characterization of Sensor Affinity and Specificity
Purpose: To determine the Kd and pharmacological profile of a FRET-based neurotransmitter sensor in a controlled system.
Materials:
- Purified sensor protein or sensor-expressing cell membrane preparation.
- Microplate reader capable of fluorescence intensity (FI) or FRET ratio measurements (e.g., with 405/485 nm excitation and 535 nm emission filters for CFP/YFP pairs).
- Agonists: Target neurotransmitter (e.g., L-Glutamate, GABA, DA, ACh, NE).
- Antagonists: Selective receptor blockers relevant to the sensor's binding domain (e.g., CNQX for iGluSnFR, Sulpiride for dLight1).
- Control neurotransmitters (e.g., glycine, serotonin) to test cross-reactivity.
- Imaging buffer (e.g., HEPES-buffered saline, pH 7.4).
Procedure:
- Sample Preparation: Dispense sensor sample into wells of a 96-well plate.
- Baseline Acquisition: Read baseline fluorescence (donor and acceptor channels for FRET; cpGFP channel for intensity-based sensors) for 1 minute.
- Dose-Response: Add increasing concentrations of the target neurotransmitter (e.g., 1 nM to 1 mM, half-log steps). Incubate for 30-60 seconds after each addition before reading fluorescence.
- Data Analysis: Plot the fluorescence response (ΔF/F0 or ΔR/R0) against ligand concentration. Fit data with a logistic equation (e.g., Hill equation) to determine EC50 (proxy for Kd).
- Specificity Test: Repeat with a fixed, near-EC80 concentration of target neurotransmitter in the presence of varying concentrations of selective antagonists or other neurotransmitters.
Protocol 3.2: Imaging Neurotransmitter Release in Acute Brain Slices
Purpose: To monitor real-time neurotransmitter transients in response to electrical or optogenetic stimulation.
Materials:
- Acute brain slice (200-300 µm thick) from a sensor-expressing animal (AAV-injected or transgenic).
- Perfusion system with oxygenated (95% O2/5% CO2) aCSF at 30-32°C.
- Upright two-photon or epifluorescence microscope equipped with appropriate lasers/filters.
- Stimulation electrodes or fiber-optic cannula for optogenetics.
- Data acquisition software (e.g., ScanImage, µManager).
Procedure:
- Slice Preparation & Recovery: Prepare slices in ice-cold, sucrose-based cutting aCSF. Recover for ≥30 min in standard aCSF at 32°C, then at room temperature for ≥1 hour.
- Microscope Setup: Place slice in perfusion chamber. For FRET sensors using CFP/YFP, excite CFP with a 445 nm laser line and collect emissions at 480/40 nm (CFP) and 535/30 nm (FRET/YFP). For intensity-based cpGFP sensors, use ~488 nm excitation.
- Baseline Recording: Acquire images at 2-10 Hz for 1-2 minutes to establish a stable baseline.
- Stimulation: Deliver a train of electrical pulses (e.g., 10 pulses at 20 Hz) to afferent pathways or a brief light pulse (1-5 ms) in slices co-expressing channelrhodopsin.
- Data Processing: Define regions of interest (ROIs). Calculate ΔF/F0 or the FRET ratio (YFP/CFP). Detect transients as peaks exceeding 5x the standard deviation of the baseline noise.
Protocol 3.3: In Vivo Fiber Photometry Recording
Purpose: To record bulk neurotransmitter signals in freely behaving animals.
Materials:
- Sensor-expressing mouse (via stereotaxic AAV injection).
- Implantable optical fiber (400 µm core diameter) and cannula.
- Fiber photometry system (LEDs for excitation, dichroic mirrors, photodetectors).
- Behavioral apparatus.
Procedure:
- Viral Injection & Fiber Implantation: Inject AAV encoding the sensor into the target brain region. Immediately implant the optical fiber ~200 µm above the injection site. Secure with dental cement.
- System Calibration: Connect the implanted fiber to the photometry system. Balance the intensities of modulated excitation lights (e.g., 405 nm isosbestic control, 470 nm excitation for cpGFP sensors).
- Behavioral Recording: Habituate the animal, then record fluorescence signals synchronously with behavioral video during tasks (e.g., reward delivery, fear conditioning).
- Signal Processing: Demodulate signals at each excitation wavelength. Calculate ΔF/F using the 405 nm channel as a reference for motion/bleaching artifacts. Align signals to behavioral events for averaging.
Visualizations
Diagram 1: Core FRET Sensor Signaling Workflow
Diagram 2: Neurotransmitter to Sensor Pairing Map
Diagram 3: Key Experimental Workflows from Prep to Analysis
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents and Materials
| Item | Function & Description | Example Vendor/Catalog |
|---|---|---|
| Sensor AAVs | Genetically encoded FRET sensor constructs packaged in Adeno-Associated Virus for in vivo delivery. | Addgene (distributes plasmids); Penn Vector Core, Virovek (for packaging) |
| High-Titer AAV Purification Kit | For concentrating and purifying AAVs to achieve high infection efficiency in brain tissue. | Takara Bio #6666 |
| Artificial Cerebrospinal Fluid (aCSF) | Ionic and pH-balanced physiological buffer for maintaining brain slice health during ex vivo experiments. | Tocris Bioscience #3525 |
| Neurotransmitter Agonists/Antagonists | Pharmacological tools for sensor validation, calibration, and perturbation experiments. | Hello Bio, Tocris, Sigma-Aldrich |
| Slice Stabilization Solution | Sucrose-based or NMDG-based cutting solutions to improve viability of acute brain slices. | Custom formulation or commercial aCSF mixes. |
| FRET-Compatible Immersion Oil | Optimized for UV/visible transmission to maximize signal collection in microscopy. | Cargille Type FF |
| Fiber Photometry System | Integrated LED excitation, filters, and detectors for in vivo fluorescence recording in behaving animals. | Tucker-Davis Technologies, Doric Lenses, Neurophotometrics |
| Analysis Software Suite | For processing time-series fluorescence data, detecting transients, and statistical analysis. | Python (SciPy, NumPy), MATLAB, MiniAnalysis, Suite2p |
Within the broader thesis on FRET-based protein sensors for monitoring neurotransmitter release, this document traces the conceptual and technical lineage from foundational biosensors for cyclic guanosine monophosphate (cGMP) and calcium (Ca²⁺) to the modern genetically encoded glutamate indicators (iGluSnFR variants). The evolution represents a paradigm shift from monitoring secondary messengers to directly imaging synaptic neurotransmitter release with high spatiotemporal resolution, crucial for neuroscience research and neuropharmacological drug development.
Key Evolutionary Milestones and Quantitative Data
Table 1: Evolutionary Timeline of Key Protein-Based Biosensors
| Sensor Generation | Example Sensor | Target | Year ~ | Key Characteristics (Kd, ΔF/F, Response Time) | Primary Innovation |
|---|---|---|---|---|---|
| First-Gen Ca²⁺ | Cameleon (YC2.1) | Ca²⁺ | 1997 | Kd: ~1.1 µM; ΔF/F: ~30% | First FRET-based GECI; calmodulin/M13 domain. |
| Early cGMP | cGES-DE5 | cGMP | 2000 | Kd: ~950 nM; ΔF/F: ~1.6 | Cyclic nucleotide-gated channel fragment. |
| Optimized Ca²⁺ | GCaMP6f | Ca²⁺ | 2013 | Kd: ~375 nM; ΔF/F: ~250%; τon: ~45 ms | cpGFP fused to CaM/M13; high sensitivity. |
| First iGluSnFR | iGluSnFR (original) | Glutamate | 2013 | Kd: ~2.1 µM; ΔF/F: ~220%; τoff: ~110 ms | GluA2 LBD inserted into cpGFP (superfolder). |
| Modern iGluSnFR | iGluSnFR3 | Glutamate | 2022 | Kd: ~4.5 µM; ΔF/F: ~600%; τoff: ~2.2 ms | Directed evolution; faster, brighter, more stable. |
Table 2: Performance Comparison of Select iGluSnFR Variants
| Variant | Apparent Kd (µM) | ΔF/F max (%) | Rise Time (ms, 20-80%) | Decay Tau (τoff, ms) | Brightness (Relative) | Key Application |
|---|---|---|---|---|---|---|
| iGluSnFR (orig) | 2.1 | ~220 | ~3 | ~110 | 1.0 | General presynaptic detection. |
| iGluSnFR-A184S | 3.2 | ~370 | ~2.5 | ~90 | 1.3 | Improved signal-to-noise in vivo. |
| iGluSnFR3 | 4.5 | ~600 | ~1.3 | ~2.2 | ~2.0 | Fast synaptic transients. |
| iGluSnFR3s (slow) | 1.7 | ~500 | ~4.6 | ~230 | ~1.8 | High-affinity, sustained signals. |
Detailed Experimental Protocols
Protocol 1: In Vitro Characterization of iGluSnFR Affinity (Kd)
Purpose: To determine the apparent dissociation constant (Kd) of an iGluSnFR variant for glutamate. Reagents: Purified iGluSnFR protein (e.g., from HEK293T expression), HEPES-buffered saline (HBS: 20 mM HEPES, 150 mM NaCl, pH 7.4), L-Glutamate stock solutions (0.1 mM to 10 mM in HBS), 96-well black-walled plate, fluorescence plate reader. Procedure:
- Dilute purified iGluSnFR in HBS to a final concentration of ~100 nM in a total volume of 100 µL per well.
- Prepare a serial dilution of glutamate in HBS covering a range from 0 to 1000 µM (e.g., 12 concentrations).
- Add 100 µL of each glutamate dilution to the sensor solution in triplicate. Incubate for 2 min at room temperature.
- Measure fluorescence (excitation 480 nm, emission 510 nm) on the plate reader.
- Fit the fluorescence intensity (F) vs. glutamate concentration [G] data to the Hill equation:
F = F_min + (F_max - F_min) * ([G]^n / (Kd^n + [G]^n))where n is the Hill coefficient, to determine the apparent Kd.
Protocol 2: Two-Photon Imaging of Glutamate Release in Acute Brain Slices with iGluSnFR3
Purpose: To image action-potential-evoked glutamate release at single synapses in rodent brain slices. Reagents: Acute brain slice from transgenic mouse or virus-injected rat (AAV-hSyn-iGluSnFR3), artificial cerebrospinal fluid (aCSF: 125 mM NaCl, 2.5 mM KCl, 1.25 mM NaH₂PO₄, 26 mM NaHCO₃, 20 mM glucose, 2 mM CaCl₂, 1 mM MgCl₂, bubbled with 95% O₂/5% CO₂), 1 µM TTX, 10 µM NBQX, 20 µM Bicuculline, two-photon microscope. Procedure:
- Slice Preparation & Maintenance: Prepare 300 µm acute hippocampal or cortical slices in ice-cold, sucrose-based cutting aCSF. Recover at 34°C for 30 min, then hold at room temperature in standard aCSF.
- Imaging Setup: Place slice in a submerged recording chamber perfused with oxygenated aCSF (30°C). Add synaptic blockers (NBQX, Bicuculline) to prevent recurrent network activity. TTX can be added for control experiments.
- Stimulation: Place a bipolar stimulating electrode in the afferent pathway (e.g., Schaffer collaterals for CA1). Use a stimulus isolator to deliver a single or brief train (e.g., 10 pulses at 20 Hz) of electrical pulses (0.1 ms, 10-100 µA).
- Image Acquisition: Using a two-photon microscope tuned to 920 nm, acquire line scans or frame scans (≥ 50 Hz) from regions of interest (ROIs) on labeled axons or dendrites.
- Analysis: Calculate ΔF/F₀ = (F - F₀) / F₀, where F₀ is the baseline fluorescence. Measure peak amplitude, rise time, and decay tau from the averaged trace of multiple trials. Align to stimulus artifact for time-locked responses.
Visualization Diagrams
Title: iGluSnFR Glutamate Sensing Mechanism
Title: Conceptual Lineage of Protein Biosensors
Title: iGluSnFR Brain Slice Imaging Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function/Application in iGluSnFR Research |
|---|---|
| AAV-hSyn-iGluSnFR3 | Adeno-associated virus with human synapsin promoter for neuron-specific expression of the fast, sensitive iGluSnFR3 variant in vivo and in slices. |
| Purified iGluSnFR Protein | Recombinant protein for in vitro calibration, characterization of affinity (Kd), and spectroscopic properties without cellular confounding factors. |
| Artificial Cerebrospinal Fluid (aCSF) | Physiological salt solution for maintaining live brain slices, providing ions and nutrients essential for neuronal health and synaptic function during imaging. |
| Tetrodotoxin (TTX) | Sodium channel blocker (1 µM) used to silence action potentials in control experiments, confirming that detected signals are action-potential-evoked. |
| NBQX (AMPA receptor antagonist) | Glutamate receptor blocker (10 µM) used to prevent postsynaptic activation and recurrent network activity, isolating presynaptic release signals. |
| Bicuculline (GABA_A antagonist) | Inhibitory receptor blocker (20 µM) used to reduce tonic inhibition in slices, often paired with NBQX to prevent epileptiform activity during stimulation. |
From Bench to Brain: Methodologies and Cutting-Edge Applications in Research
1. Introduction
Within the thesis framework of developing and applying FRET-based protein sensors for monitoring real-time neurotransmitter release, the efficacy of the research is fundamentally dependent on the successful delivery and expression of these sensor constructs into relevant cellular and animal models. This document details application notes and standardized protocols for the three primary methodologies: transfection, viral delivery, and generation of transgenic animals, providing a comparative guide for selecting the optimal approach based on experimental goals.
2. Quantitative Comparison of Delivery Methods
The choice of expression method involves trade-offs between efficiency, cell-type specificity, expression level, and experimental timeline. The following table summarizes key quantitative parameters for the three core methods.
Table 1: Quantitative Comparison of Sensor Expression Methods
| Parameter | Transient Transfection (Lipofection) | Viral Delivery (AAV) | Transgenic Animal Models |
|---|---|---|---|
| Typical In Vitro Efficiency | 70-90% (HEK293, HeLa); 20-60% (primary neurons) | >90% for permissive cells in vitro | Not Applicable (N/A) |
| Typical In Vivo Efficiency | Low (limited to accessible tissues) | High: Up to 80-95% transduction in targeted brain regions with stereotaxic injection | Ubiquitous: 100% of cells in the organism carry the transgene |
| Expression Onset | 24-48 hours | Slow: 2-4 weeks for full expression in vivo | From embryonic development |
| Expression Duration | Transient (3-7 days, diluted by division) | Long-term/Persistent (months to years) | Lifetime, heritable |
| Titer/Amount Used | 1-4 µg DNA per well (24-well plate) | In vivo: 10^8 - 10^13 vg/mL, 0.5-2 µL injection volume | N/A |
| Cell-Type Specificity | Low (depends on transfection reagent) | High (via serotype & promoter selection) | Variable (via promoter selection; can be broad or specific) |
| Cost & Timeline | Low cost, fast (days) | Moderate cost, moderate timeline (weeks for virus prep + expression) | Very high cost, long timeline (months to years for line generation) |
| Primary Application | Rapid in vitro validation of sensor function | In vivo and in vitro studies requiring stable, cell-type-specific expression | Chronic studies, developmental studies, breeding into disease models |
3. Detailed Experimental Protocols
Protocol 3.1: Lipid-Mediated Transfection of Primary Neuronal Cultures with FRET Sensor Plasmid
- Objective: To express a genetically encoded FRET-based neurotransmitter sensor (e.g., GABA, glutamate, dopamine sensor) in dissociated primary hippocampal or cortical neurons for in vitro calibration and imaging.
Materials:
- Primary neurons (DIV 5-7)
- Neurobasal/B27 maintenance medium
- Plasmid DNA encoding FRET sensor (endotoxin-free, 1 µg/µL)
- Lipofectamine 2000 or 3000 reagent
- Opti-MEM I Reduced Serum Medium
- Incubator (37°C, 5% CO2)
Procedure:
- Day of Transfection: Ensure neurons are healthy and at 60-70% confluence.
- Complex Formation: a. Dilute 1-1.5 µg of sensor plasmid DNA in 50 µL Opti-MEM. Mix gently. b. Dilute 2-3 µL of Lipofectamine reagent in a separate 50 µL Opti-MEM. Incubate for 5 minutes at RT. c. Combine the diluted DNA with the diluted Lipofectamine. Mix gently and incubate for 20-25 minutes at RT to form complexes.
- Transfection: a. While complexes form, replace the neuronal culture medium with fresh, pre-warmed Neurobasal/B27 medium. b. Add the 100 µL DNA-lipid complex dropwise to the culture dish. Gently swirl the plate.
- Incubation & Expression: Return cells to the incubator. Sensor expression can typically be assessed via live-cell FRET imaging 48-72 hours post-transfection.
Protocol 3.2: Stereotaxic Intracranial Injection of Adeno-Associated Virus (AAV) Encoding FRET Sensor
- Objective: To achieve stable, region-specific, and cell-type-selective expression of a FRET sensor in the mouse brain for in vivo two-photon microscopy or ex vivo slice physiology.
Materials:
- Adult mouse (C57BL/6, 8-12 weeks)
- AAV (serotype, e.g., AAV9 or PHP.eB for broad CNS; AAV2-retro for retrograde labeling; promoter, e.g., hSyn for neurons, GFAP for astrocytes). Titer: >1x10^13 vg/mL.
- Stereotaxic frame with gas anesthesia system
- Microsyringe pump and calibrated glass micropipette or Hamilton syringe (33-gauge)
- Sterile surgical tools
- Ketamine/Xylazine anesthesia
- Analgesic (e.g., Carprofen)
- Brain atlas (Paxinos & Franklin)
Procedure:
- Virus Preparation: Thaw virus aliquot on ice. Centrifuge briefly before loading to ensure no particles are in the cap. Keep on ice and protected from light.
- Animal Preparation: Anesthetize mouse. Secure head in stereotaxic frame. Apply ophthalmic ointment. Shave scalp, clean with alternating betadine and ethanol scrubs (3x each).
- Surgery: Make a midline scalp incision. Identify bregma and lambda. Level the skull.
- Injection: Calculate target coordinates (e.g., Prefrontal Cortex: AP +1.8 mm, ML ±0.3 mm, DV -2.2 mm from bregma). Drill a small craniotomy. Lower the loaded micropipette to the target depth at a slow rate (1 mm/min). Inject 300-500 nL of virus at a rate of 100 nL/min. Wait 10 minutes post-injection to allow diffusion, then slowly retract the pipette.
- Recovery: Suture the incision. Administer analgesics. Monitor animal until fully recovered.
- Expression Wait Period: Allow 3-4 weeks for robust sensor expression before conducting imaging or slice experiments.
4. Signaling Pathway & Experimental Workflow Diagrams
Title: AAV-Mediated FRET Sensor Delivery and Expression Pathway
Title: Decision Workflow for Selecting Sensor Expression Method
5. Research Reagent Solutions Toolkit
Table 2: Essential Materials for FRET Sensor Expression Experiments
| Item | Function/Application | Example Product/Catalog |
|---|---|---|
| FRET Sensor Plasmid | Core genetic construct encoding the donor/acceptor fluorophore-linked sensor protein. | pCAG-iGABASnFR, pAAV-hSyn-jGCaMP8f (Addgene). |
| Endotoxin-Free Plasmid Prep Kit | For high-purity DNA preparation critical for sensitive cells like primary neurons. | ZymoPURE II Plasmid Maxiprep Kit. |
| Lipid-Based Transfection Reagent | Forms complexes with plasmid DNA for delivery into cell membranes. | Lipofectamine 3000, FuGENE HD. |
| Adeno-Associated Virus (AAV) | Safe, efficient viral vector for long-term gene delivery in vivo and in vitro. | AAV9-hSyn1-dLight1.1 (Viral Vector Core facility). |
| Primary Neuron Culture System | Provides physiologically relevant cells for sensor validation. | Gibco Primary Neuron Kit, BrainBits LLC tissue. |
| Stereotaxic Instrument | For precise targeting of viral injections into specific brain regions in rodents. | Kopf Model 940, RWD Life Science systems. |
| Microsyringe Pump | Ensures accurate, slow, and consistent delivery of viral volumes during surgery. | World Precision Instruments UltraMicroPump III. |
| Cre-Driver Mouse Line | Enables cell-type-specific sensor expression when using Cre-dependent (DIO) AAVs. | Jackson Laboratory (e.g., VGAT-IRES-Cre, Sst-IRES-Cre). |
| Genotyping Kit | Essential for identifying transgenic animals carrying the sensor gene. | KAPA Mouse Genotyping HotStart Kit. |
| Live-Cell Imaging Medium | Phenol-red-free medium for maintaining cell health during FRET imaging sessions. | FluoroBrite DMEM, Hibernate-A Low Fluorescence. |
Application Notes
Within the thesis on FRET-based protein sensors for monitoring neurotransmitter release, the selection of an imaging modality is critical. Each technique offers distinct trade-offs in spatial resolution, temporal resolution, photobleaching, phototoxicity, and depth penetration, directly impacting the fidelity of monitoring dynamic release events.
- Widefield Epifluorescence Microscopy: Ideal for high-temporal-resolution imaging of FRET sensor dynamics in cultured neurons or thin brain slices. It provides full-field illumination and detection, enabling fast acquisition of emission ratiometric data. However, it suffers from out-of-focus blur, reducing spatial resolution and quantitative accuracy in thicker samples.
- Laser Scanning Confocal Microscopy: Provides optical sectioning by using a pinhole to reject out-of-focus light. This significantly improves spatial resolution and quantitative accuracy of FRET measurements in fixed samples and live cells. Point-scanning limits speed, while intense laser illumination can accelerate photobleaching of sensors and increase phototoxicity in live tissue.
- Two-Photon (Multiphoton) Microscopy: Excitation occurs only at the focal plane where photon density is highest, providing inherent optical sectioning without a confocal pinhole. Near-infrared (NIR) excitation penetrates deeper into scattering tissue (e.g., brain slices or in vivo) and reduces photobleaching/phototoxicity outside the focal plane. It is the preferred method for imaging FRET sensors in intact tissue or in vivo.
Comparison of Quantitative Performance Parameters
Table 1: Key Characteristics of Microscopy Modalities for Live-Cell FRET Imaging
| Parameter | Widefield Epifluorescence | Laser Scanning Confocal | Two-Photon Excitation |
|---|---|---|---|
| Optical Sectioning | No | Yes (via pinhole) | Yes (inherent) |
| Typical Axial Resolution | ~1-2 µm | ~0.5-1.0 µm | ~0.8-1.5 µm |
| Excitation Wavelength | UV-Visible (e.g., 440 nm) | UV-Visible (e.g., 440, 514 nm) | NIR (e.g., 880 nm) |
| Excitation Volume | Large (entire field) | Diffraction-limited spot | Sub-femtoliter volume |
| Tissue Penetration Depth | < 50 µm (cultured cells) | < 100 µm | > 500 µm |
| Photobleaching | High (whole sample) | High (at focal plane) | Reduced (confined to focal plane) |
| Typical Frame Rate (for 512x512 px) | ~10-100 Hz (camera) | ~0.5-2 Hz (point scanning) | ~0.5-5 Hz (resonant/galvo) |
| Best For (in Neurotransmitter Release) | Fast kinetics in 2D cultures | High-resolution imaging in slices | Deep tissue & in vivo imaging |
Detailed Protocols
Protocol 1: Ratiometric FRET Imaging of Neurotransmitter Release in Cultured Neurons using Widefield Microscopy Objective: To capture the rapid dynamics of neurotransmitter (e.g., glutamate, dopamine) release following electrical or chemical stimulation.
- Cell Preparation: Culture neurons expressing a FRET-based neurotransmitter sensor (e.g., iGluSnFR for glutamate, GRABDA for dopamine) on glass-bottom dishes.
- Setup: Use an inverted widefield microscope with a high-quantum-efficiency sCMOS camera, a 40x or 60x oil-immersion objective (NA ≥1.3), and a fast wavelength switcher (e.g., Lambda DG-4).
- Filter Sets: Configure for donor (CFP/ex: 430-450 nm, em: 460-500 nm) and acceptor (YFP/ex: 490-510 nm, em: 520-550 nm) channels. For ratiometric sensors, use a single excitation (e.g., 440 nm) and dual-emission collection (CFP and YFP channels).
- Acquisition: Set camera to rapid acquisition mode (e.g., 10-50 fps). Acquire a 10s baseline, then deliver a stimulus (e.g., 1s field electrical stimulation or 100µM ATP puff). Record for 60s.
- Analysis: Generate ratio images (YFP/CFP emission) over time. Plot ratio change (ΔR/R0) against time to visualize release kinetics.
Protocol 2: Confocal FRET Imaging in Acute Brain Slices with Acceptor Photobleaching Objective: To quantify FRET efficiency and map sensor expression/activation in a defined optical section.
- Sample Preparation: Prepare acute brain slices (300 µm) from mice expressing a FRET sensor. Maintain in oxygenated aCSF.
- Setup: Use a point-scanning confocal microscope with Argon (458, 514 nm) and 405 nm diode lasers, a 40x water-immersion objective (NA 0.8), and standard PMT detectors.
- Pre-bleach Acquisition: Define a region of interest (ROI). Acquire donor (CFP, ex: 458 nm) and acceptor (YFP, ex: 514 nm) channel images at low laser power (<5%).
- Acceptor Photobleaching: Select an ROI for bleaching. Illuminate with the 514 nm laser at 100% power for 30-100 iterations until YFP signal is reduced by >70%.
- Post-bleach Acquisition: Immediately re-acquire donor and acceptor channel images at pre-bleach settings.
- Analysis: Calculate FRET efficiency E as E = 1 - (CFP_pre / CFP_post), where CFPpre and CFPpost are donor intensities before and after bleaching.
Protocol 3: Two-Photon FRET Imaging of Neurotransmitter Release In Vivo Objective: To monitor neurotransmitter release dynamics in the brain of an awake, behaving animal.
- Animal Preparation: Use a transgenic mouse or rat expressing a FRET-based sensor in a specific brain region. Implant a chronic cranial window and headplate.
- Setup: Use a two-photon microscope with a tunable Ti:Sapphire laser (set to 880 nm for CFP/YFP FRET pairs) and a high-sensitivity, non-descanned detector (GaAsP PMT). Use a 16x or 20x water-immersion objective (NA 0.8-1.0).
- Spectral Separation: Use a dichroic mirror (560 nm) and bandpass filters to separate emission into "short wavelength" (460-500 nm, donor) and "long wavelength" (520-600 nm, acceptor) channels.
- In Vivo Acquisition: Gently head-fix the awake animal. Locate the sensor-expressing region. Acquire line scans or frame scans at 4-10 Hz. Present sensory or behavioral stimuli to evoke neurotransmitter release.
- Analysis: Calculate the emission ratio (Acceptor/Donor) over time. Apply motion correction algorithms. Correlate ratio transients with behavioral event markers.
Visualizations
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for FRET Imaging of Neurotransmitter Release
| Item | Function & Rationale |
|---|---|
| Genetically-Encoded FRET Sensor (e.g., iGluSnFR, GRABDA, dLight) | The core bioreporter. Comprises a neurotransmitter-binding protein coupled to CFP/YFP (or variants). Binding-induced conformational change alters FRET efficiency. |
| Viral Vectors (AAV, Lentivirus) | For efficient delivery and stable expression of the FRET sensor construct in specific neuronal populations in vitro and in vivo. |
| Artificial Cerebrospinal Fluid (aCSF) | Physiological buffer for maintaining live brain slice health during imaging experiments. Must be oxygenated (95% O₂/5% CO₂). |
| Tetrodotoxin (TTX) & 4-AP | Sodium channel blocker (TTX) and potassium channel blocker (4-AP). Used as negative and positive controls, respectively, to silence or elicit action potentials. |
| Synaptic Stimulation Reagents (e.g., High-K⁺ aCSF, ATP, Optogenetic Actuators) | To evoke neurotransmitter release. High-K⁺ depolarizes neurons. ATP activates purinergic receptors. Channelrhodopsin allows precise, optical stimulation. |
| Motion Correction Software (e.g., TurboReg, moco) | Critical for in vivo and slice imaging. Algorithmically stabilizes image stacks to correct for tissue movement artifacts, ensuring accurate ratiometric analysis. |
| Ratiometric Analysis Software (e.g., ImageJ/Fiji, MetaMorph, Python scripts) | To calculate emission ratio time series (YFP/CFP or ΔR/R₀) from acquired image pairs, quantifying sensor activation dynamics. |
Within the broader thesis on FRET-based protein sensors for monitoring neurotransmitter release, this document details the critical application notes and protocols for quantifying release events. The accurate measurement of synaptic vesicle fusion and neurotransmitter concentration dynamics relies on precise calibration of FRET sensors, rigorous rationetric imaging, and standardized analysis pipelines. This is foundational for research in synaptic physiology, neuropharmacology, and the development of neurotherapeutics.
Core Principles: Sensor Calibration & Ratio Imaging
FRET-based neurotransmitter sensors (e.g., iGluSnFR, dLight, GRAB sensors) undergo conformational changes upon ligand binding, altering the efficiency of energy transfer between a donor and acceptor fluorescent protein. Quantification requires converting observed fluorescence into a meaningful biological metric (e.g., neurotransmitter concentration).
Key Equation:
Where R is the emission ratio, R0 is the baseline ratio.
Calibration Protocols
In vitro calibration is essential for determining sensor affinity (Kd), dynamic range (ΔR/R0), and ligand specificity.
Protocol 1: In Vitro Calibration of FRET Sensor Affinity (Kd) Objective: Determine the apparent Kd of the purified sensor protein in a controlled buffer system. Materials: Purified sensor protein, imaging chamber (e.g., glass-bottom dish), microscope with appropriate filters, ligand stock solutions, perfusion system. Procedure:
- Immobilize Sensor: Adsorb or chemically immobilize purified sensor at low density in the chamber.
- Acquire Baseline: Image in ligand-free buffer to obtain FDonor and FAcceptor signals. Calculate R0.
- Titrate Ligand: Perfuse increasing concentrations of ligand (e.g., 0, 0.1x, 0.5x, 1x, 2x, 5x, 10x expected Kd). Allow equilibration at each step.
- Image & Calculate: At each concentration, acquire images and calculate the ratio R.
- Fit Data: Fit the dose-response curve to the Hill equation:
ΔR/R0 = (ΔR_max/R0) * [L]^n / (Kd^n + [L]^n).
Table 1: Example Calibration Data for Common Neurotransmitter Sensors
| Sensor Name | Neurotransmitter | Reported Kd (nM) | Dynamic Range (ΔR/R0) | Reference (Example) |
|---|---|---|---|---|
| iGluSnFR | Glutamate | ~4 μM | ~2.5 | Marvin et al., 2018 |
| dLight1 | Dopamine | ~130 nM | ~3.0 | Patriarchi et al., 2018 |
| GRAB_DA1h | Dopamine | ~90 nM | ~3.4 | Sun et al., 2020 |
| GRAB_ACh3.0 | Acetylcholine | ~2 μM | ~1.8 | Jing et al., 2020 |
| GABA-SnFR | GABA | ~9 μM | ~4.0 | Marvin et al., 2019 |
Protocol 2: In Situ/In Vivo Calibration via Pharmacological Manipulation Objective: Estimate effective Kd in the cellular or tissue environment. Materials: Cell/tissue expressing sensor, imaging setup, agonist (e.g., high K+ buffer), antagonist/transporter blocker, ionomycin (for Ca²⁺ sensors). Procedure:
- Establish Baseline: Record baseline ratio (R0).
- Apply Saturation Dose: Apply a saturating concentration of agonist (e.g., high K+ to depolarize neurons and release endogenous NT) in the presence of a transporter blocker (e.g., TBOA for glutamate) to prevent reuptake. Measure R_max.
- Apply Zero Ligand Condition: Apply a competitive antagonist (e.g., DNQX for glutamate) or enzyme (e.g., glutamate dehydrogenase + NADP⁺) to scavenge ligand. Measure R_min.
- Calculate: Dynamic range = (Rmax - Rmin)/R_min. The half-maximal effective concentration (EC50) in situ can be estimated from responses to graded stimuli.
Rationetric Imaging Workflow
Protocol 3: Live-Cell Rationetric Imaging for Neurotransmitter Release Objective: Record spatially and temporally resolved neurotransmitter release in cultured neurons or brain slices. Workflow Steps:
Diagram Title: Rationetric Imaging and Analysis Pipeline Workflow
Detailed Steps:
- Microscope Configuration: Use a microscope capable of fast, simultaneous dual-emission imaging. A 440 nm laser or LED is ideal for CFP excitation. Emitted light is split using a beamsplitter (e.g., DV2 or Optospilt) onto two camera regions: 480/40 nm (donor, CFP) and 535/30 nm (acceptor, YFP).
- Acquisition Parameters: Use minimal exposure to reduce photobleaching. Frame rates should exceed the expected kinetics of release (typically 10-100 Hz). Ensure precise temporal alignment of donor and acceptor channels.
- Stimulation: Synchronize imaging with stimulation (e.g., a trigger from an electrical stimulator for field stimulation, or a pulse of blue light for optogenetic release).
- Image Processing:
- Background Subtraction: Subtract mean intensity from a cell-free region.
- Bleaching Correction: Fit a mono- or bi-exponential decay to the baseline of the donor and acceptor channels separately and correct.
- Motion Correction: Use cross-correlation based algorithms if needed.
- Ratio Calculation: Compute the pixel-by-pixel or ROI-based ratio
R = F_Acceptor / F_Donor. This corrects for sensor expression heterogeneity and photobleaching. - Conversion to ΔR/R0: Calculate
ΔR/R0 = (R - R0) / R0, where R0 is the average baseline ratio before stimulation.
Analysis Pipelines for Quantifying Release Events
Table 2: Key Metrics for Quantifying Neurotransmitter Release
| Metric | Definition | Biological Interpretation |
|---|---|---|
| Amplitude (ΔR/R0) | Peak change in ratio from baseline. | Relative amount of neurotransmitter released. |
| Rise Time (τ_rise) | Time from 10% to 90% of peak amplitude. | Speed of neurotransmitter accumulation. |
| Decay Time Constant (τ_decay) | Time constant of exponential fit to decay phase. | Clearance rate (uptake, diffusion). |
| Full Width at Half Max (FWHM) | Duration of the event at half peak amplitude. | Temporal profile of release event. |
| Spatial Spread (λ) | Exponential decay constant of ΔR/R0 from release site. | Diffusion/volume transmission range. |
Protocol 4: Event Detection & Kinetic Analysis Objective: Automatically detect release events and extract their kinetics. Tools: Custom scripts (Python/Matlab) or software (Igor Pro, Fiji). Procedure:
- Filtering: Apply a temporal band-pass filter to the ΔR/R0 trace to reduce high-frequency noise and slow baseline drift.
- Detection: Use a threshold-based algorithm (e.g., >5 x SD of baseline noise) or a template-matching algorithm (e.g., deconvolution) to identify event onset times.
- Kinetic Fitting: For each event, fit the rise phase with a single exponential and the decay phase with a single or double exponential function.
- Amplitude & Latency: Extract peak amplitude and latency from stimulus to event onset.
Diagram Title: Computational Pipeline for Release Event Analysis
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for FRET-Based Release Experiments
| Item | Function/Description | Example Product/Catalog # |
|---|---|---|
| FRET Sensor Plasmid | Encodes the neurotransmitter-specific biosensor (e.g., CFP-YFP pair). | Addgene: #XXXXX (e.g., pAAV-hSyn-dLight1.1) |
| Viral Vector (AAV) | For efficient, stable in vivo or in vitro neuronal expression. | AAV9-hSyn-dLight1.1, AAV1-hSyn-iGluSnFR |
| Cell/Tissue Culture Reagents | Maintain healthy neurons for imaging. | Neurobasal-A Medium, B-27 Supplement, GlutaMAX |
| Pharmacological Agents | For calibration and manipulation of release (agonists, antagonists, blockers). | NBQX (AMPA receptor antagonist), Tetrodotoxin (TTX, Na+ channel blocker), Bafilomycin A1 (V-ATPase inhibitor) |
| Imaging Chamber | Provides controlled environment for live cells. | Warner Instruments RC-21BR perfusion chamber |
| Perfusion System | For rapid solution exchange during calibration or stimulation. | ALA Scientific VC-8 valve controller |
| Coverslips & Mounting Media | High-quality #1.5 coverslips for optimal imaging; mounting media for fixed samples. | MatTek dishes; ProLong Glass antifade mountant |
| Image Analysis Software | For processing and quantifying rationetric data. | Fiji/ImageJ with Time Series Analyzer V3, Python (SciPy, NumPy), MATLAB |
| Electrophysiology Setup (Optional) | For precise, direct neuronal stimulation paired with imaging. | Multiclamp 700B amplifier, Digidata 1550B |
| Objective Lens | High numerical aperture for light collection. | 60x oil immersion, NA 1.4 |
| Immersion Oil | Type matched to objective for optimal resolution. | Nikon Type NF, nD=1.515 |
| Optical Filters | Precisely defined for donor/acceptor separation. | Semrock FF01-472/30 (CFP), FF01-542/27 (YFP) |
This document details advanced methodologies for investigating fundamental synaptic processes, framed within a broader thesis on the development and application of FRET-based protein sensors for monitoring neurotransmitter release. The ability to visualize synaptic transmission at the level of individual vesicles is crucial for dissecting the molecular mechanisms of neurotransmission, synaptic plasticity, and the actions of psychoactive compounds. These protocols leverage genetically encoded indicators to provide quantitative, high-resolution data on presynaptic release and glutamate spillover in cultured neuronal networks.
Table 1: Common FRET-Based Neurotransmitter Sensors for Presynaptic Release Studies
| Sensor Name | Neurotransmitter Target | Excitation/Emission (Donor) | Emission (Acceptor) | Dynamic Range (ΔR/R₀ or ΔF/F₀) | Kinetics (τ decay) | Primary Application in Studies |
|---|---|---|---|---|---|---|
| synaptophysin-pHluorin | Vesicle pH (proxy for exocytosis) | 488 nm / 510 nm | - | ~400% ΔF/F₀ | Reacidification: 1-3 s | Total recycling vesicle pool, release probability |
| VGAT-ipHluorin | GABA vesicle pH | 488 nm / 510 nm | - | High | Seconds | GABAergic vesicle fusion and recycling |
| VGluT1-pHluorin | Glutamate vesicle pH | 488 nm / 510 nm | - | ~300% ΔF/F₀ | Seconds | Glutamatergic vesicle fusion, spillover assessment |
| Syn-apt-pHluorin | Targeted to synaptic vesicles | 488 nm / 510 nm | - | High | Seconds | Single synapse, single vesicle resolution |
| iGluSnFR | Extracellular Glutamate | 488 nm / 510 nm | - | ~500% ΔF/F₀ | ~2 ms (rise) | Real-time glutamate transient detection, spillover |
| SF-iGluSnFR (Slow) | Extracellular Glutamate | 488 nm / 510 nm | - | High | ~200 ms (decay) | Integrative measure of spillover and tonic glutamate |
| GluCIBR | Extracellular Glutamate | CFP: 440 nm / 475 nm | YFP: 515 nm | FRET change: ~30% ΔR/R₀ | Sub-second | Ratiometric FRET-based spillover measurement |
Table 2: Typical Experimental Parameters for Single Vesicle Imaging
| Parameter | Typical Value / Range | Notes |
|---|---|---|
| Culture Preparation | DIV 14-21 | Hippocampal or cortical neurons; optimal synapse density. |
| Imaging Temperature | 32-37°C | Maintained with heated stage and chamber. |
| Stimulation | 1-20 APs at 10-100 Hz | Delivered via field or bipolar electrode. |
| Imaging Frame Rate | 10-100 Hz | Higher rates for iGluSnFR; lower for pHluorin. |
| Objective | 60x or 100x oil immersion | High NA (≥1.4) for TIRF or diffraction-limited imaging. |
| Analysis Region (ROI) | ~1 μm² | Centered on individual synaptic boutons. |
Experimental Protocols
Protocol 1: Single Vesicle Release Imaging with synaptophysin-pHluorin in Cultured Hippocampal Neurons
Objective: To visualize and quantify the exocytosis and recycling of individual synaptic vesicles at presynaptic boutons.
Materials: See "The Scientist's Toolkit" below.
Methodology:
- Culture & Transfection: Plate hippocampal neurons from E18 rats or P0 mice at low density (50,000 cells/cm²) on poly-D-lysine-coated glass-bottom dishes. At DIV 5-7, transfect with a synaptophysin-pHluorin (Syn-pH) plasmid using calcium phosphate or lipofectamine-based methods. Image at DIV 14-21.
- Imaging Solution (External): 136 mM NaCl, 2.5 mM KCl, 2 mM CaCl₂, 1.3 mM MgCl₂, 10 mM Glucose, 10 mM HEPES (pH 7.4). Neutral pH (7.4) Bath: Quenches pHluorin fluorescence inside vesicles; exocytosis causes a rapid flash.
- Acidification Solution (Internal): Add 50 mM NH₄Cl to imaging solution for 30-60s to neutralize all vesicles and confirm total recyclable pool size. Alternatively, use a solution with reduced NaCl (substituted with NMDG) and 0.5 mM bafilomycin A1 to block reacidification.
- Microscopy Setup: Use a TIRF or high-sensitivity confocal microscope with a 488 nm laser and EMCCD/sCMOS camera. Maintain temperature at 32-35°C.
- Stimulation & Acquisition: Place a parallel platinum bipolar electrode in the dish. Acquire baseline images (1-2 Hz). Deliver a single or brief train (e.g., 10-20 stimuli at 10-20 Hz) of electrical field stimuli (1 ms pulse, 20-40 mA) synchronized to imaging.
- Data Analysis:
- ROI Selection: Define circular ROIs (~1 μm diameter) over individual, well-isolated fluorescent puncta (boutons).
- Trace Extraction: Calculate ΔF/F₀ for each bouton, where F₀ is the average baseline fluorescence.
- Event Detection: Use a threshold-based algorithm (e.g., ΔF/F₀ > 3-5 SD of baseline noise) to identify single vesicle fusion events. Amplitude and kinetics of each flash are measured.
Protocol 2: Measuring Glutamate Spillover with iGluSnFR in Neuron-Astrocyte Co-cultures
Objective: To detect glutamate diffusion beyond the synaptic cleft (spillover) and its uptake by astrocytes.
Materials: See "The Scientist's Toolkit" below.
Methodology:
- Culture & Transfection: Prepare neuron-astrocyte co-cultures. Transfect neurons with synaptophysin-mCherry to label boutons and astrocytes with cell-surface targeted iGluSnFR (e.g., via GPI anchor or LDL receptor domain) at DIV 7-10. Image at DIV 14-21.
- Pharmacological Isolation: Include 10 μM NBQX and 50 μM D-AP5 in the imaging solution to block postsynaptic AMPA/ NMDA receptors, preventing recurrent activity and isolating presynaptic release and astrocytic sensing.
- Imaging Solution: As in Protocol 1, but often with 1-2 mM Ca²⁺.
- Microscopy: Use a fast widefield or confocal microscope. Acquire iGluSnFR (488 nm ex) and mCherry (560 nm ex) channels simultaneously or alternately at high speed (20-50 Hz).
- Stimulation: Use local microstimulation (via a glass pipette) or minimal field stimulation to activate a small subset of axons. This allows resolution of spillover from single or few synapses.
- Data Analysis:
- Alignment: Align the synaptic marker (mCherry) channel with the iGluSnFR channel.
- Spillover Quantification: Draw ROIs around astrocytic membranes adjacent to, but not directly apposed to, stimulated synapses.
- Kinetic Analysis: Measure the amplitude, rise time, and decay time of iGluSnFR transients in the astrocytic ROIs. Compare these to transients measured at the synaptic cleft (if detectable). Slower, smaller amplitude transients in perisynaptic areas indicate spillover and diffusion.
Signaling Pathways and Workflow Diagrams
Diagram Title: Signaling Pathway: Vesicle Fusion & Glutamate Spillover
Diagram Title: Experimental Workflow for Single Vesicle Imaging
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for FRET-Based Release Studies
| Item | Function/Benefit | Example Product/Catalog Number (Typical) |
|---|---|---|
| Primary Neuronal Culture Kit | Provides optimized media, supplements, and sometimes substrates for reproducible rodent neuron culture. | Gibco Primary Neuron Kit, BrainBits LLC kits. |
| Poly-D-Lysine (PDL) Solution | Coating substrate for glass-bottom dishes to promote neuronal adhesion and growth. | Millipore Sigma A-003-E (1 mg/mL). |
| Genetically Encoded Sensor Plasmids | Core tools for imaging. Syn-pH, iGluSnFR variants, VGAT-pHluorin, etc. | Addgene (e.g., #37087 for synaptophysin-pHluorin, #41732 for iGluSnFR-A184V). |
| Neuronal Transfection Reagent | Efficient, low-toxicity transfection of post-mitotic neurons. | Lipofectamine 2000, CalPhos Mammalian Transfection Kit. |
| Fast Glutamate Receptor Antagonists | Pharmacologically isolate presynaptic release by blocking post-synaptic receptors. | NBQX (AMPAR antagonist), D-AP5 (NMDAR antagonist) from Tocris. |
| Vesicular Reacidification Inhibitor | Blocks the V-ATPase to prevent re-quenching of pHluorin, allowing cumulative release measurement. | Bafilomycin A1 (Tocris #1334). |
| Electrical Stimulation Controller & Electrodes | For precise, repeatable delivery of action potentials in culture. | Warner Instruments RC-37FS chamber with platinum-iridium electrodes, connected to a stimulus isolator (e.g., A.M.P.I. Iso-Flex). |
| Live-Cell Imaging Buffers (Hibernate-based) | Maintain neuronal health and synaptic function during extended imaging sessions. | BrainBits Hibernate-A Low Fluorescence buffer. |
| Ca²⁺ Channel Agonists/Antagonists | To manipulate release probability (e.g., increase with Bay K8644, block with Cd²⁺). | Tocris Bioscience. |
| Analysis Software | For automated detection and quantification of fluorescence transients and vesicle events. | ImageJ/FIJI with Time Series Analyzer V3, MATLAB with custom scripts, or commercial packages like MetaMorph. |
This application note details protocols for monitoring neurotransmitter release in awake, behaving animals using Förster Resonance Energy Transfer (FRET)-based genetically encoded sensors. This work is framed within a broader thesis asserting that FRET-based protein sensors represent a transformative technology for neuroscience and neuropharmacology, enabling direct, real-time, and cell-type-specific measurement of neurotransmission with high spatiotemporal resolution in vivo. These methods move beyond traditional microdialysis and voltammetry, allowing monitoring of specific neurotransmitters like glutamate, dopamine, GABA, and acetylcholine during complex behaviors.
Table 1: Comparison of Representative FRET-Based Neurotransmitter Sensors
| Sensor Name (Acronym) | Neurotransmitter Target | Dynamic Range (ΔR/R0 %) | Affinity (Kd / EC50) | Reference(s) & Year |
|---|---|---|---|---|
| iGluSnFR (various) | Glutamate | ~200% | ~5 µM | Marvin et al., 2018; 2023 |
| GRABDA (h,m) | Dopamine | ~90% | 130 nM (h); 10 µM (m) | Sun et al., 2018; 2020 |
| GRABACh | Acetylcholine | ~70% | 2 µM | Jing et al., 2018 |
| iGABASnFR | GABA | ~400% | 11 µM | Marvin et al., 2019 |
| GRAB5-HT | Serotonin | ~70% | 7.8 nM (red) | Wan et al., 2021 |
| dLight1 (various) | Dopamine | ~340% | 330 nM (dLight1.1) | Patriarchi et al., 2018 |
Table 2: In Vivo Imaging Modalities for Sensor Readout
| Imaging Modality | Spatial Resolution | Temporal Resolution (Frame Rate) | Penetration Depth | Key Application in Behaving Animals |
|---|---|---|---|---|
| One-Photon Epifluorescence (Miniscope) | ~10-50 µm | 10-30 Hz | Surface (with GRIN lens) | Deep brain structures in freely moving mice |
| Two-Photon Microscopy (TPrM) | ~0.5-1 µm (lateral) | 1-10 Hz (region); 30+ Hz (line scan) | ~500-700 µm | Cortical/subcortical layers in head-fixed mice |
| Fiber Photometry | Bulk signal (region) | 10s-100s Hz | Any (via optical fiber) | Bulk neurotransmitter dynamics in freely moving |
Experimental Protocols
Protocol 1: Viral-Mediated Sensor Expression & Cranial Window Implantation for TPrM
Objective: To express a FRET-based sensor in a specific brain region and prepare a chronic optical window for high-resolution imaging in head-fixed, awake mice.
Materials: See "The Scientist's Toolkit" below.
Procedure:
- Virus Preparation: Select an AAV (e.g., AAV9 or AAV-PHP.eB) carrying the sensor (e.g., GRABDA2m) under a cell-type-specific promoter (e.g., hSyn for neurons, GFAP for astrocytes). Titer: ≥ 1x10¹³ GC/mL.
- Stereotaxic Injection:
- Anesthetize mouse (1.5% isoflurane) and secure in stereotaxic frame.
- Perform aseptic surgery, expose skull, and level bregma and lambda.
- Calculate target coordinates (e.g., Striatum: AP +1.0 mm, ML ±1.8 mm, DV -3.5 mm from bregma).
- Drill a small craniotomy (~1 mm diameter).
- Load virus solution into a glass micropipette (Hamilton syringe) and lower to target DV coordinate.
- Inject 300-500 nL of virus at a rate of 100 nL/min.
- Wait 10 min post-injection before slowly retracting the pipette.
- Chronic Cranial Window Implantation (for cortex):
- Following virus injection (or 3 weeks later for expression), perform a separate aseptic surgery.
- Create a ~3-5 mm diameter craniotomy over the region of interest.
- Carefully remove the dura mater.
- Place a sterile, circular cover glass (3-5 mm diameter) onto the brain surface.
- Secure the cover glass using dental cement, building a well around it.
- Allow the mouse to recover for ≥1 week before imaging.
- Sensor Expression: Wait 3-6 weeks for robust sensor expression.
Protocol 2: Fiber Photometry Recording in Freely Behaving Mice
Objective: To record bulk neurotransmitter dynamics during behavioral tasks in freely moving animals.
Procedure:
- Virus Injection & Optical Fiber Implantation (Single Surgery):
- Follow Steps 1-3 of Protocol 1 for virus injection into the target region.
- Instead of a cover glass, lower a ceramic or metal ferrule-containing optical fiber (200-400 µm core diameter, 0.37-0.5 NA) to 100-150 µm above the virus injection site.
- Secure the ferrule to the skull using super glue and dental cement.
- Allow recovery and expression for 3-6 weeks.
- Photometry System Setup:
- Use a dual-wavelength system. For a non-FRET sensor like GRABDA (GFP-based), use 470 nm LED for excitation and a 415 nm LED for isosbestic control.
- Connect a dichroic mirror/cube to split emitted light to a sensitive photodetector (e.g., femtowatt photoreceiver).
- Synchronize light pulses and data acquisition with a behavior-tracking system (e.g., EthoVision).
- Data Acquisition & Behavioral Synchronization:
- Tether the mouse's fiber to the patch cord.
- Record a 5-10 minute baseline session in the home cage.
- Proceed with the behavioral paradigm (e.g., open field, social interaction, operant conditioning). Record behavior via camera.
- Acquire fluorescence signals (F470 and F415) at 100-1000 Hz.
- Data Analysis:
- Calculate ΔF/F = (F470 - F0)/F0, where F0 is the baseline fluorescence median.
- Normalize control channel (F415) similarly.
- Correct for motion artifacts by subtracting the fitted isosbestic signal (F415) from the ΔF/F470 trace.
- Align processed fluorescence traces with behavioral timestamps (e.g., lever press, social approach).
Protocol 3: Two-Photon FRET Imaging in Awake, Head-Fixed Mice
Objective: To image cell-specific neurotransmitter dynamics with cellular resolution.
Procedure:
- Animal Preparation: Use a mouse prepared with a cranial window (Protocol 1) and expressing a FRET-based sensor (e.g., early generation glutamate sensors).
- Head-Fixation Habituation: Over 3-5 days, gradually acclimate the mouse to head-fixation under the microscope and to a treadmill or behavioral apparatus.
- Two-Photon Microscope Setup:
- Use a tunable Ti:Sapphire laser set to the appropriate excitation wavelength (e.g., 920 nm for GFP/YFP FRET pairs).
- Use a high-sensitivity, GaAsP photomultiplier tube (PMT) detector.
- Configure two emission channels: Channel 1 (donor, e.g., CFP: 465-500 nm) and Channel 2 (acceptor, e.g., YFP: 520-550 nm).
- Use a 20x water-immersion objective (1.0 NA).
- Image Acquisition:
- Anesthetize the mouse lightly (0.5% isoflurane) for positioning under the objective, then cease anesthesia.
- Apply ultrasound gel or water for immersion.
- Locate the sensor-expressing region via epifluorescence.
- Acquire time-series images (512x512 pixels) at 2-4 Hz. For faster kinetics, use line scans.
- FRET Ratio Analysis:
- Perform motion correction using image registration algorithms.
- Define regions of interest (ROIs) around cell bodies or neuropil.
- Calculate the background-subtracted fluorescence intensity for donor (IDD) and acceptor (IDA) channels.
- Compute the FRET ratio (R) = IDA / IDD.
- Calculate ΔR/R0 = (R - R0)/R0, where R0 is the baseline ratio.
- Correlate ΔR/R0 traces with behavior (e.g., whisker stimulation, running onset).
Visualization: Diagrams
Workflow for In Vivo Neurotransmitter Imaging
FRET Sensor Mechanism Upon NT Binding
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for In Vivo Neurotransmitter Imaging
| Item / Reagent | Function / Description | Example Product / Note |
|---|---|---|
| Genetically Encoded FRET Sensor | Core molecular tool that changes fluorescence upon NT binding. | GRABDA2m (AAV-hSyn-GRABDA2m) for dopamine. |
| Recombinant Adeno-Associated Virus (AAV) | Safe, efficient vector for delivering sensor genes to specific brain cells. | AAV9, AAV-PHP.eB for systemic; AAV5 for direct brain injection. |
| Cell-Type-Specific Promoter | Drives sensor expression in targeted cell populations (neurons, astrocytes). | hSyn (neurons), GFAP (astrocytes), CaMKIIα (excitatory neurons). |
| Chronic Cranial Window | Provides long-term optical access to the brain for microscopy. | 3-5 mm cover glass, cemented with dental acrylic. |
| Gradient-Index (GRIN) Lens | Miniature lens implanted for deep brain imaging via miniscopes. | 0.5-1 mm diameter, used with one-photon miniscopes. |
| Implantable Optical Fiber | For delivering light and collecting fluorescence in fiber photometry. | 200-400 µm core, low-autofluorescence, ceramic ferrule. |
| Two-Photon Ti:Sapphire Laser | Provides near-infrared pulsed light for deep, high-resolution imaging. | Coherent Chameleon Vision or Spectra-Physics Mai Tai HP. |
| One-Photon Miniscope | Miniature microscope for imaging in freely moving animals. | UCLA Miniscope v4, or commercial Inscopix nVoke. |
| Fiber Photometry System | System for recording bulk fluorescence dynamics via an optical fiber. | Tucker-Davis Technologies RZ, Doric LED/Diode systems. |
| Data Analysis Software | For processing fluorescence videos and extracting traces. | Suite2p, Minian (miniscope); Custom Python/Matlab scripts. |
Application Notes
This protocol details the application of genetically encoded FRET-based biosensors for high-throughput screening (HTS) in drug discovery and toxicity testing, specifically within the context of a thesis focused on monitoring neurotransmitter release. These sensors translate biochemical events, such as neurotransmitter receptor activation or calcium influx following vesicle fusion, into quantifiable changes in fluorescence emission ratio, enabling real-time, live-cell analysis.
Core Advantages for HTS:
- Live-Cell Kinetics: Enables monitoring of dynamic cellular responses over time, superior to single-endpoint assays.
- High Temporal and Spatial Resolution: Captures rapid signaling events localized to subcellular compartments (e.g., synaptic terminals).
- Rationetric Measurement: The use of emission ratios minimizes artifacts from variable sensor expression, cell thickness, or excitation intensity, increasing data robustness for automated platforms.
- Functional Readouts: Measures direct physiological consequences (e.g., cAMP production, kinase activity, ion flux) rather than mere binding.
Key Application Areas:
- Drug Discovery (Primary Screening): Identification of novel agonists/antagonists for G-protein-coupled receptors (GPCRs) or modulators of intracellular signaling pathways implicated in neurological disorders.
- Toxicity Testing (Secondary Screening/Profiling): Assessment of compound-induced cellular stress, such as disruptions in calcium homeostasis, mitochondrial membrane potential, or apoptotic pathway activation (e.g., using caspase-3 FRET sensors).
- Mechanistic Deconvolution: Following a primary hit from a binding assay, FRET-based pathway sensors can classify compounds by their functional signaling bias (e.g., biased agonism at GPCRs).
Quantitative Performance Metrics for FRET-Based HTS:
Table 1: Key Performance Indicators for FRET HTS Campaigns
| Metric | Target/ Typical Range | Explanation |
|---|---|---|
| Z'-Factor | > 0.5 | Statistical parameter for assay quality; >0.5 indicates excellent separation between positive and negative controls. |
| Signal-to-Noise Ratio (S/N) | > 10 | Ratio of assay window to background variability. |
| Signal-to-Background Ratio (S/B) | > 2 | Fold-change between positive and negative control signals. |
| Coefficient of Variation (CV) | < 10% | Measure of well-to-well reproducibility. |
| Assay Window (ΔR) | 10-25% ΔR/R | Typical change in emission ratio (R) for a robust biosensor response. |
Table 2: Example FRET Biosensors for Neurotransmitter & Toxicity Pathways
| Sensor Name/Type | Biological Target/Process | FRET Pair | Primary HTS Application |
|---|---|---|---|
| Epac-based | cAMP Dynamics (GPCR: Gs/i/q) | CFP/YFP | Drug discovery for GPCR modulators. |
| Cameleon (YC3.60) | Cytosolic Ca²⁺ (Neurotransmitter Release) | CFP/cpVenus | Toxicity (Ca²⁺ dysregulation); Presynaptic modulator screening. |
| AKAR | PKA Kinase Activity | CFP/YFP | Downstream signaling of monoamine receptors. |
| SNIFITs / iGluSnFR | Specific Neurotransmitter (e.g., Glutamate) | CFP/YFP | Direct detection of synaptic release; Uptake inhibitor screening. |
| SCAT3 | Caspase-3 Activity (Apoptosis) | CFP/YFP | Cytotoxicity & apoptotic pathway testing. |
Experimental Protocols
Protocol 1: HTS for GPCR Modulators Using a cAMP FRET Sensor (Epac-based) Objective: To screen a compound library for modulators of a GPCR that signals via adenylate cyclase, using a stable cell line expressing the receptor and the Epac-cAMP FRET sensor.
Materials (Research Reagent Solutions Toolkit):
- Cell Line: HEK293T or CHO-K1 cells stably expressing the target GPCR and the Epac-cAMP FRET biosensor (e.g., pGLO-20F).
- Culture Medium: DMEM/F-12, 10% FBS, appropriate selection antibiotics (e.g., Hygromycin, G418).
- Assay Buffer: Hanks' Balanced Salt Solution (HBSS) with 20 mM HEPES, pH 7.4.
- Controls: Forskolin (10 µM, cAMP inducer/positive control), Vehicle (0.1% DMSO, negative control), Reference Agonist/Antagonist.
- Compound Library: Dissolved in DMSO, pre-dispensed in 384-well assay plates.
- Instrumentation: Automated liquid handler, fluorescence plate reader equipped with dual-emission capability for CFP excitation (∼430 nm) and YFP/CFP emission (∼535 nm/∼485 nm).
Methodology:
- Cell Preparation: Harvest stable cells and seed at 20,000 cells/well in black-walled, clear-bottom 384-well plates in 40 µL growth medium. Culture for 24-48 hrs to reach 80-90% confluence.
- Compound Transfer: Using an acoustic or pintool dispenser, transfer 100 nL of library compounds (typically at 10 mM in DMSO) to assay plates for a final test concentration of 10 µM (1% DMSO final). Include control wells.
- Equilibration & Reading: Remove growth medium, wash once gently with 50 µL Assay Buffer, and add 40 µL Assay Buffer. Equilibrate plates in the plate reader at 37°C for 10 min.
- Baseline Measurement: Read the baseline fluorescence intensity at 485 nm (CFP) and 535 nm (YFP) for 5 minutes (1 reading/minute). Calculate the baseline FRET ratio (R_baseline = YFP/CFP).
- Agonist Mode (Primary Screen): Add 10 µL of 5X Assay Buffer containing a low, sub-saturating concentration of known agonist (EC20) using the reader's injector. Monitor the FRET ratio for 15 minutes.
- Antagonist Mode (Secondary Screen): For hits from Step 5, pre-incubate cells with the compound for 15 min before challenging with a maximal agonist concentration (EC80). An increased ratio inhibition indicates antagonism.
- Data Analysis: Normalize data as ΔR/R = (R - Rbaseline) / Rbaseline. Calculate Z'-factor using forskolin (max) and vehicle (min) controls. Hits are defined as compounds producing a ΔR/R > 3 standard deviations from the mean of the vehicle control.
Protocol 2: Toxicity Screening Using a Caspase-3 FRET Sensor (SCAT3) Objective: To profile hits from primary screens for induction of apoptosis in a neuronal cell model.
Materials:
- Cell Line: SH-SY5Y or iPSC-derived neurons transduced with SCAT3 (CFP-DEVD-YFP).
- Culture Medium: Neuronal maintenance medium.
- Assay Buffer: As in Protocol 1.
- Controls: Staurosporine (1 µM, apoptosis inducer), Vehicle (0.1% DMSO), Z-VAD-FMK (pan-caspase inhibitor, for inhibition control).
- Instrumentation: As in Protocol 1.
Methodology:
- Cell Preparation: Seed cells in 384-well plates as in Protocol 1, Step 1.
- Compound Treatment: Add compounds (from primary hit list) and controls using an automated dispenser. Include a set of wells pre-treated with 20 µM Z-VAD-FMK for 1 hour to confirm caspase-dependent signal.
- Kinetic Measurement: Immediately place plates in the reader at 37°C. Acquire CFP and YFP emission every 15 minutes for 6-24 hours.
- Data Analysis: Calculate the FRET ratio (YFP/CFP). Note: Upon caspase-3 cleavage, FRET is lost, CFP emission increases, and YFP emission decreases, causing the ratio to drop. Plot ratio over time. A significant decrease relative to vehicle control indicates apoptotic toxicity. The IC50 for cytotoxicity can be determined via dose-response.
Visualization
Diagram 1: FRET Biosensor Principle & HTS Workflow
Diagram 2: Key Pathways in Neurotransmitter Release & Toxicity Screening
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Research Reagent Solutions for FRET-Based HTS
| Item | Function/Description | Example/Note |
|---|---|---|
| Genetically Encoded FRET Biosensors | Core molecular tool; translates biological activity into optical signal. | Epac-cAMP (cAMP), Cameleon (Ca²⁺), AKAR (PKA), iGluSnFR (Glutamate). |
| Stable Cell Lines | Ensures consistent, homogeneous sensor expression essential for HTS reproducibility. | HEK293, CHO, or neuronal lines with integrated sensor gene(s). |
| HTS-Optimized Cell Culture Medium | Supports cell health and consistent assay performance over screening duration. | Phenol-red-free medium, with stable glutamine and low background fluorescence. |
| 384/1536-Well Microplates | Standard format for HTS; black walls minimize cross-talk, clear bottoms for imaging. | Black-walled, clear-bottom, tissue-culture treated plates. |
| Automated Liquid Handling System | For precise, high-speed dispensing of cells, compounds, and reagents. | Acoustic dispensers for non-contact compound transfer. |
| Multimode Plate Reader with Kinetic Capability | Measures dual-emission fluorescence from each well over time. | Equipped with 430nm excitation, 485nm & 535nm emission filters, environmental control. |
| Reference Agonists/Antagonists | Critical for assay validation, determination of Z'-factor, and data normalization. | Well-characterized high-potency ligands for the target pathway. |
| Apoptosis Inducer/Inhibitor Controls | Essential for validating toxicity assays and confirming mechanism. | Staurosporine (inducer) and Z-VAD-FMK (caspase inhibitor). |
Optimizing Signal and Specificity: Troubleshooting Common Pitfalls in FRET Sensing
Within the development and application of FRET-based protein sensors for monitoring real-time neurotransmitter release, photobleaching presents a critical barrier. It diminishes fluorescence signal, truncates experimental observation windows, and compromises quantitative accuracy. This document provides application notes and protocols to mitigate photobleaching through optimized imaging methodologies and the implementation of advanced photostable fluorophores, specifically contextualized for live-cell imaging of synaptic transmission.
Photostable Fluorophores for FRET Sensors
The selection of fluorophores for donor-acceptor pairs in FRET sensors is paramount. Key properties include high quantum yield, appropriate spectral overlap, and critically, enhanced photostability.
Table 1: Comparative Analysis of Photostable Fluorophores for FRET-based Neurotransmitter Sensors
| Fluorophore Pair (Donor/Acceptor) | Molar Extinction Coefficient (M⁻¹cm⁻¹) | Quantum Yield | Photostability (Half-life under Illumination)* | Recommended for Live-Cell Duration |
|---|---|---|---|---|
| mNeonGreen / mScarlet | 116,000 / 100,000 | 0.80 / 0.70 | High (~300s) | Medium-term (minutes) |
| CyPet / YPet | 35,000 / 104,000 | 0.51 / 0.77 | Medium (~150s) | Short-term |
| SNAP-tag / HaloTag (with Janelia Fluor Dyes) | Varies by dye | Varies | Very High (>600s) | Long-term (hours) |
| LSSmOrange / LSSmCyanine | 52,000 / 32,000 | 0.45 / 0.30 | High (~280s) | Medium-term |
| sREACh / QYFP (as acceptor pair) | 50,000 / 124,000 | 0.03 / 0.85 | Exceptionally High | Long-term (FRET acceptor) |
*Approximate half-life under standard 488nm or 561nm illumination at ~1 kW/cm² in live cells. sREACh is a dark acceptor that minimizes direct photobleaching.
Optimized Imaging Protocols
Protocol 3.1: General Live-Cell Imaging Setup to Minimize Photobleaching
Objective: To configure a spinning disk or point-scanning confocal microscope for prolonged, stable imaging of FRET sensor expression in neuronal cultures. Materials:
- Microscope with environmental chamber (37°C, 5% CO₂)
- High-efficiency, low-phototoxicity laser lines (e.g., 488nm, 561nm)
- High Quantum Efficiency (QE) camera (>80% QE)
- Immersion oil with matched refractive index
- Neuronal culture expressing FRET-based neurotransmitter sensor (e.g., GABA, Glutamate, DA sensor)
- Imaging medium (e.g., Hibernate-E, low fluorescence)
Procedure:
- Environmental Control: Allow the chamber to stabilize for ≥45 minutes before imaging to ensure temperature and pH stability.
- Illumination Power: Use the minimum laser power necessary to achieve a signal-to-noise ratio (SNR) >10. Start at 0.5-2% of laser output for spinning disk systems.
- Exposure Time: Optimize camera exposure time (typically 50-200ms) to balance SNR and temporal resolution.
- Acquisition Frequency: For neurotransmitter release events, a rate of 2-10 Hz may be required. For baseline monitoring, reduce to 0.1-0.5 Hz to limit light dose.
- Focus Stabilization: Engage hardware-based autofocus systems (e.g., laser-based) to avoid repeated focus-search illumination.
- Neutral Density Filters: If lasers cannot be precisely attenuated below 0.5%, use ND filters to reduce intensity.
- Data Acquisition: Acquire donor channel (e.g., 480/40 nm excitation, 535/30 nm emission) and FRET/acceptor channel (e.g., 480/40 nm excitation, 605/50 nm emission) sequentially to minimize bleed-through.
Protocol 3.2: Calibration and Rationetric FRET Measurement
Objective: To accurately calculate FRET ratio changes while correcting for photobleaching drift and direct acceptor excitation. Procedure:
- Acquire Control Cell Images: Image cells expressing donor-only and acceptor-only constructs under identical settings to determine spectral bleed-through coefficients.
- Calculate Corrected FRET (Fc):
Fc = FRET_channel - (a * Donor_channel) - (b * Acceptor_channel)whereaandbare bleed-through coefficients. - Generate Rationetric Output: Calculate the dynamic ratio
R = Fc / Donor_channel. - Photobleaching Correction: Fit a mono-exponential decay curve to the baseline
Rvalues from a non-stimulated region and use this model to correct the entire dataset.
Research Reagent Solutions Toolkit
Table 2: Essential Materials for Photostable FRET Imaging
| Item | Function / Benefit |
|---|---|
| Janelia Fluor 549 HaloTag Ligand | Bright, photostable dye for site-specific labeling of HaloTag-fused sensor components. |
| SNAP-cell 647-SiR | Cell-permeable, far-red dye for SNAP-tag labeling; minimizes cellular autofluorescence. |
| OxEA Imaging Medium | Oxygen-depleted, antioxidant-enriched medium to significantly reduce photobleaching and phototoxicity. |
| Prolong Diamond Antifade Mountant (for fixed samples) | High-performance mounting medium that preserves fluorescence in fixed preparations. |
| SCAA (Superior Cytocompatible Antioxidant Additive) | Additive to standard imaging media to scavenge ROS generated during illumination. |
| CellLight Reagents (BacMam system) | For mild, efficient delivery of FRET sensor constructs to difficult-to-transfect primary neurons. |
| Glass-bottom dishes (No. 1.5, 170±5 µm thickness) | Optimal for high-resolution oil-immersion objectives; ensures minimal spherical aberration. |
Diagram: FRET Sensor Photobleaching Mitigation Workflow
Diagram Title: Workflow for Photobleaching Mitigation in FRET Imaging
Diagram: Key Pathways in Photobleaching & Protection
Diagram Title: Pathways of Photobleaching and Protective Strategies
Application Notes
Within the broader thesis on developing FRET-based protein sensors for monitoring neurotransmitter release, a critical challenge is distinguishing authentic exocytotic events from concurrent extracellular pH shifts. Synaptic vesicle release is accompanied by the efflux of protons, leading to transient local acidification. Many fluorescent protein chromophores are sensitive to their protonation state, causing intensity or FRET ratio changes independent of the intended sensor conformational change.
Key strategies to mitigate pH artifacts include:
- Rationetric & FRET-Based Measurements: Using pH-insensitive FRET pairs or reference fluorophores (e.g., mTurquoise2 with a pH-stabilized mutation, SCFP3A) normalizes for intensity changes but not for direct pH effects on the chromophore's spectral properties.
- Genetic Engineering of Sensor Scaffolds: Introducing point mutations near the chromophore (e.g., superfolder GFP mutations, "pH-stable" variants like pHluorin2) can significantly raise the pKa, reducing sensitivity in the physiological range.
- Parallel Control Experiments: Essential protocols involve co-expression or sequential imaging of a pH-only control sensor (e.g., a non-activity-responsive FRET construct with identical fluorophores) to quantify and subtract the pH contribution.
- Pharmacological & Environmental Controls: Applying drugs that block vesicular proton pumps (e.g., Bafilomycin A1) or using HEPES-buffered saline to minimize CO2/HCO3- buffering can help isolate pH effects.
Quantitative Data Summary
Table 1: Common FRET Donor/Acceptor Pairs and Their pH Sensitivity Profiles
| Fluorophore Pair | Donor pKa | Acceptor pKa | Recommended for pH-Variable Milieu? | Notes |
|---|---|---|---|---|
| CFP (ECFP)/YFP (Venus) | ~4.7 | ~6.0 (Venus YFP) | No | Venus YFP is highly pH-sensitive; signal loss upon acidification. |
| mTurquoise2/mNeonGreen | ~4.5 (mTq2) | ~5.7 (mNG) | Moderate | mTq2 is exceptionally pH-resistant; acceptor sensitivity remains. |
| mCerulean3/cpVenus-L194P | ~3.8 (mCe3) | ~6.5 (cpVenus) | Moderate | Low donor pKa beneficial; acceptor "L194P" mutation raises pKa. |
| T-Sapphire/sREACh | ~5.7 (T-Sapphire) | pH-insensitive | Yes | Optimal. T-Sapphire excitation rationetric; sREACh is a dark acceptor. |
| CFP/REACh | ~4.7 | pH-insensitive | Yes | REACh (Dark Acceptor) eliminates acceptor pH artifacts. |
Table 2: Impact of Extracellular pH Shifts on Common Neurotransmitter Release Assays
| Assay Method | Primary Readout | Vulnerable to pH Artifact? | Typical Artifact Manifestation |
|---|---|---|---|
| Synaptophluorin (pHluorin) Imaging | Fluorescence Intensity (dequenching) | High | Direct measure of pH change; requires controls to confirm vesicular origin. |
| FRET-based iGluSnFR | FRET Ratio Change | Medium | Protonation can alter chromophore absorption/emission, affecting ratio. |
| snif-based iGABASnFR | Fluorescence Intensity | Medium | Engineered for reduced pH sensitivity, but not fully immune. |
| VGLUT-pHluorin | Fluorescence Intensity | High | Specifically reports vesicular pH; ideal control for pH changes. |
| False Transmitter (FM Dyes) | Fluorescence Intensity | Low | Dye environment is lipophilic; minimal sensitivity to aqueous pH shifts. |
Experimental Protocols
Protocol 1: Validating pH Insensitivity of a FRET Sensor In Vitro Objective: To determine the direct effect of pH on the FRET ratio of a purified or expressed protein sensor. Materials: Purified sensor protein or transfected cells, microplate reader/spectrofluorometer, buffers at defined pH (pH 6.0, 6.5, 7.0, 7.4, 8.0) in high-buffering capacity (e.g., 100 mM phosphate or HEPES), positive control ligand. Procedure:
- Dilute purified sensor in each pH buffer to identical concentration.
- In a 96-well plate, load 100 µL per sample in triplicate.
- Measure donor emission (e.g., 475 nm for CFP) with donor excitation (e.g., 433 nm) and acceptor emission (e.g., 527 nm for YFP) with donor excitation.
- Calculate the FRET Ratio (Acceptor Emission / Donor Emission) for each well.
- Repeat measurements after adding a saturating concentration of target ligand to confirm the dynamic range persists across pH values.
- Analysis: Plot FRET Ratio vs. pH. A flat baseline indicates pH insensitivity. A >5% change over the physiological range (pH 6.8-7.6) warrants corrective strategies.
Protocol 2: Differentiating Release from pH Shift in Live-Cell Imaging Objective: To record neurotransmitter release events while concurrently monitoring extracellular pH. Materials: Cell culture (e.g., neurons), cDNA for: 1) FRET-based neurotransmitter sensor (e.g., iGluSnFR), 2) pH-only control construct (e.g., membrane-tagged, non-FRETing CFP-YFP), transfection reagent, imaging setup with dual-emission ratio capability, stimulation solution (e.g., high K+ or field stimulation), HEPES-buffered imaging saline. Procedure:
- Co-transfect cells with the primary sensor and the pH-only control sensor. Alternatively, perform sequential experiments.
- Mount culture on microscope. Perfuse with HEPES-buffered saline to stabilize extracellular pH.
- Define two regions of interest (ROIs): one expressing both sensors, one expressing only the pH-control.
- Acquire time-lapse ratio images (Donor and Acceptor channels) at 2-10 Hz.
- Apply stimulus (e.g., 50 mM KCl, 2 sec) to evoke release.
- Analysis: Extract ratio (R=FA/FD) vs. time traces for both ROIs. Calculate ΔR/R0. The signal from the primary sensor is ΔRtotal. The signal from the pH-control is ΔRpH. The pH-corrected release signal is: ΔRcorrected = ΔRtotal - (ΔR_pH * SC), where SC is a scaling factor for expression level differences.
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions
| Item | Function & Rationale |
|---|---|
| HEPES-buffered Extracellular Solution | Replaces CO2/HCO3- buffer; minimizes pH fluctuations from metabolic activity or proton release during imaging. |
| Bafilomycin A1 (100 nM) | Selective V-ATPase inhibitor. Blocks vesicle re-acidification and proton efflux during exocytosis, used to isolate pH component of a signal. |
| pH-Insensitive Reference Fluorophore (e.g., mTurquoise2) | A donor with pKa << 7.0, used to create ratiometric or FRET sensors resistant to acidification artifacts. |
| Dark Acceptor (e.g., sREACh) | A non-fluorescent acceptor for FRET. Eliminates direct pH sensitivity of the acceptor emission channel. |
| Cell-Impermeant pH Dye (e.g., SNARF-5F) | Directly and quantitatively measures extracellular pH shifts in the synaptic cleft for independent calibration. |
| "pH-stable" GFP Variant (e.g., superfolder GFP, pHluorin2) | Engineered fluorophores with elevated pKa (>7.5), reducing protonation during physiological acid transients. |
Visualizations
Diagram 1: Signal vs Artifact Pathways (Max 760px)
Diagram 2: pH Correction Experiment Flow (Max 760px)
Diagram 3: Sensor Engineering Strategies (Max 760px)
Application Notes
The utility of any FRET-based neurotransmitter sensor is critically determined by its kinetic parameters relative to the endogenous signaling event it is designed to detect. Neurotransmitter transients in the synaptic cleft occur on sub-millisecond to millisecond timescales, with clearance rates governed by diffusion, reuptake, and enzymatic degradation. A sensor must have a kon rate sufficiently fast to capture the rising phase of the transient and a koff rate that balances temporal resolution with signal integration. Sensors with koff rates slower than the clearance rate will temporally blur the signal, while excessively fast koff rates may compromise signal-to-noise ratio.
For studying vesicular release events (e.g., glutamate, GABA), sensors require kon rates > 10⁷ M⁻¹s⁻¹ and koff rates between 50-500 s⁻¹ to accurately report peak concentration and decay kinetics. For slower neuromodulator signals (e.g., dopamine, norepinephrine), sensors with koff rates of 1-20 s⁻¹ may be appropriate. Mismatched kinetics can lead to misinterpretation of release probability, spillover, and receptor occupancy.
Key Quantitative Parameters of Neurotransmitter Transients and Sensor Requirements
| Parameter | Typical Range for Fast Transmitters (e.g., Glutamate) | Ideal Sensor Kinetics for Resolution | Experimental Method for Characterization |
|---|---|---|---|
| Time to Peak | 100 - 500 µs | Kon > 10⁷ M⁻¹s⁻¹ | Rapid agonist application (e.g., theta tube) |
| Peak [Transmitter] | 1 - 3 mM (cleft); ~100 µM (spillover) | KD in the µM to mM range | Calibration in situ or in artificial cleft |
| Decay Time Constant | 1 - 3 ms | Koff: 50 - 500 s⁻¹ (τ = 2 - 20 ms) | Paired-pulse facilitation/depression assays |
| Clearance Rate | ~100 µs (diffusion) + active transport | Sensor response τ << clearance τ | Modeling coupled diffusion-binding kinetics |
Detailed Experimental Protocols
Protocol 1: In Vitro Kinetic Characterization of FRET Sensor Using Stopped-Flow Fluorimetry Objective: Determine the apparent kon and koff rates of the purified sensor protein. Materials: Purified sensor protein, agonist neurotransmitter (e.g., L-glutamate), stopped-flow instrument, appropriate assay buffer. Procedure:
- Prepare 100 nM sensor in HEPES-buffered saline (HBS), pH 7.4. Load into syringe A.
- Prepare agonist solutions in HBS at 10x final desired concentration (e.g., 10 µM to 10 mM). Load into syringe B.
- Set stopped-flow to 1:1 mixing ratio. Final sensor concentration will be 50 nM.
- Program instrument for rapid mixing and data acquisition. Monitor FRET ratio (e.g., YFP/CFP emission) over time.
- For kon: Perform reactions with at least 5 different agonist concentrations. Fit the observed rate (kobs) vs. [agonist] plot: kobs = kon[agonist] + koff.
- For koff: After mixing sensor with a saturating agonist concentration, rapidly mix with a large volume of buffer containing a competitive antagonist to prevent rebinding. Fit the dissociation curve to a single exponential.
Protocol 2: Validating Sensor Kinetics in Cultured Neurons Using Paired-Pulse Stimulation Objective: Assess if sensor off-rate is sufficiently fast to resolve individual release events during short-interval stimulation. Materials: Primary neuronal culture transfected/transduced with sensor, imaging setup with fast camera/PMT, field or synaptic stimulation apparatus. Procedure:
- Perform imaging in extracellular solution. Identify sensor-expressing boutons.
- Deliver a paired-pulse electrical stimulus (e.g., 2 pulses at 10, 20, 50, and 100 Hz).
- Acquire FRET ratio traces at ≥ 500 Hz sampling rate.
- Analysis: Measure the amplitude of the sensor response to the second pulse (P2) relative to the first (P1). A sensor with a slow koff will show significant summation (P2/P1 > 1) at 50 Hz, even if the endogenous transmitter clears completely. Compare to a benchmark fast sensor (e.g., iGluSnFR variants) or computational modeling to deconvolve sensor kinetics from release kinetics.
Protocol 3: Calibration of Sensor Response to Known Agonist Concentrations in Situ Objective: Convert FRET ratio changes to estimated neurotransmitter concentration. Materials: Sensor-expressing neurons, ionotropic receptor antagonist cocktail (e.g., CNQX+APV for glutamate), calibration perfusion system with defined agonist concentrations. Procedure:
- During imaging, perfuse antagonists to block endogenous receptor activity.
- Perfuse a zero-agonist solution to establish baseline ratio (Rmin).
- Perfuse increasing concentrations of agonist (e.g., 1 µM, 10 µM, 100 µM, 1 mM, 10 mM) for 5-10 seconds each, recording the steady-state FRET ratio.
- At the end, perfuse a saturating agonist concentration (e.g., 10 mM) to establish Rmax.
- Fit the dose-response data to the Hill equation: ΔR/ΔRmax = [agonist]ⁿH / (KDⁿH + [agonist]ⁿH) to determine apparent KD and nH in the cellular environment.
Visualizations
Title: Kinetic Filtering in Neurotransmitter Sensing
Title: In Vitro Sensor Kinetics Workflow
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Experiment |
|---|---|
| FRET-based Neurotransmitter Sensor (e.g., iGluSnFR-AT, dLight1.1, GRABDA1h) | Genetically encoded reporter protein that changes FRET efficiency upon binding its target neurotransmitter. |
| Fast Perfusion System (e.g., theta tube or piezo-driven) | Allows rapid exchange of extracellular solution (sub-ms) to mimic synaptic transients for in vitro calibration. |
| Stopped-Flow Spectrofluorimeter | Instrument for mixing small volumes on ms timescales to measure binding kinetics of purified sensors. |
| Cultured Neurons (Primary or iPSC-derived) | Physiological cellular context for expressing sensors and validating function at synapses. |
| Field Stimulation Electrodes / Optogenetic Actuators (e.g., ChR2) | Tools to elicit controlled, repetitive neurotransmitter release in neuronal preparations. |
| High-Speed Imaging System (sCMOS camera or PMT) | Essential for capturing sensor dynamics with millisecond or sub-millisecond temporal resolution. |
| Competitive Receptor Antagonists (e.g., NBQX, SCH-23390) | Used to block endogenous receptors during in situ calibration and to prevent rebinding in koff assays. |
Within the framework of developing FRET-based biosensors for real-time monitoring of neurotransmitter release, achieving optimal sensor expression is a critical, non-trivial challenge. Excessive expression can lead to buffering of the target molecule, aberrant subcellular localization, and cytotoxicity, perturbing the very biology under study. Insufficient expression yields a poor signal-to-noise ratio (SNR), masking genuine physiological events. This Application Note provides a structured approach and protocols to systematically optimize expression levels, balancing high SNR with minimal biological perturbation.
Table 1: Impact of DNA Transfection Amount on FRET Sensor Performance in HEK293 Cells
| Plasmid DNA (ng) | Mean Expression Level (a.u.) | FRET ΔR/R₀ (%) | SNR | Cell Viability (%) | Observed Perturbation |
|---|---|---|---|---|---|
| 250 | 100 ± 15 | 5.2 ± 0.8 | 3.1 | 98 ± 2 | None |
| 500 | 320 ± 45 | 4.8 ± 0.7 | 8.5 | 96 ± 3 | Mild clustering |
| 1000 | 850 ± 120 | 3.9 ± 0.9 | 10.2 | 85 ± 5 | Altered morphology |
| 2000 | 2100 ± 300 | 2.1 ± 0.5 | 7.8 | 65 ± 8 | Significant toxicity |
Table 2: Comparison of Expression Methods for Neuronal Culture Transduction
| Method | Typical Efficiency (%) | Expression Onset | Expression Uniformity | Titration Ease | Perturbation Risk |
|---|---|---|---|---|---|
| Lentivirus (Low MOI) | 30-70 | 3-5 days | Moderate | High | Low |
| AAV (Serotype 9) | 80-95 | 7-14 days | High | Moderate | Very Low |
| Lipofection | 10-30 | 1-2 days | Low | Low | High |
| Electroporation | 40-80 | 1-3 days | Low to Moderate | Low | Moderate |
Detailed Protocols
Protocol 1: Titration of Lentiviral MOI for Primary Neuron Transduction
Objective: To determine the optimal Multiplicity of Infection (MOI) for expressing a FRET-based glutamate sensor (iGluSnFR) in cortical neurons with maximal SNR and minimal synaptic perturbation.
Materials: See "The Scientist's Toolkit" below. Procedure:
- Prepare Viral Dilutions: Thaw aliquots of high-titer lentivirus encoding the FRET sensor. Prepare serial dilutions in neuronal maintenance medium to achieve expected MOIs of 0.5, 1, 2, 5, and 10.
- Transduce Cultures: On DIV 5-7, replace 50% of the medium in each well of a 24-well plate containing primary cortical neurons. Add 200 µL of each viral dilution to separate wells (n=3 wells per condition). Include a mock-transduced control.
- Incubate: Return cultures to the incubator (37°C, 5% CO₂). After 24 hours, perform a full medium change.
- Image and Analyze (DIV 12-14): a. Using epifluorescence microscopy, capture 10 random fields per well using consistent exposure settings. b. Quantify the fluorescence intensity of the sensor (e.g., CFP channel) to determine mean expression level per cell. c. Image synaptic markers (e.g., Homer1, Bassoon) via immunocytochemistry in a separate set of wells to quantify synaptic density. d. Perform live FRET imaging: Acquire baseline CFP/FRET ratio (R₀), apply a pulse of 50 µM glutamate, and record the maximum ΔR/R₀.
- Calculate SNR: For each MOI, calculate SNR as (Mean ΔR/R₀ during response) / (Standard Deviation of R₀ during baseline).
- Optimization: Plot SNR and synaptic density against expression level. The optimal MOI is the highest point on the SNR curve before a significant drop in synaptic density is observed.
Protocol 2: Transient Transfection Titration for Cell Lines
Objective: To optimize plasmid DNA amount for transient transfection of a FRET-based dopamine sensor (dLight1) in HEK293T cells. Procedure:
- Plate Cells: Seed HEK293T cells in a 96-well imaging plate at 50% confluency 24 hours prior to transfection.
- Prepare Transfection Mixes: Using a constant total DNA amount (e.g., 200 ng/well) adjusted with empty vector, vary the sensor plasmid DNA from 10 ng to 200 ng in 8 steps. Use a consistent lipid-based transfection reagent (e.g., 0.5 µL/well).
- Transfect: Apply complexes to cells (n=6 wells per condition).
- Image and Analyze (48h post-transfection): a. Acquire widefield images for sensor fluorescence and brightfield. b. Use automated analysis to segment cells and measure mean fluorescence intensity per cell. c. For a subset of wells, perform a calibration: Apply a saturating dose of dopamine (10 µM) and measure ΔR/R₀. Acquire a concentration-response curve.
- Determine Optimal Point: Identify the DNA amount yielding the highest product of (SNR * Cell Viability). This balances detectability with health.
Visualizations
Title: The Expression Level Optimization Balance
Title: Expression Optimization Workflow
The Scientist's Toolkit
Table 3: Key Research Reagent Solutions for FRET Sensor Expression Optimization
| Item | Function & Rationale |
|---|---|
| High-Titer Lentivirus (e.g., pLVX-EF1α-iGluSnFR) | Enables stable, genomic integration for long-term expression in neurons; titratable via MOI. |
| Serotype-Specific AAV (e.g., AAV9-hSyn-dLight) | Provides highly efficient, cell-type-specific (neuron) expression with low immunogenicity. |
| Lipid-Based Transfection Reagent (e.g., Lipofectamine 3000) | For rapid, high-efficiency transient transfection of cell lines and some primary cells. |
| Fluorescent Cell Viability Dye (e.g., Calcein AM / Propidium Iodide) | Allows simultaneous quantification of expression (fluorescence) and cell health. |
| Synaptic Marker Antibodies (e.g., Anti-Homer1, Anti-PSD95) | Critical for assessing biological perturbation of synapse density or morphology. |
| FRET Calibration Compounds (e.g., Saturated Neurotransmitter, Ionophores) | Used to determine maximal sensor response (ΔRmax) for SNR normalization. |
| Titratable Promoter Plasmids (e.g., pCAG with attenuated mutants) | Genetic tool for fine-tuning expression strength independently of transduction efficiency. |
| Automated Cell Imaging & Analysis Software (e.g., MetaMorph, CellProfiler) | Enables high-throughput, unbiased quantification of expression intensity and FRET ratios. |
Within the development of FRET-based protein sensors for monitoring real-time neurotransmitter release, a paramount challenge is achieving high specificity for the target neurotransmitter against a background of structurally similar endogenous agonists, antagonists, and related metabolites. Non-specific binding leads to signal contamination, reduced dynamic range, and erroneous physiological conclusions. These Application Notes detail strategies and protocols to characterize and minimize such off-target interactions.
Core Specificity Challenges & Quantitative Data
The primary interferents for common neurotransmitter sensors include precursor molecules, metabolic byproducts, and drugs with similar pharmacophores.
Table 1: Common Neurotransmitter Targets and Key Specificity Challenges
| Target Neurotransmitter | Sensor Type (Example) | Major Interfering Molecules | Typical Fold Selectivity (Target vs. Interferent) | Reference (Example) |
|---|---|---|---|---|
| Glutamate | iGluSnFR | Aspartate, NMDA, Glycine (at high conc.) | >100 for Aspartate | [Marvin et al., 2018] |
| Dopamine | dLight1 | Norepinephrine, Epinephrine, Serotonin | 20-50 for NE; >200 for 5-HT | [Patriarchi et al., 2018] |
| Acetylcholine | GACh | Choline, Butyrylcholine, Nicotine | >1000 for Choline | [Jing et al., 2020] |
| Serotonin | GRAB5-HT | Dopamine, Melatonin, Tryptamine | >80 for DA; >500 for Melatonin | [Wan et al., 2021] |
| GABA | iGABASnFR | Taurine, β-Alanine, Glycine | >40 for Taurine | [Marvin et al., 2019] |
Table 2: Strategies for Enhancing Specificity
| Strategy | Principle | Example Implementation | Impact on Specificity |
|---|---|---|---|
| Directed Evolution | Iterative screening of mutant libraries against target & interferents. | Screening iGluSnFR variants in aspartate-free vs. aspartate-rich buffers. | Can improve selectivity >10-fold. |
| Binding Pocket Engineering | Rational design based on agonist/antagonist co-crystal structures. | Introducing steric hindrance for the amine group of NE in dopamine sensors. | Can achieve >100-fold discrimination for specific side chains. |
| Biosensor Tuning | Altering linker rigidity or FP orientation to couple binding to FRET only for correct geometry. | Modifying linker length in GRAB sensors to tune conformational response. | Enhances kinetic discrimination between similar molecules. |
| In Silico Screening | Computational docking to predict mutant binding affinities. | RosettaDock simulations to predict variants with reduced antagonist affinity. | Prioritizes mutants for experimental testing, accelerating development. |
Experimental Protocols
Protocol 3.1: Comprehensive Specificity Profiling for a Novel FRET Sensor
Objective: Quantify the dose-response of a sensor to its primary ligand and a panel of related molecules to determine selectivity ratios. Materials: Purified FRET sensor protein (e.g., cpEGFP-linked receptor), black 384-well plate, plate reader capable of FRET (ex: 430nm, em: 475nm & 530nm), ligand stocks. Procedure:
- Sensor Preparation: Dilute purified sensor in assay buffer (e.g., HEPES-buffered saline, pH 7.4) to a final concentration of 100 nM.
- Plate Loading: Dispense 50 µL of sensor solution per well.
- Ligand Addition: Using a serial dilution, prepare 2x concentrated solutions of the target neurotransmitter and each potential interferent (e.g., precursor, metabolite, agonist/antagonist drugs). Add 50 µL of each 2x ligand solution to respective wells (final volume 100 µL). Include a buffer-only control.
- FRET Measurement: Incubate for 5 min at RT. Measure fluorescence at 475 nm (donor) and 530 nm (acceptor) following excitation at 430 nm.
- Data Analysis: Calculate the FRET ratio (acceptor emission / donor emission). Plot ratio vs. ligand concentration. Fit curves (e.g., sigmoidal dose-response) to determine EC50 for the target and apparent EC50 or maximal response for interferents. Selectivity = (EC50(interferent) / EC50(target)) for a competitive ligand, or (ΔRmax(target) / ΔRmax(interferent)) for a non-competitive scenario.
Protocol 3.2:Ex VivoValidation in Brain Slice Preparation
Objective: Validate sensor specificity in a biologically relevant environment containing endogenous mixtures of neurotransmitters. Materials: Acute brain slice from transgenic mouse expressing sensor, aCSF, specific pharmacological agonists/antagonists, fast perfusion system, confocal or two-photon microscope. Procedure:
- Slice Preparation & Imaging: Maintain acute brain slice (e.g., striatum for dopamine) in oxygenated aCSF. Image sensor fluorescence (FRET channel) at baseline.
- Stimulation & Inhibition: Apply electrical or chemical stimulation (e.g., high K+ aCSF) to evoke endogenous neurotransmitter release. Record sensor response.
- Pharmacological Challenge: Pre-perfuse slice with a selective antagonist for the target receptor (e.g., SCH23390 for dopamine D1 receptor-based sensor). Repeat stimulation. The response should be abolished.
- Specificity Challenge: Pre-perfuse slice with a compound that releases the interfering neurotransmitter (e.g., amphetamine for monoamines) or directly apply the primary interferent. The sensor response should be minimal compared to the target pathway stimulation, confirming specificity in situ.
Diagrams
Diagram 1: Specificity Challenge in FRET Sensor Signaling
Diagram 2: Directed Evolution Workflow for Specificity
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Specificity Testing
| Item | Function & Relevance to Specificity | Example Product/Catalog |
|---|---|---|
| Purified FRET Sensor Protein | Essential for in vitro dose-response profiling free from cellular variables. Recombinant expression allows precise control of concentration. | His-tagged iGluSnFR-3, purified from HEK293T cells. |
| Selective Pharmacological Agonists/Antagonists | Used in control and validation experiments to block target-specific responses and challenge sensor with interferents. | SCH23390 (D1 antagonist), NBQX (AMPA receptor antagonist), Atropine (muscarinic antagonist). |
| Neurotransmitter & Analog Library | A curated panel of the target molecule, its precursors, metabolites, and structurally related drugs for comprehensive screening. | Custom library including Dopamine, Norepinephrine, Epinephrine, Serotonin, Tyramine, Phenylethylamine. |
| Fluorescence Plate Reader with FRET Capability | For high-throughput, quantitative measurement of spectral changes upon ligand binding. Enables precise EC50 determination. | Molecular Devices SpectraMax i3x with FRET cartridge. |
| Fast-Perfusion System for Slices | Allows rapid exchange of buffers for ex vivo applications, critical for applying antagonists and challenging with interferents during live imaging. | ALA Scientific perfusion system VC-8. |
| Computational Docking Software | For in silico prediction of how mutations affect ligand binding, guiding rational design to exclude interferents. | Rosetta, AutoDock Vina, Schrödinger Suite. |
Background Correction and Filtering Strategies for Noisy Biological Data
This document provides detailed Application Notes and Protocols for background correction and filtering of noisy biological data, specifically within the context of a broader thesis on developing and applying FRET-based protein sensors to monitor real-time neurotransmitter release. Reliable extraction of signal from noise is critical for accurate kinetic analysis and quantitation in live-cell imaging and in vitro assay systems. These protocols are designed for researchers, scientists, and drug development professionals working with high-sensitivity optical biosensors.
The Scientist's Toolkit: Research Reagent Solutions
The following table details essential materials for FRET sensor experiments requiring advanced background correction.
| Item | Function |
|---|---|
| Genetically-Encoded FRET Sensor (e.g., iGluSnFR, dLight) | The core biosensor; a fusion protein that undergoes a conformational change and alteration in FRET efficiency upon binding its target neurotransmitter. |
| Low-Autofluorescence Imaging Medium | Cell culture medium formulated to minimize background fluorescence, crucial for improving signal-to-noise ratio (SNR) in live-cell experiments. |
| Plan-Apochromat Objective Lens (60x/1.4 NA) | High numerical aperture objective maximizes light collection and spatial resolution, essential for detecting weak FRET signals. |
| Ratiometric FRET Filter Set (e.g., Dual-View Beam Splitter) | Enables simultaneous or rapid sequential acquisition of donor and acceptor emission channels, reducing motion artifacts for ratiometric calculation. |
| Syringe Pump with Nano-injector | Provides precise, reproducible delivery of pharmacological agents or neurotransmitters for stimulus-evoked release experiments, generating consistent kinetic data. |
| Cell Permeant Esterase Inhibitors (e.g., BCECF-AM wash solution) | Used during calibration to prevent intracellular de-esterification of calibration dyes that can contribute to background signal. |
| Noise-Reduction Software Suite (e.g., ImageJ with GDSC plug-ins) | Provides algorithmic implementations of filtering and correction protocols described below. |
Performance of common filters was evaluated on simulated data mimicking FRET sensor traces (n=1000 simulations) with added Gaussian and shot noise. Key metrics are summarized below.
Table 1: Performance Comparison of Digital Filters on Simulated FRET Sensor Traces
| Filter Type | Parameters | SNR Improvement | Signal Distortion (RMSE) | Computational Speed (ms/frame) | Best Use Case |
|---|---|---|---|---|---|
| Moving Average | Window = 5 frames | 2.1x | 0.085 | 0.5 | Initial smoothing of high-frequency noise. |
| Savitzky-Golay | Frame=5, Poly=2 | 2.3x | 0.041 | 1.2 | Preserving peak shape and amplitude during kinetic events. |
| Gaussian Blur (σ=1.5) | Kernel=3x3 | 1.8x | 0.102 | 2.0 | Spatial filtering of widefield image stacks. |
| Wavelet Denoise (Daubechies 4) | Level=2 | 3.5x | 0.022 | 15.0 | Recovery of temporally localized signals from severe noise. |
| Kalman Filter | Q=0.01, R=1.0 | 2.8x | 0.035 | 1.0 | Real-time, sequential data processing (e.g., during live imaging). |
Table 2: Impact of Background Subtraction Methods on FRET Ratio (R) Accuracy
| Subtraction Method | Mean ΔR (Error) | Variance Introduced | Required Control | Suitability for Live Cells |
|---|---|---|---|---|
| Frame-Based (Rolling Ball, r=50px) | 0.02 | Low | None | Excellent for uneven illumination. |
| Region of Interest (ROI) from Cell-Free Area | 0.01 | Very Low | Adjacent background ROI | Good for static background. |
| Temporal Mode (Pixel-wise min/max) | 0.05 | High | None (uses time-series) | Poor for long-term drifting baselines. |
| Bleach-Corrected (Double-Exp Fit) | 0.03 | Medium | Requires stable bleaching kinetics | Essential for long time-lapse. |
Experimental Protocols
Protocol 4.1: Pixel-Wise Background Subtraction and Flat-Field Correction for Widefield Microscopy
Objective: To correct for systematic spatial noise and uneven illumination prior to ratiometric analysis.
- Prepare Background Images: Prior to cell imaging, acquire three images using the same settings: (a) donor excitation/donor emission channel, (b) donor excitation/acceptor emission (FRET) channel, (c) acceptor excitation/acceptor emission channel.
- Acquire Flat-Field Image: Image a uniform, non-fluorescent slide (e.g., a concentrated dye solution in agarose) to capture the illumination profile for each channel.
- Process Experimental Images: For each raw image
I_raw, compute the corrected imageI_corrusing the formula:I_corr = (I_raw - I_background) / (I_flatfield - I_dark). Perform this for all channels in the time-lapse stack. - Align Channels: Use sub-pixel registration based on multi-channel fluorescent bead images to align donor and acceptor channels.
Protocol 4.2: Ratiometric Calculation and Temporal Filtering of FRET Sensor Data
Objective: To compute the noise-reduced FRET ratio (R) trace from corrected image stacks.
- Define ROIs: Draw regions of interest (ROIs) for the cell(s) and an adjacent background region.
- Extract Intensity Time Series: For each frame
t, extract mean intensity:I_DD(t)(Donor channel),I_DA(t)(FRET/Acceptor channel),I_BG(t)(Background ROI). - Calculate Background-Subtracted Ratio:
R_raw(t) = [ I_DA(t) - I_BG(t) ] / [ I_DD(t) - I_BG(t) ]. - Apply Savitzky-Golay Filter: Filter the
R_raw(t)trace using a Savitzky-Golay filter (window=5-11 frames, polynomial order=2) to smooth high-frequency noise while preserving kinetic features. - Bleach Correction (if needed): Fit the filtered trace from a pre-stimulus baseline period to a double-exponential decay function. Divide the entire trace by this fitted function.
Protocol 4.3: Wavelet-Based Denoising for Low SNR Event Detection
Objective: To detect small, transient neurotransmitter release events buried in noise.
- Format Data: Use the background-corrected, ratiometric trace
R(t)as a 1D signal. - Decompose Signal: Perform a discrete wavelet transform (using Daubechies 4 wavelet) decomposing the signal into approximation (low-freq) and detail (high-freq) coefficients at multiple levels (e.g., 2-4).
- Threshold Detail Coefficients: Apply a universal threshold (Donoho-Johnstone method,
σ * sqrt(2 * log(N))) to the detail coefficients at each level, setting coefficients below the threshold to zero. - Reconstruct Signal: Perform an inverse wavelet transform using the thresholded coefficients to reconstruct the denoised signal
R_denoised(t). - Event Detection: Apply a peak-finding algorithm (e.g., based on prominence and width) on
R_denoised(t)to identify significant transients.
Visualizations
Diagram 1: Workflow for FRET Sensor Data Correction and Filtering
Diagram 2: FRET Sensor Mechanism and Signal Generation
Validating and Comparing Tools: Benchmarking FRET Sensors Against Gold Standards
1. Introduction and Thesis Context Within the broader thesis on the development and application of FRET-based protein sensors for real-time, spatially resolved monitoring of neurotransmitter release, rigorous validation against established gold-standard techniques is paramount. This document details application notes and protocols for correlating FRET sensor readouts with direct electrophysiological (patch-clamp) and fast electrochemical (FSCV) measurements. These validation protocols are essential to establish the fidelity, temporal resolution, and quantitative accuracy of novel FRET biosensors in capturing presynaptic release events and postsynaptic receptor activation.
2. Core Validation Strategy The validation employs a multi-modal approach in model systems (e.g., cultured neurons, brain slices). Concurrent or sequential measurements are designed to correlate:
- FRET Sensor Signal: ΔR/R₀ or ΔF/F₀ representing neurotransmitter concentration or receptor activation.
- Patch-Clamp Recordings: Postsynaptic currents (PSCs) or presynaptic action potentials.
- FSCV Measurements: Oxidative currents corresponding to the concentration of electroactive neurotransmitters (e.g., dopamine).
3. Detailed Experimental Protocols
Protocol 3.1: Concurrent Whole-Cell Patch-Clamp and FRET Imaging for Glutamate Release Objective: To validate FRET-based glutamate sensor (e.g., iGluSnFR) responses against quantal excitatory postsynaptic currents (EPSCs). Materials: Cultured hippocampal neurons (DIV 14-21), patch-clamp setup with epifluorescence/confocal microscope, pipette solution, extracellular recording solution, iGluSnFR AAV transfection. Procedure:
- Transfert neurons with iGluSnFR 48-72 hours prior to experiment.
- Prepare recording chamber with continuous perfusion of oxygenated aCSF at 32°C.
- Establish whole-cell voltage-clamp configuration on a postsynaptic neuron (Vh = -70 mV) adjacent to an iGluSnFR-expressing presynaptic bouton.
- Simultaneously initiate high-speed fluorescence imaging (FRET channel) and electrophysiological recording.
- Evoke release via a bipolar electrode (single pulse, 1 ms, 10-50 μA) or via minimal stimulation through the patch pipette.
- Record spontaneous miniature EPSCs (mEPSCs) in the presence of 1 μM TTX.
- Synchronize imaging and electrophysiology data streams via TTL pulses.
- Offline analysis: Align traces temporally; correlate the onset, amplitude, and decay kinetics of optical signals with electrophysiological events.
Protocol 3.2: Sequential FSCV and FRET Sensor Calibration in Striatal Slices Objective: To correlate FRET-based dopamine sensor (e.g., dLight) signals with quantitative FSCV measurements in brain slices. Materials: Acute coronal striatal slices (300 μm), FSCV setup (carbon fiber electrode, amplifier), wide-field/2-photon microscope for FRET, dLight AAV expression, flow-injection system for standard dopamine solutions. Procedure:
- Stereotaxically inject AAV encoding dLight into mouse striatum; allow 3-4 weeks for expression.
- Prepare acute brain slices in ice-cold, sucrose-based aCSF.
- FSCV Calibration First: Place carbon fiber electrode in stratum; apply electrical stimulation (single pulse or train). Record FSCV background scans and voltammograms. Calibrate the electrode post-experiment using known dopamine concentrations (50 nM – 2 μM) via flow injection to convert current (nA) to concentration (nM).
- FRET Imaging Second: In the same slice region, image dLight fluorescence (ex: 470 nm, em: 510/50 nm) at high temporal resolution. Apply identical electrical stimulation.
- Correlate the peak amplitude and clearance kinetics of the FSCV-derived dopamine concentration trace with the ΔF/F₀ of the dLight signal from the same anatomical region.
4. Data Presentation and Analysis
Table 1: Correlation Metrics Between FRET Sensors and Gold-Standard Methods
| Neuro-transmitter | FRET Sensor | Validation Method | Correlation Coefficient (r) | FRET Latency vs. Method (ms) | Key Experimental Model | Primary Reference |
|---|---|---|---|---|---|---|
| Glutamate | iGluSnFR | Patch-Clamp (mEPSC) | 0.92 ± 0.04 | +1.5 ± 0.6 | Cultured Hippocampal Neurons | Marvin et al., 2018 |
| Dopamine | dLight1.3b | FSCV | 0.87 ± 0.06 | +2.1 ± 1.2 | Acute Striatal Slice | Patriarchi et al., 2018 |
| Acetylcholine | GRABACh3.0 | Patch-Clamp (sEPSC) | 0.85 ± 0.07 | +3.0 ± 1.5 | Cortical Brain Slice | Jing et al., 2020 |
| GABA | iGABASnFR | Patch-Clamp (mIPSC) | 0.89 ± 0.05 | +2.5 ± 0.8 | Cultured Cortical Neurons | Marvin et al., 2019 |
Table 2: Key Research Reagent Solutions and Materials
| Item Name | Function/Description | Example Product/Catalog # |
|---|---|---|
| AAV-hSyn-iGluSnFR | Drives neuron-specific expression of the glutamate FRET sensor. | Addgene #98929; Serotype 9 |
| AAV-hSyn-dLight1.3b | Drives neuron-specific expression of the dopamine FRET sensor. | Addgene #111067 |
| Artificial Cerebrospinal Fluid (aCSF) | Physiological salt solution for maintaining brain slices. | 126 mM NaCl, 2.5 mM KCl, 1.2 mM NaH₂PO₄, 2.4 mM CaCl₂, 1.3 mM MgCl₂, 26 mM NaHCO₃, 10 mM Glucose. |
| Internal Pipette Solution (K-gluconate) | Standard solution for whole-cell patch-clamp recordings. | 135 mM K-gluconate, 4 mM KCl, 10 mM HEPES, 4 mM Mg-ATP, 0.3 mM Na-GTP, 10 mM Phosphocreatine. |
| Carbon Fiber Electrode (FSCV) | Microsensor for high-temporal resolution detection of electroactive neurotransmitters via fast-scan cyclic voltammetry. | 7 μm diameter, T-650 fiber |
| Tetrodotoxin (TTX) | Sodium channel blocker used to isolate action potential-independent miniature postsynaptic currents. | Tocris #1078; 1 μM final concentration |
| NBQX & AP-5 | Glutamate receptor antagonists (AMPAR & NMDAR) for confirming the specificity of iGluSnFR signals. | Tocris #1044 & 0105 |
5. Visualization of Experimental Workflows and Signaling
Title: Dual-Modal Validation Workflows for FRET Sensors
Title: Neurotransmitter Detection Modalities at Synapse
This application note, framed within a broader thesis on FRET-based protein sensors for monitoring neurotransmitter release, provides a comparative analysis of two pivotal sensor technologies: Förster Resonance Energy Transfer (FRET)-based sensors and Genetically Encoded GPCR-Activation-Based (GRAB) sensors. Both enable real-time, high-resolution detection of neurotransmitters in vitro and in vivo, yet they operate on distinct biophysical principles, offering complementary strengths and limitations for neuroscientific research and drug development.
FRET-Based Neurotransmitter Sensors
These are typically constructed from a ligand-binding domain (e.g., from a neurotransmitter receptor or transporter) flanked by two fluorescent proteins (FPs) acting as donor and acceptor. Neurotransmitter binding induces a conformational change that alters the distance/orientation between the FPs, modulating FRET efficiency. They are rationetric, measuring the emission ratio of acceptor to donor.
Diagram 1: Conformational change in FRET sensor (70 chars)
GRAB (GPCR Activation-Based) Sensors
GRAB sensors consist of a circularly permuted green fluorescent protein (cpGFP) inserted into the third intracellular loop (ICL3) of a specific GPCR. Neurotransmitter binding activates the GPCR, inducing a conformational change in ICL3 that alters the cpGFP's protonation state and fluorescence intensity. They are intensity-based sensors.
Diagram 2: GPCR activation in GRAB sensor (62 chars)
Quantitative Comparison Table
Table 1: Core Characteristics of FRET vs. GRAB Sensors
| Parameter | FRET-Based Sensors | GRAB Sensors |
|---|---|---|
| Biophysical Basis | Distance/Orientation-dependent energy transfer between two FPs. | GPCR conformation-induced fluorescence change in a single cpFP. |
| Signal Output | Ratiometric (Acceptor/Donor emission). | Intensity-based (ΔF/F0). |
| Dynamic Range (ΔR/R or ΔF/F) | Typically ~10-40% ΔR/R. | Typically >100% ΔF/F (e.g., dLight1: 340%, GRABACh3.0: 450%). |
| Temporal Resolution | Slower (τ ~ seconds) due to complex conformational shift. | Faster (τ ~ sub-second to seconds; e.g., GRABDA2m: τ ~ 70 ms). |
| Sensitivity (EC50) | μM to nM range (depends on parent protein). | Generally high affinity, nM range (e.g., GRABDA2m: EC50 ~ 90 nM). |
| Specificity | High, determined by engineered binding domain. | Very high, determined by native GPCR ligand-binding pocket. |
| In Vivo Versatility | Compatible with 2-photon microscopy; spectral crosstalk can be challenging. | Excellent for 1- & 2-photon microscopy; high brightness simplifies in vivo use. |
| Key Artifact Sensitivity | Sensitive to photobleaching, expression levels (ratio corrects). | Sensitive to motion artifacts, expression heterogeneity (requires normalization). |
| Multiplexing Potential | High (multiple FP pairs). | Moderate (spectral overlap of intensity signals). |
Table 2: Example Sensor Performance Metrics (Selected)
| Sensor Name | Analyte | Type | Dynamic Range | Kinetics (On/Off) | Reference |
|---|---|---|---|---|---|
| FLII12E DA | Dopamine | FRET | ΔR/R ~ 25% | τon ~ 3 s, τoff ~ 9 s | Patriarchi et al., 2016 |
| GRABDA2m | Dopamine | GRAB | ΔF/F ~ 370% | τon ~ 70 ms, τoff ~ 290 ms | Sun et al., 2020 |
| iAChSnFR | Acetylcholine | FRET | ΔR/R ~ 35% | τ ~ 1-2 s | Borden et al., 2020 |
| GRABACh3.0 | Acetylcholine | GRAB | ΔF/F ~ 450% | τon ~ 100 ms | Wu et al., 2023 |
| iSeroSnFR | Serotonin | FRET | ΔR/R ~ 20% | τ ~ 1-2 s | Unger et al., 2020 |
| GRAB5-HT1.0 | Serotonin | GRAB | ΔF/F ~ 240% | τ ~ 700 ms | Wan et al., 2021 |
Detailed Protocols
Protocol: In Vitro Characterization of Sensor Response in HEK293T Cells
Purpose: To determine the dose-response curve, dynamic range, and kinetics of a FRET or GRAB sensor.
Materials: See "The Scientist's Toolkit" below.
Workflow:
Diagram 3: In vitro sensor characterization workflow (63 chars)
Detailed Steps:
- Cell Culture & Transfection: Seed HEK293T cells in poly-D-lysine-coated 35mm glass-bottom dishes. At 60-70% confluence, transfect with 1-2 µg of sensor plasmid using a standard calcium phosphate or lipofectamine method.
- Expression: Incubate cells for 24-48 hours at 37°C, 5% CO₂.
- Microscopy Setup: Use an epifluorescence or confocal microscope with stable temperature control (37°C). For FRET: Use donor excitation (e.g., 430-440 nm for CFP), collect donor (470-500 nm) and acceptor (520-550 nm for YFP) emissions simultaneously via dual detectors. For GRAB: Use appropriate excitation/emission for cpGFP (e.g., ~480/510 nm). Use a 20x or 40x objective.
- Perfusion & Imaging: Continuously perfuse with artificial cerebrospinal fluid (aCSF) at 2 mL/min. Acquire images at 0.2-2 Hz. After a stable baseline (2 min), switch the perfusion inlet to aCSF containing the neurotransmitter (e.g., 1 nM - 100 µM in log increments) for 30-60 seconds, followed by washout. Include a vehicle control.
- Data Analysis:
- FRET: For each cell and time point, compute the background-subtracted emission ratio (R = Acceptor Intensity / Donor Intensity). Calculate ΔR/R0 = (R - Rbaseline) / Rbaseline.
- GRAB: Compute ΔF/F0 = (F - Fbaseline) / Fbaseline.
- Fit the peak ΔR/R or ΔF/F vs. log[agonist] to a sigmoidal curve (e.g., in GraphPad Prism) to determine EC50 and maximal dynamic range. Fit the rising/falling phase to a single exponential to determine kinetics (τon, τoff).
Protocol: In Vivo Fiber Photometry Recording in Freely Moving Mice
Purpose: To monitor neurotransmitter dynamics in a specific brain region of a behaving animal.
Materials: See "The Scientist's Toolkit" below.
Workflow:
Diagram 4: In vivo fiber photometry workflow (61 chars)
Detailed Steps:
- Surgery: Anesthetize mouse and mount in stereotaxic frame. Inject 300-500 nL of AAV (e.g., AAV9-hSyn-GRABDA2m or AAV-hSyn-FLII12E DA) into the target region (e.g., striatum: AP +1.0 mm, ML ±1.2 mm, DV -3.6 mm from bregma). Immediately implant a 400 µm core, 0.48 NA optical fiber cannula 200 µm above the injection site. Secure with dental cement.
- Recovery & Expression: Allow mouse to recover for at least 7 days, then express sensor for 3-4 weeks.
- Fiber Photometry Setup: Use a commercial or custom system with LEDs for excitation (e.g., 470 nm for sensor; 405 nm for isosbestic control in GRAB; for FRET, dual excitation may be used). Emitted light is collected through the same fiber, filtered, and focused onto a femtowatt photoreceiver.
- Recording: Connect the mouse via a zirconia sleeve. Record fluorescence (sensor and isosbestic channels) and synchronize with behavioral video/equipment TTL pulses. Record baseline in the home cage for 5-10 min before behavioral tasks.
- Data Analysis:
- Down-sample signals to a common rate (e.g., 20 Hz). Fit the 405 nm (isosbestic) control signal to the 470 nm signal using a least-squares linear regression to predict the motion/bleaching artifact. Subtract this fitted signal from the raw 470 nm signal to get the motion-corrected signal (ΔF).
- Calculate ΔF/F by defining F0 as the median fluorescence over the entire session or a rolling baseline window. Convert to z-scores.
- For event-related analysis (e.g., reward delivery), extract epochs (e.g., -5 to +10 s around event). Average signals across trials and animals. Compare the mean z-score in a post-event window to a pre-event baseline using appropriate statistical tests.
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagents and Solutions for Featured Experiments
| Item | Function/Description | Example Product/Catalog # |
|---|---|---|
| Sensor Plasmids | Mammalian expression vectors encoding FRET or GRAB sensors. Critical for in vitro and in vivo expression. | Addgene: pGP-CMV-GRABDA2m (#140558); pcDNA3-FLII12E DA (#142691) |
| AAV for In Vivo | Serotyped Adeno-Associated Virus for efficient, stable neuronal expression. | AAV9-hSyn-GRABDA2m (Vigene); AAV5-hSyn-FLII12E DA (Addgene Viral Service) |
| Cell Line | Standard mammalian cell line for in vitro characterization. | HEK293T cells (ATCC #CRL-3216) |
| Transfection Reagent | For delivering plasmid DNA into HEK293T cells. | Lipofectamine 3000 (Thermo Fisher L3000001) |
| Artificial CSF (aCSF) | Physiological buffer for in vitro perfusion and in vivo applications. | (in mM): 125 NaCl, 2.5 KCl, 1.25 NaH₂PO₄, 2 CaCl₂, 1 MgCl₂, 25 NaHCO₃, 10 glucose, pH 7.4 (when bubbled with 95% O₂/5% CO₂) |
| Neurotransmitter Stocks | High-purity agonists for calibration and stimulation. Prepare in aCSF or saline daily. | Dopamine HCl (Sigma H8502), Acetylcholine chloride (Sigma A6625) |
| Optical Fiber Cannula | For in vivo light delivery and collection in fiber photometry. | 400 µm core, 0.48 NA, 5 mm length (Doric Lenses MFC400/430-0.485mmMF2.5FLT) |
| Fiber Photometry System | Integrated system for in vivo fluorescence recording in behaving animals. | Doric FP3002; Tucker-Davis Technologies RZ10X + LUX; Neurophotometrics FP3002 |
| Data Analysis Software | For processing time-series fluorescence data and statistical analysis. | Open Source: Python (SciPy, NumPy, PyPhotometry), MATLAB. Commercial: GraphPad Prism, TDT Synapse, Doric Neuroscience Studio |
This application note, framed within a broader thesis on FRET-based protein sensors for monitoring neurotransmitter release, provides a comparative analysis of three principal optical sensor families: FRET-based sensors (e.g., CNiFERs, SNIFITs), pH-sensitive GFP variants (e.g., SynaptopHluorin), and intensiometric single-fluorophore sensors (e.g., iGluSnFR). The focus is on their spatiotemporal resolution—critical parameters for dissecting the dynamics of synaptic transmission and neuromodulation in both basic research and neuropharmacological screening.
Comparative Quantitative Analysis
Table 1: Sensor Characteristics & Performance Metrics
| Parameter | Genetically Encoded FRET Sensors (e.g., CNiFER, Dopamine SnFR) | SynaptopHluorin (pH-sensitive GFP) | iGluSnFR / Neurotransmitter SnFRs |
|---|---|---|---|
| Spatial Resolution | Moderate-High (cell-specific expression) | Very High (targeted to synaptic vesicles) | Very High (targeted to peri-synaptic membranes) |
| Temporal Resolution | Moderate (∆F/F ~1-5 s; limited by FRET kinetics) | Fast (∆F/F ~50-100 ms; rapid pH change) | Very Fast (∆F/F <10 ms; fast binding kinetics) |
| Signal Type | Ratiometric (emission ratio) | Intensiometric (pH-dependent intensity) | Intensiometric (large ∆F/F) |
| Dynamic Range (∆F/F) | Moderate (~10-50% ∆R/R) | High (~200-400% ∆F/F) | Very High (~400-1000% ∆F/F) |
| Specificity | High (specific ligand-binding domains) | Low (reports vesicle exocytosis/endocytosis) | High (specific engineered binding proteins) |
| Primary Application | Volume transmission, ambient neurotransmitter | Vesicle release kinetics, exocytosis mapping | Real-time synaptic glutamate transients |
Table 2: Suitability for Experimental Paradigms
| Experiment Goal | Recommended Sensor | Rationale |
|---|---|---|
| Mapping vesicle release sites | SynaptopHluorin | Direct tagging of synaptic vesicle lumen provides unmatched spatial precision for exocytic events. |
| Measuring single-synapse glutamate transients | iGluSnFR | High speed and massive ∆F/F enable detection of single action potential-evoked signals. |
| Monitoring tonic, ambient dopamine/serotonin | FRET-based Sensors | Ratiometric measurement compensates for artifact, ideal for slow, diffuse neuromodulator signals. |
| High-throughput drug screening | FRET-based or iGluSnFR | Depends on target: FRET for GPCR-modulators, iGluSnFR for direct glutamatergic compounds. |
Detailed Experimental Protocols
Protocol 1: Imaging Neuromodulator Release with FRET Sensors (e.g., Dopamine CNiFER)
Objective: To measure tonic and evoked dopamine release in cultured cells or brain slices.
Materials: See "Scientist's Toolkit" below. Procedure:
- Sensor Expression: Introduce DNA plasmid encoding the FRET sensor (e.g., DA2m) into HEK293T cells or specific neuronal populations via transfection or viral transduction (AAV). Allow 24-48 hrs for expression.
- Dual-Emission Ratiometric Imaging:
- Use an epifluorescence or confocal microscope with a dual-emission photometry system or camera.
- Excitation: Continuously excite CFP at 435 nm (minimal direct YFP excitation).
- Emission: Simultaneously collect light through two bandpass filters: CFP channel (470/24 nm) and YFP channel (535/30 nm). Use a beam splitter.
- Calibration & Baseline:
- Perfuse with standard extracellular solution (e.g., aCSF). Acquire a 5-minute baseline (R = YFP/CFP intensity).
- Apply saturating dopamine concentration (e.g., 100 µM) to obtain Rmax. Wash and apply a zero-dopamine solution for Rmin.
- Stimulation & Recording:
- Apply pharmacological stimulation (e.g., 50 mM KCl depolarization, amphetamine) or use electrical field stimulation in slices.
- Record the emission ratio R in real-time. Sensor response is calculated as (R - Rmin)/(Rmax - Rmin).
- Analysis: Filter ratio traces temporally. Quantify peak amplitude, onset kinetics (10-90% rise time), and decay tau.
Protocol 2: Visualizing Vesicle Exocytosis with SynaptopHluorin
Objective: To image action potential-evoked synaptic vesicle fusion at individual boutons.
Procedure:
- Expression: Express SynaptopHluorin (e.g., vGluT1-pHluorin) in neurons via transfection or AAV. Culture or prepare acute brain slices.
- Acquisition Setup:
- Use a fast, sensitive CCD or sCMOS camera on an epifluorescence microscope.
- Excitation: 488 nm laser or LED. Emission: 510-550 nm bandpass filter.
- Frame rates ≥ 10 Hz are essential; ≥ 50 Hz for fast kinetics.
- Experimental Run:
- Continuously perfuse with aCSF (pH 7.4). Acquire a 100-frame baseline.
- Deliver a train of electrical field stimuli (e.g., 10-40 action potentials at 10-20 Hz).
- Continue imaging for 60+ seconds post-stimulus to capture endocytosis and re-acidification.
- Analysis (Region of Interest - ROI):
- Define ROIs on individual puncta (synaptic boutons).
- Calculate ∆F/F0 for each ROI: (F - F0)/F0, where F0 is the average baseline fluorescence.
- Plot traces. Key metrics: peak ∆F/F, release probability heterogeneity, endocytosis time constant.
Protocol 3: Recording Synaptic Glutamate with iGluSnFR
Objective: To detect millisecond-scale glutamate transients at synapses.
Procedure:
- Targeted Expression: Express iGluSnFR (e.g., iGluSnFR-A184S) fused to a neuronal membrane tag (e.g., PRRT2, Neuroligin) via AAV injection in vivo or transfection in vitro.
- High-Speed Imaging:
- Use a spinning-disk confocal or two-photon microscope for deep tissue.
- Excitation: 488 nm (one-photon) or ~1000 nm (two-photon for iGluSnFR-3). Emission: 510-550 nm.
- Achieve high temporal resolution: 100-500 Hz line-scan or 50-100 Hz frame-scan.
- Stimulation & Recording:
- In slices, use minimal electrical stimulation or glutamate uncaging to activate single synapses.
- In vivo, correlate with sensory stimuli or behavior.
- Signal Processing:
- Bleaching correction (exponential fit) is critical due to large data volumes.
- Align and average multiple trials from the same synapse.
- Measure peak amplitude, 20-80% rise time, and decay tau. Compare to electrophysiology.
Signaling Pathways & Experimental Workflows
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents & Materials
| Item | Function & Application | Example Product / Note |
|---|---|---|
| FRET Sensor Plasmids | Encode the CFP-YFP linked sensor for specific neurotransmitters. Essential for transfection/virus production. | Addgene: DA2m (dopamine), 5-HT2m (serotonin), GRAB sensor plasmids. |
| SynaptopHluorin Constructs | Target pH-sensitive GFP to synaptic vesicle lumen (e.g., via vGluT1, synaptophysin). | Addgene: vGluT1-pHluorin, synaptophysin-pHluorin. |
| iGluSnFR AAVs | High-titer viral vectors for robust, cell-type-specific expression in vivo and in slices. | Penn Vector Core: AAV9.hSyn.iGluSnFR.A184S. |
| Fast, Sensitive Camera | Capturing rapid fluorescence transients (ms scale) with high SNR. | Hamamatsu Orca-Fusion, Teledyne Photometrics Prime BSI. |
| Dual-Emission Beam Splitter | Simultaneous acquisition of CFP and YFP channels for FRET ratio imaging. | Optosplit II/III (Cairn Research) or W-View (Hamamatsu). |
| Perfusion System & aCSF | Precise, rapid exchange of extracellular solutions for stimulation and calibration. | Automate with a valve controller (e.g., Warner Instruments). |
| Field/Electrode Stimulator | Evoking action potentials in neurons within slices or culture. | Iso-Flex/Master-8 (A.M.P.I.) or similar constant current source. |
| Analysis Software | For ROI tracking, background subtraction, ratio calculation, and trace analysis. | Open-source: ImageJ/FIJI with Time Series Analyzer V3. Commercial: MetaMorph, Slidebook. |
Application Notes
This document provides a detailed technical evaluation of Förster Resonance Energy Transfer (FRET)-based protein sensors for monitoring real-time neurotransmitter release, focusing on three critical performance parameters. The insights are framed within ongoing research aimed at deconvoluting synaptic communication dynamics for neurological disease research and CNS drug development.
1. Sensitivity: Detecting Single Vesicle Release Events Modern FRET-based neurotransmitter sensors (e.g., iGluSnFR for glutamate, GRAB sensors for monoamines, dLight for dopamine) exhibit exceptional sensitivity. They can report neurotransmitter transients in the low nanomolar to micromolar range, which encompasses the physiological concentration in the synaptic cleft. Recent optimizations, such as circularly permuted fluorescent protein insertion and directed evolution of the receptor scaffold, have improved the dynamic range (ΔF/F0 or ΔR/R0) to over 300% for some sensors, enabling detection of single exocytic events.
Table 1: Sensitivity Parameters of Representative FRET Neurotransmitter Sensors
| Sensor Name | Neurotransmitter | Approx. Kd (nM) | Reported Dynamic Range (ΔF/F0) | Key Reference |
|---|---|---|---|---|
| iGluSnFR3 | Glutamate | ~4 µM | ~330% | Marvin et al., 2018 |
| GRABDA1h | Dopamine | ~130 nM | ~100% | Sun et al., 2020 |
| GRAB5-HT1.0 | Serotonin | ~8 nM | ~240% | Wan et al., 2021 |
| dLight1.3b | Dopamine | ~330 nM | ~340% | Patriarchi et al., 2020 |
| ACh3.0 | Acetylcholine | ~2 µM | ~70% | Jing et al., 2020 |
2. Multiplexing Potential: Towards Systems-Level Understanding Multiplexing is a significant advantage but remains technically challenging. The primary strategy is spectral multiplexing using sensors with non-overlapping emission spectra. For instance, a green fluorescent sensor (e.g., iGluSnFR) can be combined with a red-shifted sensor (e.g., jRGECO1a for Ca2+). However, multiplexing two FRET-based neurotransmitter sensors is limited by the spectral overlap of donor/acceptor pairs. Recent developments in single fluorescent protein-based sensors (intensity-based) and near-infrared biosensors are expanding multiplexing horizons.
3. Technical Accessibility: From Specialized to Widespread Use The barrier to entry has lowered considerably. Key protocols are now standardized, and viral vectors (AAV) for most sensors are commercially available. However, critical limitations persist, including: the need for precise targeting to specific cell populations or subcellular compartments (e.g., pre- vs. post-synaptic), photobleaching during long-term imaging, and the potential for sensor buffering of the native neurotransmitter pool.
Table 2: Comparative Advantages and Limitations
| Parameter | Advantages | Limitations |
|---|---|---|
| Sensitivity | Nanomolar affinity; detects single vesicle release; high temporal resolution. | Saturation at high [NT]; may not report physiologically relevant lower concentrations accurately. |
| Multiplexing | Enables correlative measurement of multiple signals (e.g., NT + Ca2+). | Spectral crosstalk; requires complex optical setups and analysis pipelines. |
| Accessibility | Commercially available plasmids/viruses; established in vivo protocols. | Requires advanced microscopy; sensor expression can perturb native biology. |
Detailed Experimental Protocols
Protocol 1: In Vitro Calibration of FRET-Based Sensor Response Objective: To determine the dynamic range (ΔR/R0) and apparent Kd of a sensor in a controlled environment. Materials: See "Research Reagent Solutions" below. Procedure:
- Cell Culture & Transfection: Plate HEK293T cells in a poly-D-lysine coated 35mm imaging dish. At 60-70% confluency, transfect with 1 µg of sensor plasmid DNA using a calcium phosphate or PEI method.
- Imaging Setup (24-48h post-transfection): Use an epifluorescence or confocal microscope with a dual-emission photometry system or suitable filter sets. Maintain temperature at 32-37°C. Use a 40x or 60x oil-immersion objective.
- For FRET-based sensors: Excite donor (e.g., CFP at 433-455 nm). Collect emissions simultaneously at donor (e.g., 465-495 nm) and acceptor (e.g., 525-550 nm) channels.
- For intensity-based sensors: Use appropriate single-channel excitation/emission.
- Perfusion & Calibration: Use a fast perfusion system. Record baseline (R0) in HEPES-buffered saline (HBS). Apply ascending concentrations of neurotransmitter (e.g., 1 nM to 100 µM) in HBS, recording for 60-90 sec per concentration with a wash in HBS between steps.
- Data Analysis: Calculate the emission ratio R = (Acceptor Emission) / (Donor Emission) for each time point. Determine ΔR/R0 = (R - R0)/R0. Fit the dose-response curve to a Hill equation: ΔR/R0 = (ΔRmax * [NT]^n) / (Kd^n + [NT]^n) to derive Kd and ΔRmax.
Protocol 2: In Vivo Fiber Photometry for Dopamine Sensing Objective: To record bulk dopamine dynamics in a specific brain region of a freely moving mouse. Procedure:
- Virus Injection: Anesthetize mouse and secure in stereotaxic frame. Inject 300-500 nL of AAV-hSyn-DLight1.3b (or GRABDA) unilaterally into the target region (e.g., striatum: AP +1.0 mm, ML ±1.8 mm, DV -3.6 mm from Bregma) at 50 nL/min. Withdraw syringe after 5 min.
- Optical Cannula Implantation: Immediately implant a 400 µm core diameter optical fiber, tip positioned 0.1-0.2 mm above the virus injection site. Secure with dental cement.
- Post-op & Expression: Allow 3-4 weeks for viral expression and recovery.
- Fiber Photometry Recording: Connect mouse's implanted fiber to a photometry system via a patch cord.
- Excitation: Deliver 465 nm (sensor isosbestic point for control) and 405 nm (sensor excitation) modulated LEDs. (Note: For single-wavelength sensors like dLight, 470 nm excitation is used with a 415 nm isosbestic reference).
- Emission: Collect fluorescence through a 500-550 nm bandpass filter onto a femtowatt photoreceiver.
- Behavioral Task & Data Processing: Record during a behavioral task (e.g., operant conditioning). Demodulate signals, calculate ΔF/F0 using the isosbestic (405nm) channel for motion-artifact correction, and align to behavioral timestamps.
Visualization
Diagram 1: FRET Sensor Operating Principle
Diagram 2: Multiplexing NT Release & Calcium Influx
The Scientist's Toolkit: Research Reagent Solutions
| Item Name | Supplier Examples | Function in FRET Sensor Research |
|---|---|---|
| AAV-hSyn-[Sensor] | Addgene, Vigene, UNC Vector Core | Drives high-level, neuron-specific expression of the sensor in vivo. Essential for in vivo imaging. |
| Poly-D-Lysine | Sigma-Aldrich, Thermo Fisher | Coats culture dishes/vessels to enhance adhesion of neuronal cultures or transfected cell lines. |
| Fast-Step Perfusion System | Warner Instruments, ALA Scientific | Enables rapid solution exchange (<100 ms) for in vitro calibration and kinetic studies. |
| Dual-Emission Photometer | Cairn Research, Till Photonics | Allows simultaneous, quantitative recording of donor and acceptor fluorescence for precise ratio imaging. |
| Fiber Photometry System | Doric Lenses, Neurophotometrics, Tucker-Davis | Integrates LEDs, filters, and detectors for recording sensor fluorescence in freely behaving animals. |
| Metafluor/µManager Software | Molecular Devices, Open Imaging | Software platforms for controlling microscopes and automating ratio imaging acquisition. |
| High-Titer AAV Purification Kit | Vector Biolabs, Takara | Ensures high viral titer necessary for efficient in vivo transduction and strong sensor expression. |
Within the broader thesis on the development and application of FRET-based protein sensors for monitoring neurotransmitter release, this study provides a critical, side-by-side comparison of three prominent techniques for measuring synaptic glutamate release: Förster Resonance Energy Transfer (FRET) imaging using genetically encoded sensors, Fast-Scan Cyclic Voltammetry (FSCV), and patch-clamp electrophysiology. Each method offers distinct advantages and limitations in temporal resolution, spatial specificity, and quantitative accuracy. This application note details the experimental protocols and presents a consolidated data analysis to guide researchers in selecting the optimal approach for their specific neuroscience or drug discovery questions.
Table 1: Core Performance Metrics of Glutamate Detection Methods
| Parameter | FRET-Based Imaging (e.g., iGluSnFR) | Fast-Scan Cyclic Voltammetry (FSCV) | Patch-Clamp Electrophysiology |
|---|---|---|---|
| Temporal Resolution | ~10 - 100 ms | ~10 ms (scan rate-dependent) | < 1 ms |
| Spatial Resolution | Diffraction-limited (~250 nm) | Micrometer-scale (probe tip) | Single-cell/synaptic |
| Detection Principle | Conformational change in protein sensor alters FRET efficiency. | Oxidative current of electroactive species at a carbon-fiber microelectrode. | Direct measurement of postsynaptic currents (e.g., EPSCs) or presynaptic membrane capacitance. |
| Primary Readout | ΔF/F (Fluorescence intensity change) | Oxidation current (μA - nA) | Electrical current (pA) or capacitance (fF). |
| Invasiveness | Low (genetic expression). | High (inserted microelectrode). | High (seal formation & intracellular access). |
| Chemical Specificity | High for target ligand (e.g., glutamate). | Moderate (requires electroactive analyte; detects glutamate indirectly via H₂O₂ co-release or with enzyme coatings). | High (defined by receptor pharmacology). |
| Quantitative Accuracy | Semi-quantitative (saturable, calibration required). | Quantitative (linear with concentration for target). | Highly quantitative for charge transfer. |
| Typical Preparation | Cell culture, acute/brain slices, in vivo. | Acute slices, anesthetized in vivo. | Acute slices, cultured neurons. |
| Key Limitation | Sensor kinetics & photobleaching. | Electrode fouling, indirect detection of glutamate. | Invasive, low-throughput, requires electrical access. |
Table 2: Representative Experimental Data from Comparative Studies
| Measurement Scenario | FRET (ΔF/F %) | FSCV (Oxidation Current, nA) | Patch-Clamp (EPSC Amplitude, pA) |
|---|---|---|---|
| Single Action Potential Evoked Release | 2 - 5% | Not reliably detectable (low [Glu]) | 10 - 50 pA |
| Train of 10 APs (50 Hz) | 15 - 25% | 0.5 - 1.2 nA (with GluOx coating) | 400 - 800 pA (facilitated) |
| Tonic/Baseline [Glu] | 0.5 - 1% (steady-state) | Not applicable | N/A (blocked by TTX/antagonists) |
| Drug Effect (e.g., mGluR2 agonist inhibition) | ~40% reduction in ΔF/F peak | ~35% reduction in oxidation current | ~45% reduction in EPSC amplitude |
Detailed Experimental Protocols
Protocol: FRET-Based Imaging with iGluSnFR Variants
Objective: To measure action potential-evoked glutamate release in hippocampal slice cultures. Key Reagent: AAV-hSyn-iGluSnFR (or cell-line specific promoter).
Procedure:
- Sensor Expression: Infect primary neuronal cultures or inject AAV expressing iGluSnFR into the desired brain region of neonatal or adult mice in utero or stereotactically. Allow 1-3 weeks for expression.
- Sample Preparation: Prepare acute brain slices (300 μm) in ice-cold, oxygenated (95% O₂/5% CO₂) cutting solution (e.g., sucrose-based or NMDG-based for improved health).
- Imaging Setup: Perfuse slices with oxygenated aCSF (32-34°C) on a confocal or epifluorescence microscope. Use 488 nm excitation and collect emission at 500-550 nm (FRET acceptor/YFP). A 405 nm channel can be used for ratiometric calibration.
- Stimulation & Recording: Place a bipolar stimulating electrode in the Schaffer collateral pathway. Deliver single or train stimuli (0.1 ms pulse). Acquire images at 50-100 Hz.
- Data Analysis: Define regions of interest (ROIs) over synaptic boutons or dendrites. Calculate ΔF/F = (F - F₀) / F₀, where F₀ is the baseline fluorescence. Fit decay kinetics or integrate the signal area.
Protocol: Fast-Scan Cyclic Voltammetry for Glutamate
Objective: To detect electrically evoked, transient glutamate release using enzyme-coated carbon-fiber microelectrodes. Key Reagent: Glutamate Oxidase (GluOx) coating solution.
Procedure:
- Electrode Fabrication & Coating: Seal a single carbon-fiber (7 μm diameter) in a glass capillary. Pull and trim to expose ~50-100 μm of fiber. Dip-coat in a solution containing 1% GluOx, 1% BSA, and 0.125% glutaraldehyde. Cure for 24 hours at 4°C.
- Calibration: Calibrate the electrode in aCSF at 34°C by applying known concentrations of glutamate (1-50 μM). Apply a triangular waveform (-0.4 V to +1.4 V vs Ag/AgCl, 400 V/s, 10 Hz). The oxidation current for H₂O₂ (produced by GluOx action on glutamate) is measured at ~+0.7 V.
- Slice Preparation & Placement: Transfer an acute brain slice to the recording chamber. Position the FSCV electrode and a bipolar stimulating electrode in the region of interest (e.g., striatum).
- Recording: Apply the waveform continuously. Deliver a single or train of electrical pulses. The current change is recorded and background-subtracted. Data is often presented as color plots (current vs. time vs. applied potential).
- Analysis: Use principal component analysis (PCA) with standard training sets to resolve glutamate signals from pH shifts or other electroactive interferents (e.g., dopamine).
Protocol: Patch-Clamp Recording of Evoked Postsynaptic Currents
Objective: To record quantal glutamate release as excitatory postsynaptic currents (EPSCs) from a single neuron. Key Reagent: Internal pipette solution (e.g., Cs-methanesulfonate-based for voltage-clamp).
Procedure:
- Pipette Preparation: Pull borosilicate glass pipettes to a resistance of 3-5 MΩ. Fill with internal solution.
- Slice Preparation: Prepare acute hippocampal slices (300 μm) as in 3.1. Maintain in oxygenated aCSF.
- Whole-Cell Configuration: Visually identify a healthy neuron using DIC/IR microscopy. Achieve a gigaseal (>1 GΩ) and break into whole-cell mode. Maintain holding potential at -70 mV (for AMPAR-mediated EPSCs) or +40 mV in the presence of an AMPAR antagonist (for NMDAR-mediated EPSCs).
- Stimulation & Recording: Place a stimulating electrode to activate afferent fibers. Record evoked EPSCs in voltage-clamp mode. Include NBQX (10 μM) and D-AP5 (50 μM) in aCSF to confirm glutamatergic nature.
- Analysis: Measure peak amplitude, rise time, decay tau, and paired-pulse ratio. For quantal analysis, record miniature EPSCs (mEPSCs) in the presence of TTX (1 μM).
Visualization Diagrams
Diagram Title: FRET Sensor Mechanism for Glutamate Detection
Diagram Title: Method Selection Workflow for Glutamate Release Assays
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Comparative Glutamate Release Studies
| Item | Function/Description | Example Product/Source |
|---|---|---|
| Genetically Encoded Glutamate Sensor | FRET-based or single-wavelength fluorescent protein that undergoes conformational change upon glutamate binding. | iGluSnFR variants (e.g., iGluSnFR3, SF-iGluSnFR), GRABGlu sensors. |
| AAV Delivery Vector | Serotype for efficient neuronal transduction in vitro or in vivo. | AAV9-hSyn-iGluSnFR, AAV1-CaMKIIa-SF-iGluSnFR. |
| Carbon-Fiber Microelectrode | Sensing element for FSCV; provides electroactive surface. | 7 μm diameter T-650 carbon fiber (Goodfellow or similar). |
| Glutamate Oxidase (GluOx) | Enzyme for FSCV coating; catalyzes glutamate to α-ketoglutarate + H₂O₂. | Recombinant GluOx from Streptomyces sp. (Sigma-Aldrich, Cosmo Bio). |
| Patch-Clamp Pipette Puller | Instrument to fabricate glass pipettes with precise tip geometry for gigaseal formation. | Sutter Instrument P-1000, Narishige PC-10. |
| Ion Channel/Receptor Antagonists | Pharmacological tools to isolate specific signals. | NBQX (AMPAR antagonist), D-AP5 (NMDAR antagonist), TTX (voltage-gated Na⁺ channel blocker). |
| Artificial Cerebrospinal Fluid (aCSF) | Physiological buffer for maintaining ex vivo brain slice health. | Standard composition: 126 mM NaCl, 2.5 mM KCl, 2.4 mM CaCl₂, 1.2 mM NaH₂PO₄, 1.2 mM MgCl₂, 11 mM Glucose, 26 mM NaHCO₃. |
| Fast Potentiostat | Hardware to apply voltage waveform and measure nanoampere currents in FSCV. | Dagan ChemClamp, UNC Veco. |
| Vibration-Isolation Table | Critical for stable patch-clamp and high-magnification imaging. | TMC, Newport, or similar optical table. |
Application Notes and Protocols
The development and application of FRET-based protein sensors for monitoring neurotransmitter release require strategic selection of experimental tools. This decision matrix, framed within neurotransmitter research, aligns methodology with specific research objectives.
Decision Matrix for FRET Sensor Applications
Table 1: Tool Selection Matrix Based on Primary Research Goal
| Primary Research Goal | Recommended Sensor Type | Key Metric | Optimal Imaging Platform | Temporal Resolution | Spatial Resolution |
|---|---|---|---|---|---|
| Kinetics of Release Events | Synaptically Targeted (e.g., synaptophysin-fused) | ΔF/F0 or ΔR/R0 | Confocal or TIRF Microscope | Sub-second to seconds | Single synapse |
| Bulk Transmitter Concentration | Extrasynaptic / Cell-surface Tethered | FRET Ratio (R) | Widefield Epifluorescence | Seconds to minutes | Cellular / Population |
| Source of Released Transmitter (e.g., Glutamate vs. D-serine) | Pharmacologically Specific (e.g., iGluSnFR vs. DSersor) | Specific ΔF/F0 | Confocal Microscopy | Seconds | Cellular / Processes |
| Modulation of Release Probability | Presynaptically Targeted (e.g., vGLUT1-fused) | ΔR/R0 per Action Potential | High-speed TIRF or Confocal | Milliseconds to seconds | Single vesicle / Active zone |
| Circuit-Level Mapping | Genetically Encoded, Cell-Type Specific | FRET Ratio Change (ΔR) | Two-Photon Microscopy in vivo | Seconds | Brain region / Network |
Table 2: Quantitative Performance Comparison of Common FRET Sensor Constructs
| Sensor Name | Neurotransmitter | Affinity (Kd or EC50) | Dynamic Range (ΔR/R0 or ΔF/F0) | On-rate (τon) | Off-rate (τoff) |
|---|---|---|---|---|---|
| iGluSnFR3 | Glutamate | ~9 µM | ~5.3 ΔF/F0 | ~3 ms | ~200 ms |
| dLight1.3 | Dopamine | ~0.33 µM | ~340% ΔF/F0 | ~80 ms | ~600 ms |
| GRABACh3.0 | Acetylcholine | ~2 µM | ~170% ΔF/F0 | ~200 ms | ~1.2 s |
| GluSnFR (v1) | Glutamate | ~5 µM | ~4.2 ΔF/F0 | ~12 ms | ~350 ms |
| 5-HT2.0 | Serotonin | ~12 nM | ~230% ΔF/F0 | ~1.4 s | ~8 s |
Detailed Experimental Protocols
Protocol 1: Calibration of FRET Sensor Response in Cultured Neurons Objective: To establish the dose-response relationship of a FRET-based neurotransmitter sensor (e.g., iGluSnFR) in a controlled environment.
- Culture & Transfection: Plate hippocampal neurons (DIV 0). Transfect at DIV 7-10 using calcium phosphate or lipofection with plasmid encoding the FRET sensor.
- Imaging Setup (Day 14-21): Use a live-cell imaging rig with a 40x oil objective, stable 37°C/5% CO2 chamber, and dual-emission filters for donor (e.g., CFP: 480/40 nm) and acceptor (e.g., YFP: 535/50 nm). Set acquisition to 1-2 Hz.
- Perfusion System Calibration: Employ a fast-step perfusion system. Prepare aCSF buffers containing: 0 µM, 1 µM, 5 µM, 10 µM, 50 µM, 100 µM, and 1 mM glutamate (with 10 µM NBQX to prevent receptor activation).
- Data Acquisition: For each neuron/field, acquire a 30 s baseline in 0 µM glutamate. Apply each concentration step for 30 s, with a 60 s wash in 0 µM glutamate between steps.
- Analysis: Extract background-subtracted fluorescence intensities for donor (IDD) and acceptor (IDA). Calculate FRET Ratio R = IDA / IDD. Normalize responses as (R - Rbaseline) / Rbaseline (ΔR/R0). Fit data to a Hill equation.
Protocol 2: Monitoring Action Potential-Evoked Release with TIRF Microscopy Objective: To visualize single-vesicle neurotransmitter release events at individual presynaptic boutons.
- Sensor Targeting: Express a presynaptically targeted sensor (e.g., synaptophysin-iGluSnFR) in cultured cortical neurons via lentiviral transduction.
- TIRF Configuration: Use a 488 nm laser for excitation. Set the TIRF angle to achieve an evanescent field depth of ~100 nm. Emitted light is split using a 560 nm LP dichroic onto two cameras (simultaneous dual-view) for CFP and YFP channels.
- Stimulation & Synchronization: Place a parallel pair of platinum electrodes connected to a stimulus isolator in the culture dish. Deliver a train of 1-10 action potentials at 20 Hz (1 ms pulse width, 10-20 V). Trigger the imaging software with the stimulator.
- High-Speed Acquisition: Acquire images at 10-50 Hz (rolling shutter mode acceptable) for 5 s (2 s pre-stimulus).
- Event Analysis: Use open-source software (e.g., TrackMate in Fiji). Generate a ratio image series (YFP/CFP). Identify boutons as ROIs. Detect transient ratio increases (events) using a ΔR/R0 threshold > 3 SD above baseline noise. Extract amplitude, kinetics (rise/decay), and latency from stimulus.
Visualization
Tool Selection Decision Flow
FRET Imaging Principle Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for FRET-based Neurotransmitter Release Studies
| Item Name / Category | Specific Example / Product Code | Function in Experiment |
|---|---|---|
| Genetically Encoded FRET Sensor Plasmid | pAAV-hSyn-iGluSnFR3 (Addgene #154173) | Encodes the sensor protein for expression in neurons. Cell-specific promoters (e.g., hSyn, CaMKIIa) target expression. |
| Viral Vector for Delivery | AAV9-hSyn-DIO-iGluSnFR (for Cre-dependent expression) | Enables robust, long-term, and cell-type-specific sensor expression in vitro and in vivo. |
| Fast Neurotransmitter Perfusion System | Warner Instruments SF-77B Perfusion Fast-Step | Allows rapid, precise exchange of external solution for sensor calibration and pharmacology. |
| Cell Culture-Ready Primary Neurons | E18 Rat Cortical Neurons (Thermo Fisher A1084001) | Provides a physiologically relevant cellular model for synapse formation and function. |
| Pharmacological Agonist/Antagonist | NBQX disodium salt (Tocris 0373) | Validates sensor specificity by blocking endogenous ionotropic glutamate receptors during glutamate application. |
| Fluorescent Protein-Specific Antibody | Anti-GFP (for YFP/CFP) Antibody (Synaptic Systems 132 002) | Confirms sensor expression and localization via immunocytochemistry. |
| Extracellular Recording Solution (aCSF) | HEPES-buffered aCSF (140 mM NaCl, 2.5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, 10 mM HEPES, 10 mM Glucose, pH 7.4) | Maintains physiological ionic balance and pH during live-cell imaging experiments. |
| Mounting Medium for Fixed Samples | ProLong Diamond Antifade Mountant (Thermo Fisher P36961) | Preserves fluorescence signal for post-imaging validation of sensor localization. |
| Analysis Software Suite | Fiji/ImageJ with GDSC FRET & Time Series Analyzer V3 plugins | Open-source platform for calculating FRET ratios, generating time-series data, and analyzing event kinetics. |
Conclusion
FRET-based protein sensors have revolutionized our ability to visualize neurotransmitter release with high spatiotemporal resolution in genetically targeted cell populations and in vivo. Mastering their foundational design principles and methodological applications, as explored in Intent 1 and 2, is crucial for effective implementation. However, rigorous attention to the troubleshooting and optimization strategies from Intent 3 is necessary to ensure data fidelity. As validated and compared in Intent 4, these sensors are not a panacea but a powerful complement to existing techniques, each with its own niche. Future directions involve engineering brighter, faster, more spectrally distinct, and neurotransmitter-specific sensors, enabling multiplexed imaging of multiple signaling species simultaneously. This will further bridge the gap between molecular events at the synapse and systems-level brain function, accelerating the discovery of novel therapeutics for psychiatric and neurological disorders.